Clear Sky Science · en
Y chromosome-linked EIF1AY deletion drives sex differences in multiple myeloma
Why some blood cancers hit men harder
Multiple myeloma is a cancer of antibody-making cells in the bone marrow. Around the world, men develop and die from this disease more often than women, but the reasons have been unclear. This study digs into one surprising suspect: the Y chromosome. The authors uncover how the loss of a single Y‑linked gene, called EIF1AY, helps tip the immune environment in men toward supporting tumor growth, revealing a potential new target for more precise treatment.
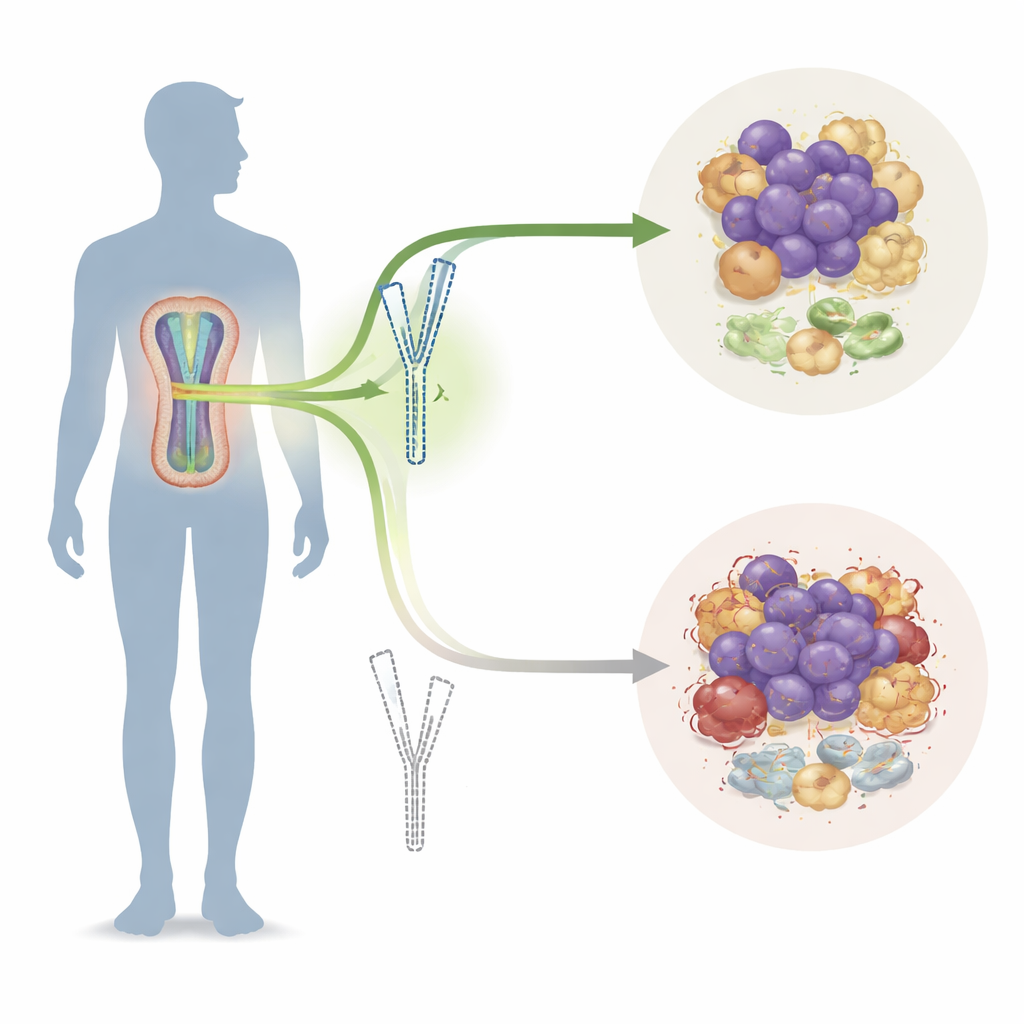
A sex-linked clue hidden in tumor DNA
Researchers first confirmed that men with multiple myeloma tend to do worse than women, with higher incidence, more advanced disease at diagnosis, and shorter survival. They then scanned large genetic and clinical databases to look for genes that differed between male and female patients. Among many candidates, three Y‑linked genes stood out as being lower in men with aggressive disease, with EIF1AY emerging as the strongest predictor of poor outcome. In blood and bone marrow samples from male patients, EIF1AY was often partially deleted from the Y chromosome, especially in older men and in those with advanced-stage myeloma and complex chromosomal damage. Patients with lower EIF1AY levels had higher tumor burden and worse responses to therapy, marking this gene as a key protective factor that some tumors manage to shed.
