Clear Sky Science · en
T-cell receptor clonotypic diversity and specialization in digestive system cancers
Why this research matters for cancer patients
Cancer immunotherapy has transformed treatment for many people, but it does not work equally well for everyone. This study looks deep into the "barcodes" of immune cells—T-cell receptors—inside tumors of the digestive system, including colorectal and gastric cancers. By reading these barcodes at large scale, the researchers show how the immune system recognizes tumors, how this recognition differs between cancer types, and how it changes when colorectal cancer spreads. These insights could help doctors better predict outcomes and design more precise, personalized immunotherapies.
The immune system’s fingerprint inside tumors
T cells are the body’s targeted defenders, each carrying a unique receptor that lets it recognize specific abnormal cells, including cancer. The team sequenced T-cell receptors from 415 tumor samples taken from 145 patients with colorectal, gastric, and liver cancers, and combined this with detailed clinical information. They examined several receptor chains that define two key T-cell families: conventional αβ T cells, which drive most adaptive immune responses, and γδ T cells, which act more like rapid-response sentinels. They found that αβ T cells tended to form large, highly varied clones, while γδ T cells formed smaller, more specialized groups. Patients with more advanced disease, blood-vessel invasion, or distant spread had more complex T-cell receptor patterns, suggesting that tumor progression pushes the immune system into a broader, more complicated battle.
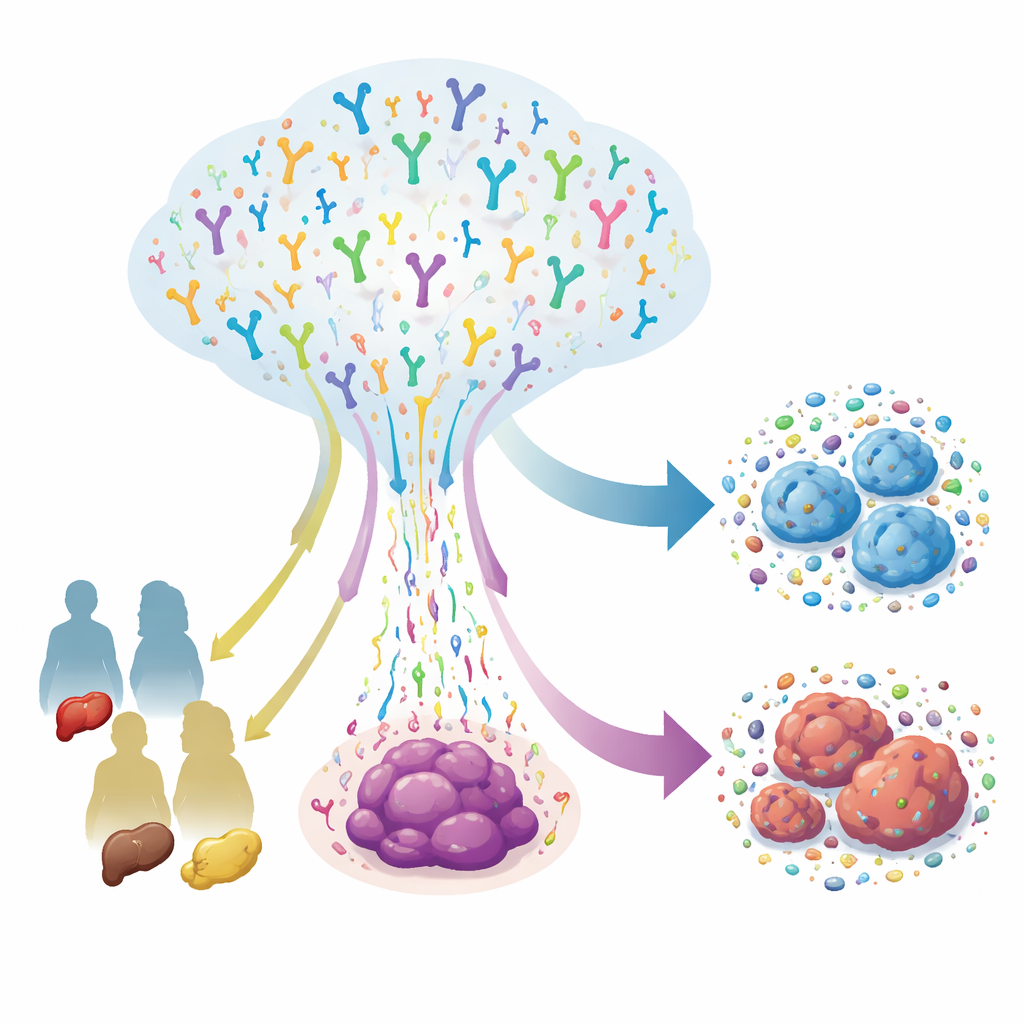
Different cancers, different immune strategies
Although these tumors all arise in the digestive system, they do not attract or shape T cells in the same way. Colorectal cancers were enriched for particular combinations of receptor gene segments in the classic β chain, pointing to a strong role for conventional T cells. Gastric cancers, by contrast, showed heavier use of γδ T-cell receptors, hinting that these less-studied sentinels may play a special role in stomach tumors. When the researchers zoomed in on the most critical part of the receptor—the short loop that actually contacts targets—they found shared sequence patterns across cancer types, signs that T cells are being steered by common selective pressures. Yet there were also clear differences in length, amino-acid composition, and tiny recurring “motifs” that suggest each tumor type forces T cells to solve a slightly different recognition puzzle.
