Clear Sky Science · en
AI-driven virtual screening platform identifies novel NSUN2 inhibitor candidates for targeted cancer therapy: a computational drug discovery approach
New Ways to Outsmart Stubborn Cancers
Many cancers become deadly not just because they start, but because they learn to evade our best drugs. This study explores a promising new way to shut down a key helper of tumor growth, using artificial intelligence to sift through more than one hundred million potential medicines on a computer before any are mixed in a real test tube.
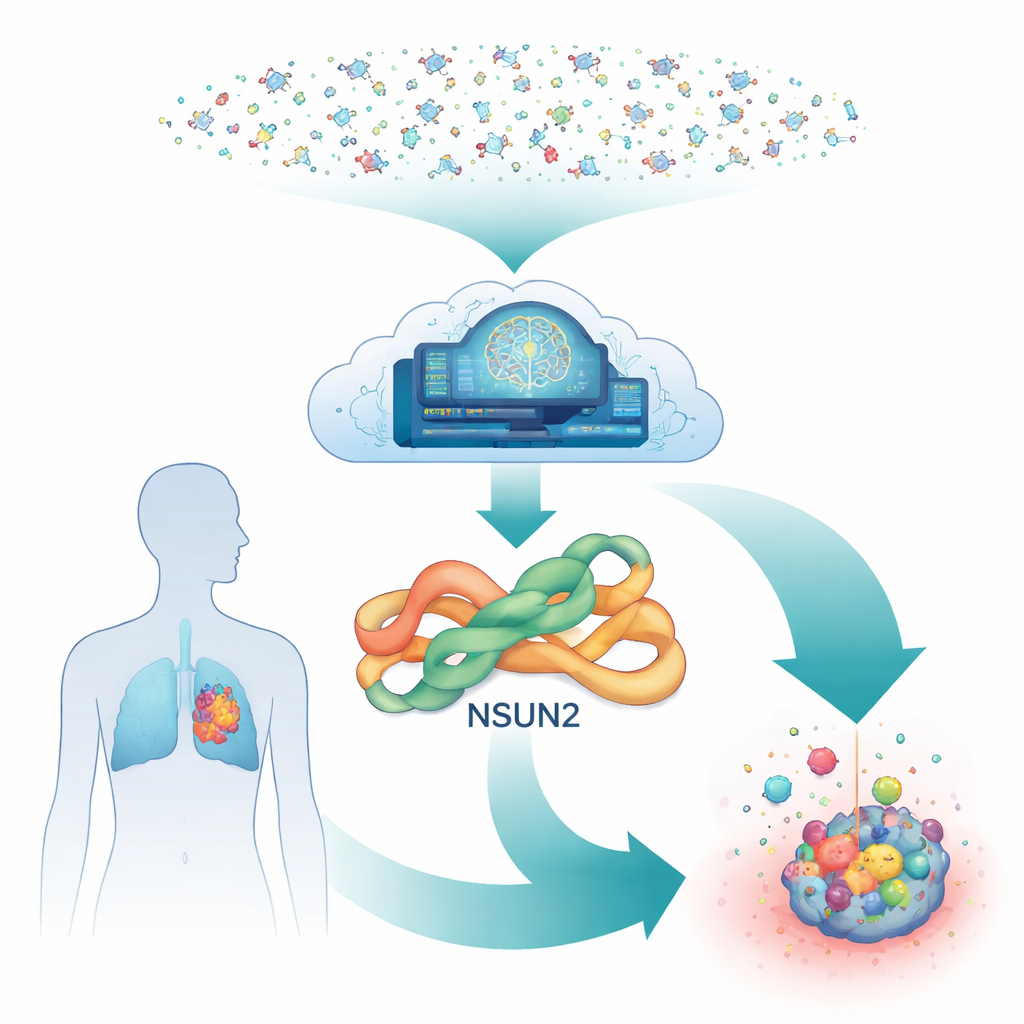
A Hidden Switch Inside Cancer Cells
Deep inside our cells, an enzyme called NSUN2 acts like a chemical highlighter for RNA, the molecule that helps turn genes into proteins. By adding tiny chemical marks to RNA, NSUN2 can make growth-related messages more stable and easier to read. Many tumors, including lung, stomach, pancreatic, and breast cancers, crank NSUN2 levels up, which boosts their ability to divide, spread, and resist targeted drugs. Yet, despite its importance, very few drug candidates exist that can safely turn NSUN2 down, especially reversible ones that do not permanently damage the enzyme.
Letting AI Search a Sea of Molecules
Traditional drug discovery would struggle to test millions of molecules against NSUN2 in the lab, because each experiment requires complex mixtures of RNA and helper chemicals. Instead, the researchers built an all-digital pipeline. They started from a predicted three-dimensional shape of human NSUN2 generated by AlphaFold, an AI system that has revolutionized structure prediction. To ensure this model was trustworthy, they aligned it with a closely related enzyme whose structure is known from X-ray studies. The critical pocket where NSUN2 binds its natural helper molecule turned out to be strongly conserved, giving the team confidence that virtual docking of potential drugs into this site would be meaningful.