How immune cells can be turned into helpers of cancer
The team next asked how EIF1AY affects the tumor’s surroundings, especially macrophages—immune cells that can either attack cancer (the “M1” state) or support it (the “M2” state). Analyses of patient datasets showed that male myeloma samples were richer in M2‑like macrophages than female samples. In laboratory models, myeloma cells engineered to make more EIF1AY grew more slowly in dishes and in mice. Their surrounding macrophages were less likely to adopt the tumor‑supporting M2 program and were recruited into tumors less efficiently. When EIF1AY was reduced or lost, the opposite occurred: more M2‑polarized macrophages gathered at tumor edges and within the cancer mass, and these conditioned macrophages in turn released factors that drove myeloma cells to proliferate. In essence, EIF1AY acted as a brake on a vicious partnership between cancer cells and macrophages.
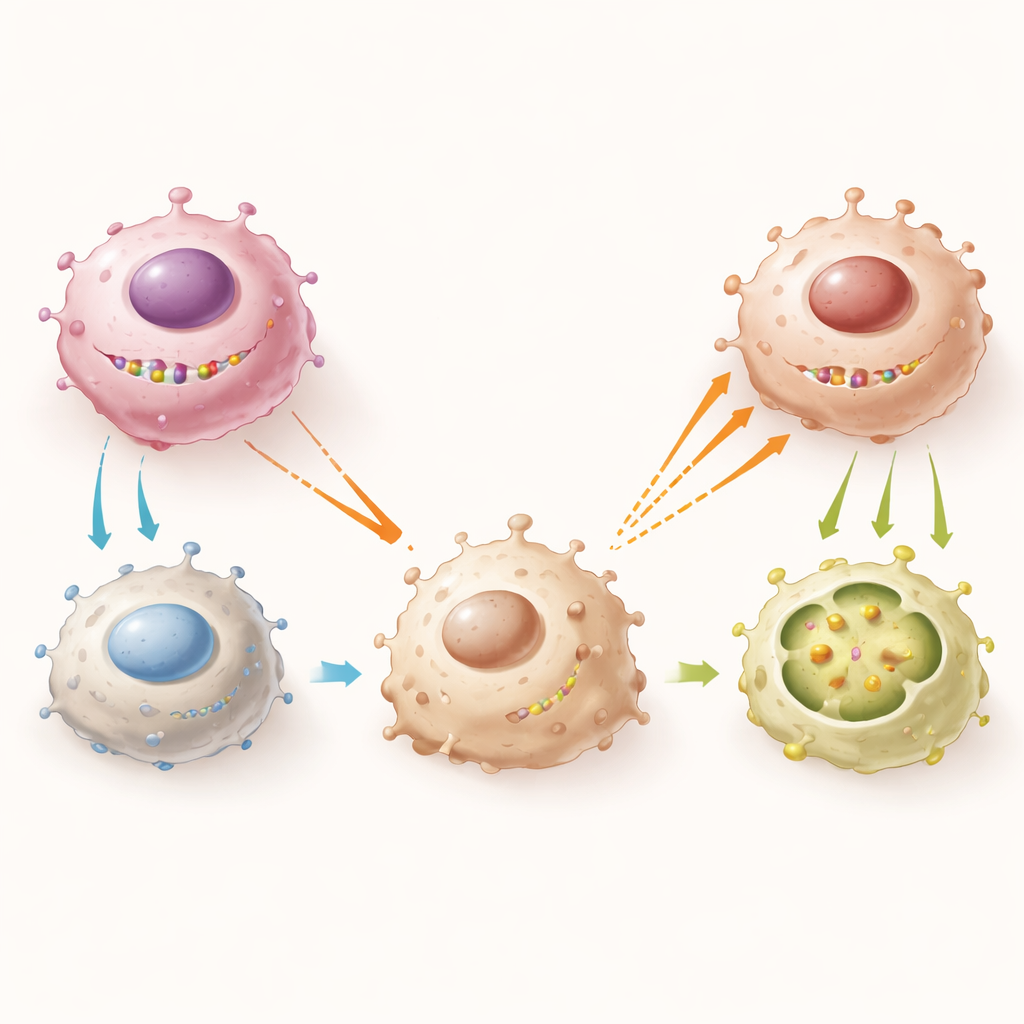
A molecular chain reaction from tumor cell to macrophage
Diving deeper, the authors mapped the molecular chain that links a Y‑chromosome gene inside a tumor cell to the behavior of nearby immune cells. EIF1AY teams up with another Y‑linked protein, RPS4Y1, to form a complex that physically binds and stabilizes the messenger RNA for a surface receptor called CD134 on myeloma cells. When EIF1AY and RPS4Y1 are present, CD134 is maintained at higher levels. This, in turn, keeps the tumor cells from releasing large amounts of two signaling proteins, IL‑4 and IL‑13. Low IL‑4 and IL‑13 mean that macrophages express less of a collagen‑sensing receptor called DDR1, are less likely to become M2‑like, and are less drawn into the tumor. When EIF1AY is deleted, this entire axis collapses: CD134 levels fall, IL‑4 and IL‑13 rise, DDR1 increases on macrophages, and more M2 macrophages flood in to nurture the cancer.