Shared and unique targets for immune attack
Using a global database of known T-cell–antigen pairs, the authors linked specific receptor patterns to their likely targets. Some antigens appeared in both colorectal and gastric cancers, including well-known drivers and immune-related proteins such as KRAS, SF3B1, BST2, and several classic cancer-testis antigens. These shared targets could support "pan-cancer" strategies that use the same immune tools across multiple tumor types. At the same time, each cancer type also showed its own private targets. Colorectal tumors were linked to antigens such as MAGEA10, WT1, and a fusion gene found only in cancer cells, while gastric tumors uniquely featured PABPC1, a protein involved in controlling messenger RNA. Such tumor-specific antigens are attractive candidates for highly selective vaccines or engineered T-cell therapies that spare healthy tissues.
How immune signatures shift when cancer spreads
The study paid particular attention to colorectal cancer that had already metastasized, comparing tumors from patients with and without distant spread. Metastatic tumors were larger, more likely to invade blood vessels, and carried higher blood levels of standard tumor markers. Their T-cell receptor repertoires were also richer and more complex, especially in the β chain, consistent with a tougher and more varied immune contest. By tracking clones shared between non-metastatic and metastatic settings, the researchers grouped them into stable, shrinking, or expanding populations. A striking finding was that some γδ T-cell clones, initially rare in primary tumors, expanded strongly in metastatic disease and became more numerous as tumors grew. Within conventional T cells, an especially expanded set of clones carried a specific sequence motif in the β chain and frequently targeted MLANA, a tumor-associated antigen. High MLANA expression in colorectal tumors was linked to better patient survival, suggesting that this antigen may mark tumors that remain visible to the immune system and responsive to immune-based therapies.
