Clear Sky Science · en
ACACA modulates R-loop homeostasis to enhance lipid metabolism and microenvironmental interactions in ccRCC
Why this kidney cancer story matters
Clear cell kidney cancer is notorious for being both common and hard to treat once it spreads. One of its hallmarks is that tumor cells become remarkably fatty, stockpiling lipids and rewiring how they fuel themselves. This study asks a surprising question: how are changes in a cell’s genetic stress signals connected to this oily makeover and to the way tumors talk with their surroundings? By following a single enzyme, ACACA, the researchers uncover a molecular link between genome stress, altered fat metabolism, and the growth-friendly neighborhood that forms around kidney tumors.
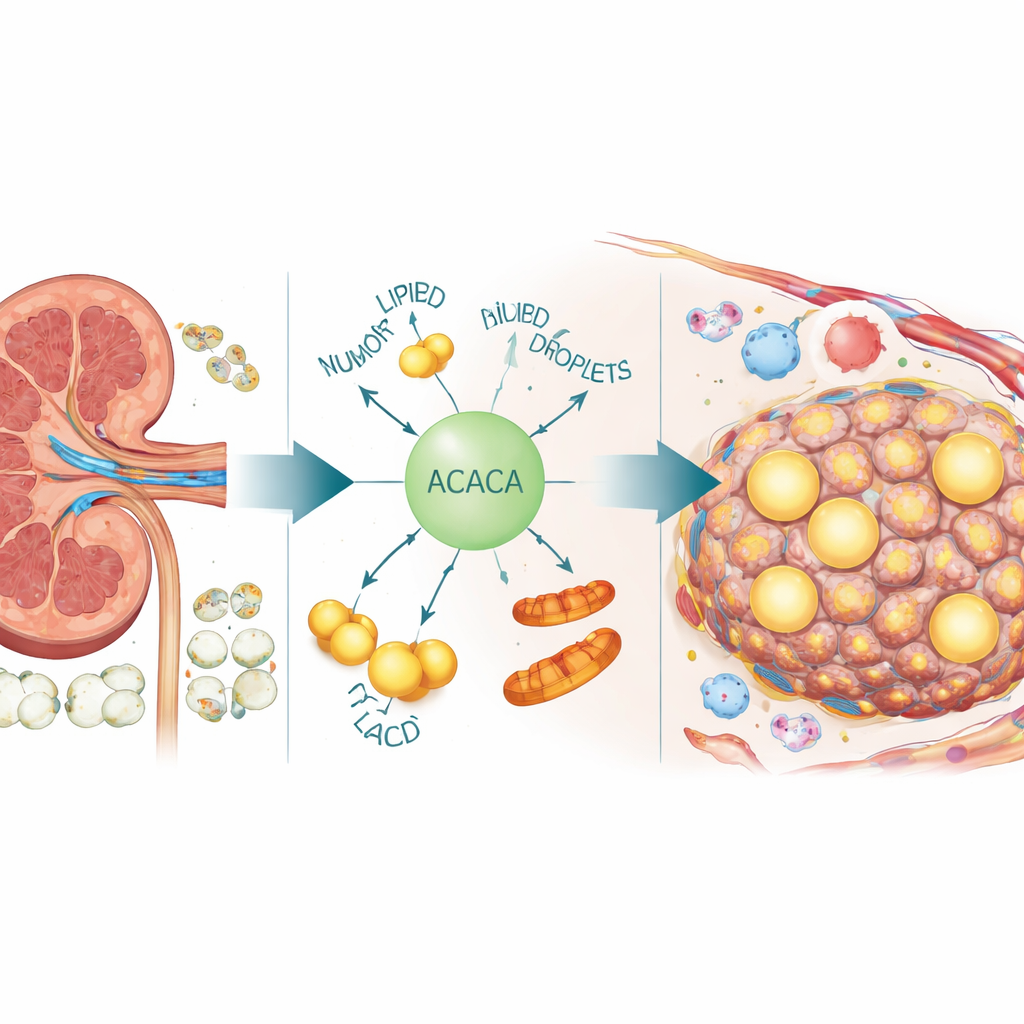
Hidden three-stranded knots in tumor DNA
Inside our cells, DNA and RNA can briefly form three-stranded knots called R-loops during the process of reading genes. In moderation, these structures help regulate the genome, but in excess they can stall DNA copying and trigger damage. Using large public datasets of kidney tumors and healthy tissue, the team built a score that reflects how active R-loop-related genes are in each patient. They found that this activity is clearly higher in clear cell kidney cancers than in normal kidney tissue and climbs even further in advanced tumors and those that have spread. Patients whose tumors showed stronger R-loop signatures generally had poorer survival, suggesting that disrupted control of these structures goes hand-in-hand with more aggressive disease.
A single enzyme stands out from the crowd
From more than a thousand R-loop-associated genes, the researchers narrowed the field to 44 that were both misregulated in tumors and linked to patient outcomes. They then used several linear machine-learning models to see which combination best predicted survival. Across methods and across independent patient cohorts, one gene kept rising to the top: ACACA, a key enzyme that drives the first committed step in making new fatty acids. High ACACA levels helped identify patients at higher risk of relapse or death. When the team combined ACACA expression with standard clinical information—tumor size, spread, and microscopic grade—they could build a practical scoring tool that closely matched actual patient survival over many years.