From Hundreds of Millions to a Handful
With the target pocket in place, the team turned to a vast public database of purchasable molecules. They first docked a training set of compounds into the NSUN2 pocket and used the resulting scores to teach a machine-learning model which shapes were most promising. That model then rapidly screened about 350 million molecules and flagged roughly 101 million as likely “hits.” To further narrow the field, the top fraction were redocked with more careful calculations, and only the best 12,000, with strong predicted binding, were kept. These were then run through a battery of computer-based safety checks that estimated how the body might absorb, distribute, break down, and tolerate each candidate. After these filters, just 34 molecules remained that looked both potent and drug-like.
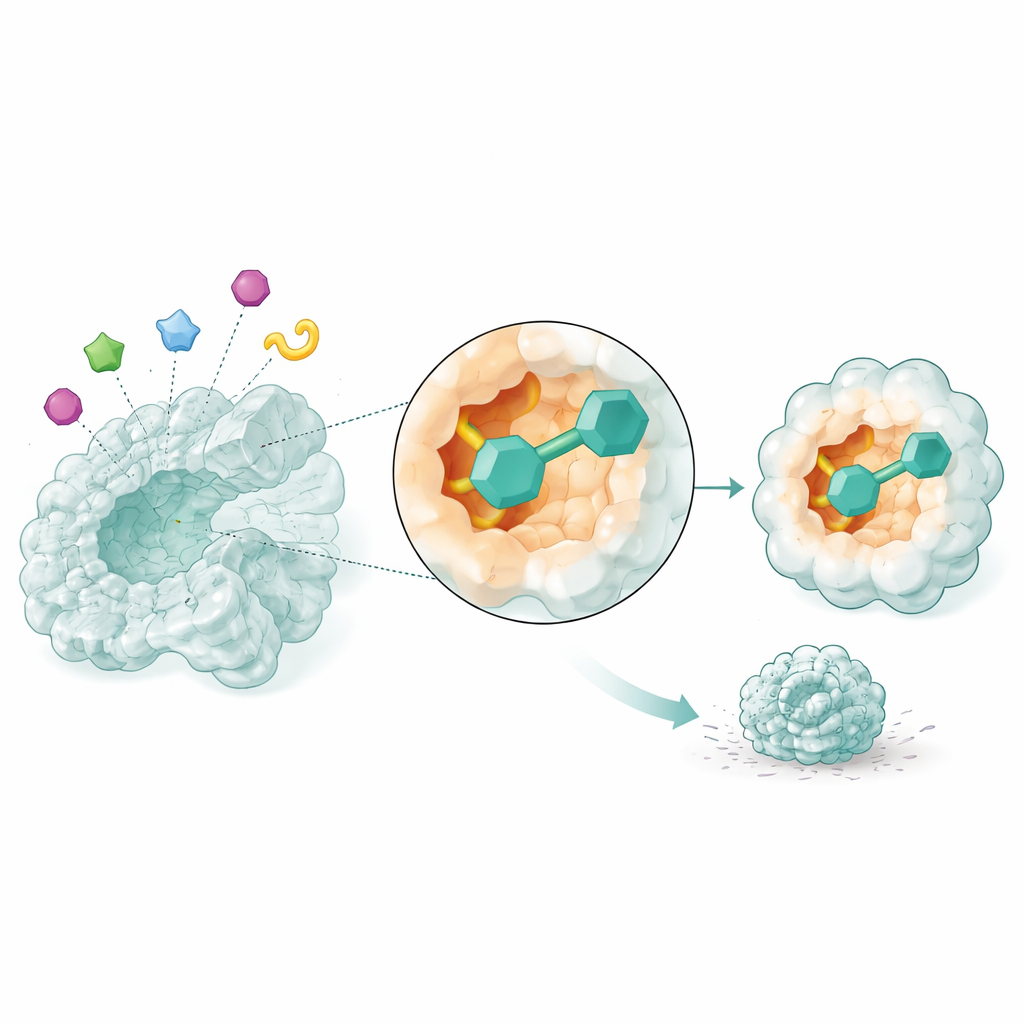
Watching Candidate Drugs in Motion
Static snapshots are not enough to know whether a drug will truly hold on to its target. The researchers therefore used molecular dynamics simulations, which model how atoms move over time, to watch the most promising three candidates interacting with NSUN2 over 50 billionths of a second. Two compounds, identified only by their database codes, formed especially stable complexes: the enzyme structure stayed compact, its key regions remained steady rather than floppy, and the small molecules kept a snug fit inside the pocket while forming persistent contacts. These simulations suggest that the two compounds could reliably block NSUN2’s activity in real cells.
What This Could Mean for Future Treatments
Although every result in this work comes from computations and still needs to be proven in the lab, the study delivers a short list of realistic starting points for new NSUN2-blocking drugs. Because NSUN2 helps cancer cells stabilize growth and survival signals, such medicines could weaken tumors and potentially resensitize them to existing targeted therapies, particularly in lung cancers that have become resistant. Just as important, the study showcases a general recipe for using AI and physics-based modeling to explore enormous chemical spaces quickly and cheaply, offering a powerful shortcut toward the next generation of precision cancer treatments.
Citation: Yu, S., Peng, Q., Wei, W. et al. AI-driven virtual screening platform identifies novel NSUN2 inhibitor candidates for targeted cancer therapy: a computational drug discovery approach. npj Precis. Onc. 10, 98 (2026). https://doi.org/10.1038/s41698-026-01296-2
Keywords: NSUN2, epitranscriptomics, AI virtual screening, cancer drug discovery, RNA methylation