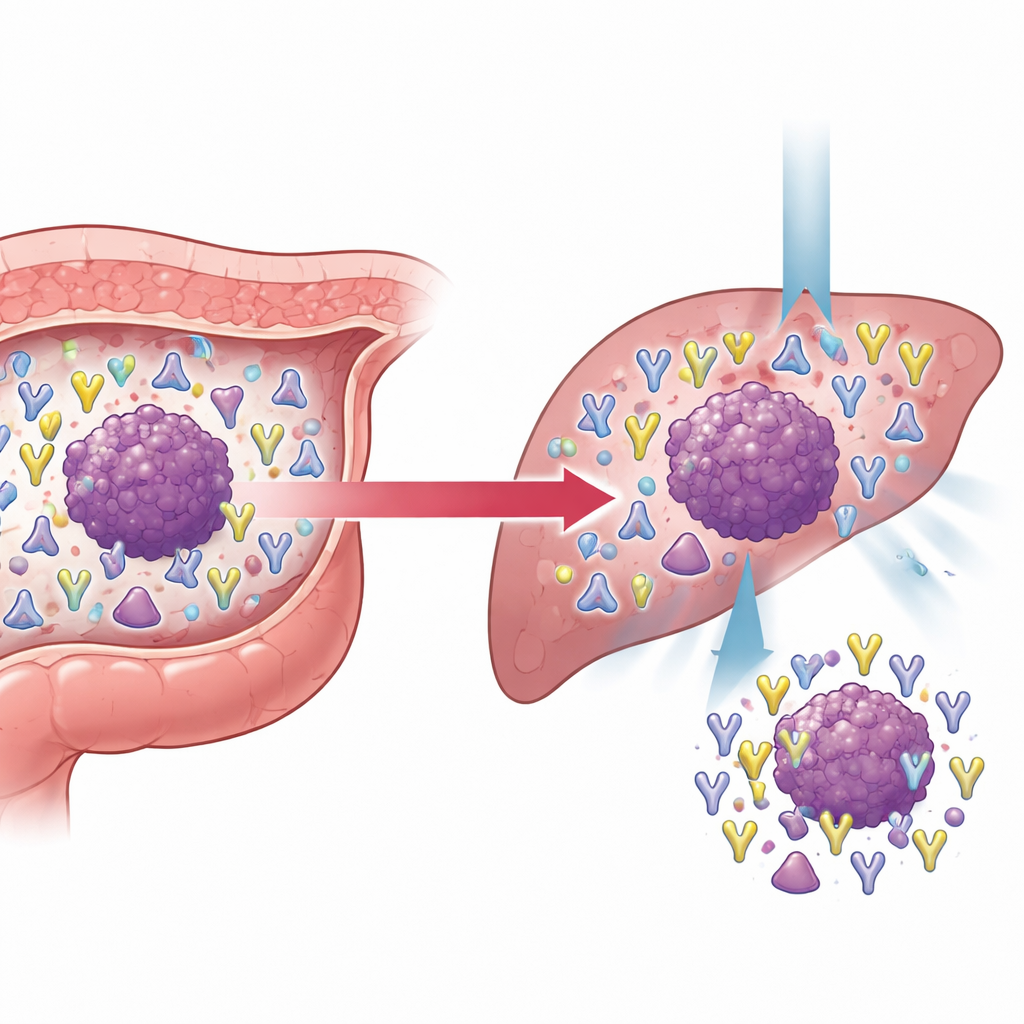
What this means for future cancer care
Overall, the work shows that the immune landscape inside digestive system cancers is not random but shaped by cancer type, stage, and spread. Specific patterns of T-cell receptors act as readable fingerprints of how the immune system is engaging the tumor, highlighting both shared and cancer-specific antigens. For patients, this points toward a future in which doctors could profile T-cell receptor repertoires from a tumor sample to refine prognosis, choose immunotherapies more intelligently, and even design custom treatments that call on the most effective T-cell clones. In colorectal and gastric cancers, the receptor signatures and antigens identified here provide a roadmap for such precision immunotherapy efforts.
Citation: Li, L., Li, J., Wang, F. et al. T-cell receptor clonotypic diversity and specialization in digestive system cancers. npj Precis. Onc. 10, 91 (2026). https://doi.org/10.1038/s41698-026-01294-4
Keywords: T-cell receptors, colorectal cancer, gastric cancer, tumor immunity, immunotherapy biomarkers