Linking male and female versions of the same safeguard
Interestingly, women do not carry EIF1AY or RPS4Y1, but they do have closely related genes on the X chromosome, EIF1AX and RPS4X. The study found that high levels of these X‑linked counterparts in female myeloma patients were also associated with better outcomes. When the researchers experimentally reduced EIF1AX or RPS4X in female‑derived myeloma cell lines, the cells grew faster, mirroring what happens in males when EIF1AY and RPS4Y1 are lost. This suggests that both sexes possess a similar protective pathway, but men are uniquely vulnerable because the Y chromosome can be partially lost in aging blood cells, removing EIF1AY and RPS4Y1 altogether, whereas the X‑linked genes in women are more stably maintained.
What this means for patients and future treatments
In clear terms, this work shows that many male myeloma patients lose a protective circuit that normally tells tumor cells to keep quiet and stops nearby macrophages from turning into cancer accomplices. When the EIF1AY–RPS4Y1–CD134 pathway is intact, it keeps certain immune signals low, prevents macrophages from adopting a tumor‑friendly identity, and slows myeloma growth. When parts of the Y chromosome—and EIF1AY in particular—are deleted, that brake is lifted, allowing a self‑reinforcing loop of inflammatory signals and supportive macrophages that accelerates disease. These findings help explain why men fare worse than women in multiple myeloma and suggest that measuring EIF1AY status, or restoring its downstream pathway, could guide more tailored immunotherapies for male patients.
Citation: Feng, Z., Bai, J., Li, Y. et al. Y chromosome-linked EIF1AY deletion drives sex differences in multiple myeloma. npj Precis. Onc. 10, 131 (2026). https://doi.org/10.1038/s41698-026-01317-0
Keywords: multiple myeloma, Y chromosome, tumor microenvironment, macrophage polarization, sex differences in cancer