A fatty hub inside malignant cells
To see where ACACA sits in the tumor ecosystem, the authors turned to single-cell and spatial transcriptomics, technologies that read gene activity one cell at a time and map it back onto tissue slices. These analyses showed that ACACA is not evenly expressed: it is concentrated in malignant cells, where it coincides with active cell-division programs and signs of DNA damage and repair. ACACA-high cancer cells appeared as communication hubs, sending and receiving many signals from nearby immune cells and blood vessel cells. Many of these signals travel through lipid-related pathways involving ANGPTL molecules, which are known to coordinate fat handling with inflammation and blood vessel growth. In tissue sections, regions rich in ACACA overlapped with dense tumor areas rather than with surrounding normal tissue.
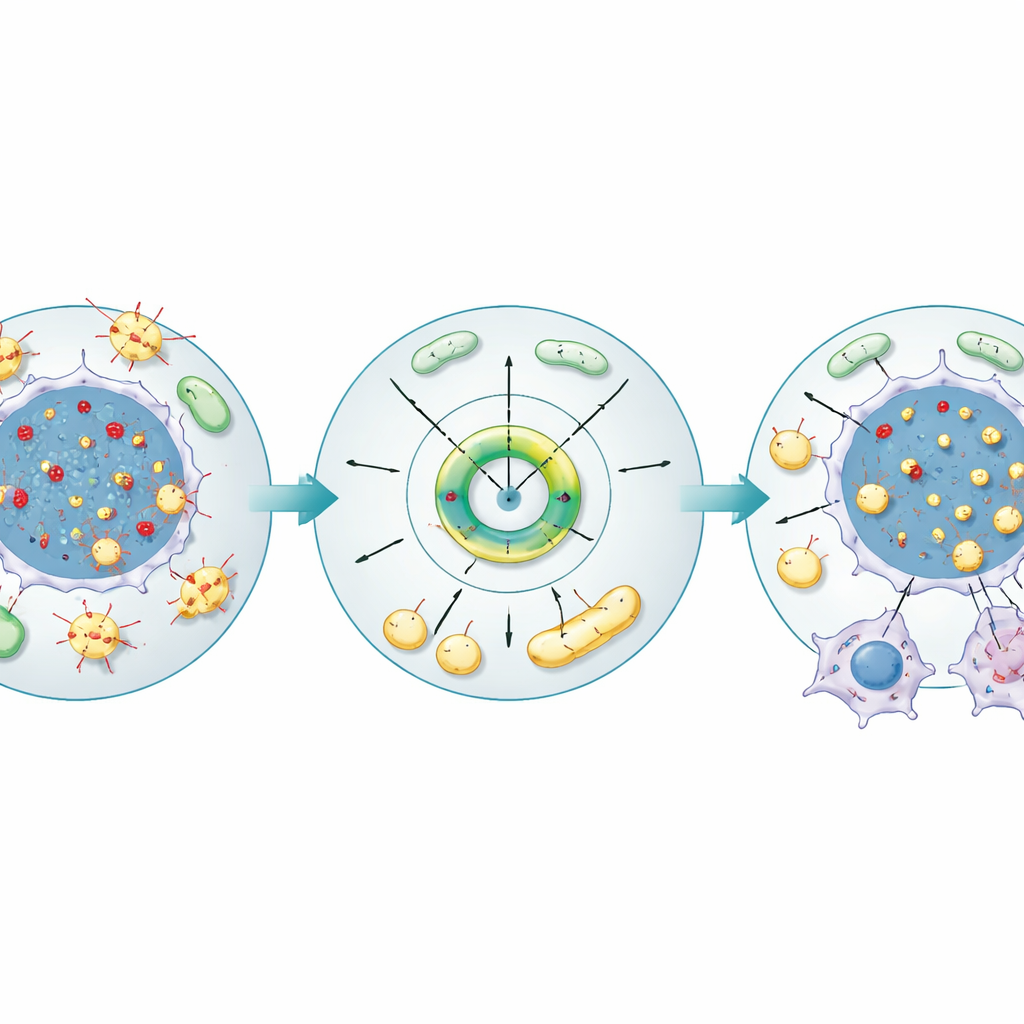
From genetic stress to fuel supply and tumor growth
The authors then tested ACACA directly in kidney cancer cell lines and in mouse tumors. Turning down ACACA slowed cell growth, reduced movement, and increased cell suicide, while turning it up had the opposite effect. Microscopy showed that low ACACA led to a buildup of R-loops and more marks of DNA injury, whereas high ACACA reduced these structures. At the same time, ACACA boosted the number of lipid droplets inside cells, raised levels of fatty acids and triglycerides, and improved mitochondrial health, as judged by stronger membrane potential, more mitochondria, and lower levels of reactive oxygen molecules. In mice, blocking ACACA in implanted kidney cancer cells curtailed tumor growth, increased DNA damage signals, diminished lipid stores, and weakened mitochondrial function, tying the enzyme to both genome stability and the tumor’s energy budget.
What this means for future treatment
Taken together, the study paints ACACA as a molecular switch that helps clear cell kidney cancers survive under genetic stress by reshaping fat metabolism and strengthening their interactions with the surrounding microenvironment. Elevated R-loop activity marks more dangerous tumors, and ACACA emerges as a central player that connects this stress to lipid-rich, energy-efficient cancer cells that grow and spread more readily. Because ACACA is an enzyme that can, in principle, be targeted by drugs, these findings suggest new ways to both predict which patients are at highest risk and to design therapies that simultaneously disrupt the tumor’s genome-protection tactics and its metabolic lifelines.
Citation: Zhang, D., Chen, X., He, X. et al. ACACA modulates R-loop homeostasis to enhance lipid metabolism and microenvironmental interactions in ccRCC. npj Precis. Onc. 10, 102 (2026). https://doi.org/10.1038/s41698-026-01319-y
Keywords: clear cell renal cell carcinoma, R-loops, ACACA, lipid metabolism, tumor microenvironment