Clear Sky Science · en
Identification and characterization of MARCO-expressing tumor-associated macrophages in pancreatic ductal adenocarcinoma with pan-cancer relevance
Why the Body’s Own Defenders Can Help Pancreatic Cancer
Pancreatic ductal adenocarcinoma is one of the most lethal cancers, in part because it hides behind a powerful shield built from the body’s own cells. This study explores a particular kind of immune cell inside pancreatic tumors that, instead of fighting cancer, appears to help it grow and resist treatment. Understanding and disarming these cells could open a new way to make immunotherapy work for patients who currently have very few options.
A Deadly Cancer with a Hostile Neighborhood
Pancreatic tumors are notorious for forming a tough, scar-like tissue packed with immune cells that should, in theory, attack the cancer. Yet most patients respond poorly to modern immunotherapies. The researchers focused on tumor-associated macrophages—“big eaters” of the immune system that normally clear debris and pathogens. In pancreatic cancer, these macrophages are abundant, but their exact roles and identities remained murky. Using single-cell RNA sequencing, which reads the activity of thousands of genes in individual cells, the team mapped the diverse macrophage types present in primary tumors, metastatic sites, and nearby noncancerous tissue.
A Problematic Macrophage Subtype Emerges
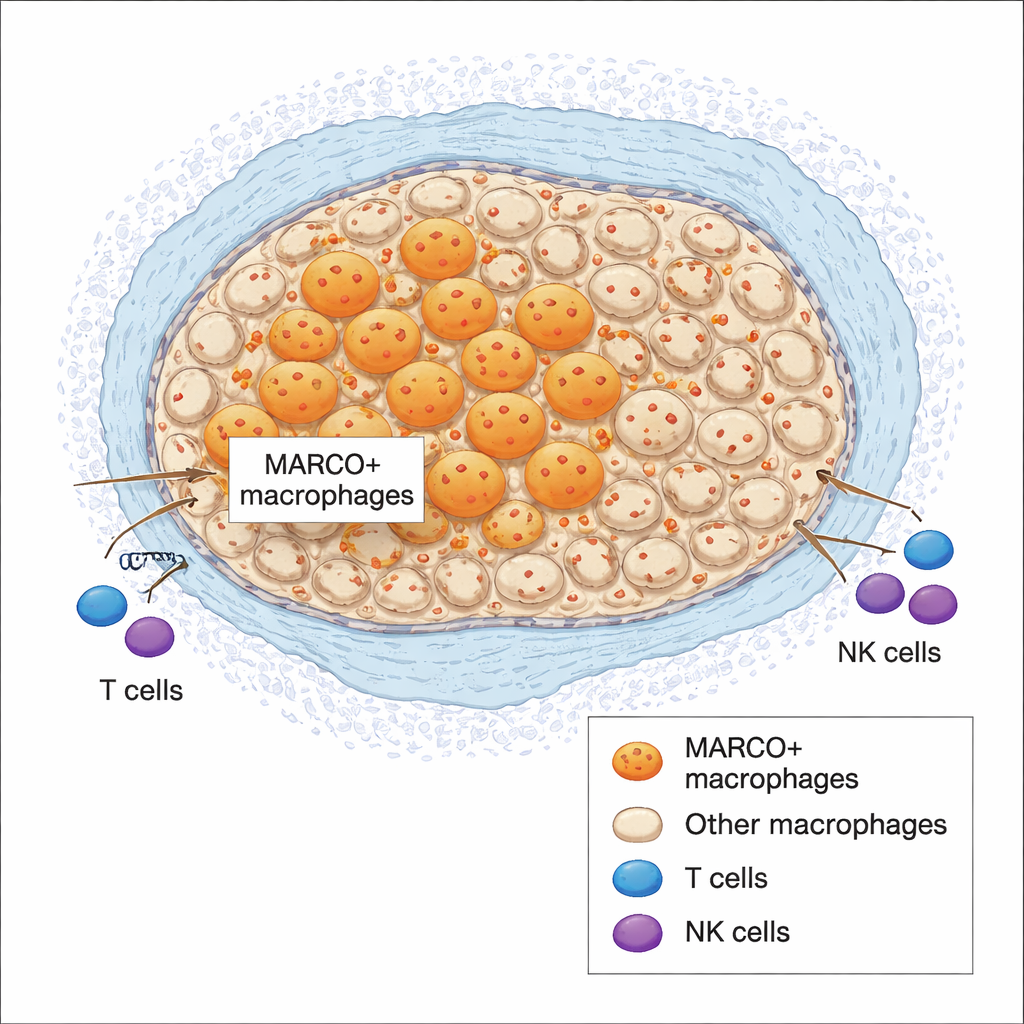
The analysis revealed a distinct subtype of macrophages that strongly expressed a surface protein called MARCO. These MARCO-positive cells were rare in healthy or adjacent tissue but markedly increased in both primary and metastatic pancreatic tumors, as well as in tumors that had already been exposed to chemotherapy. Gene activity patterns showed that MARCO-positive macrophages were geared toward wound-healing, tissue remodeling, and blood vessel growth rather than attack, and they carried many hallmarks of an immunosuppressive, tumor-promoting state. Patients whose tumors had high MARCO levels tended to have fewer cancer-killing CD8 T cells and natural killer (NK) cells, more regulatory T cells that dampen immune responses, and poorer overall survival.
How Tumors Turn Macrophages to Their Side
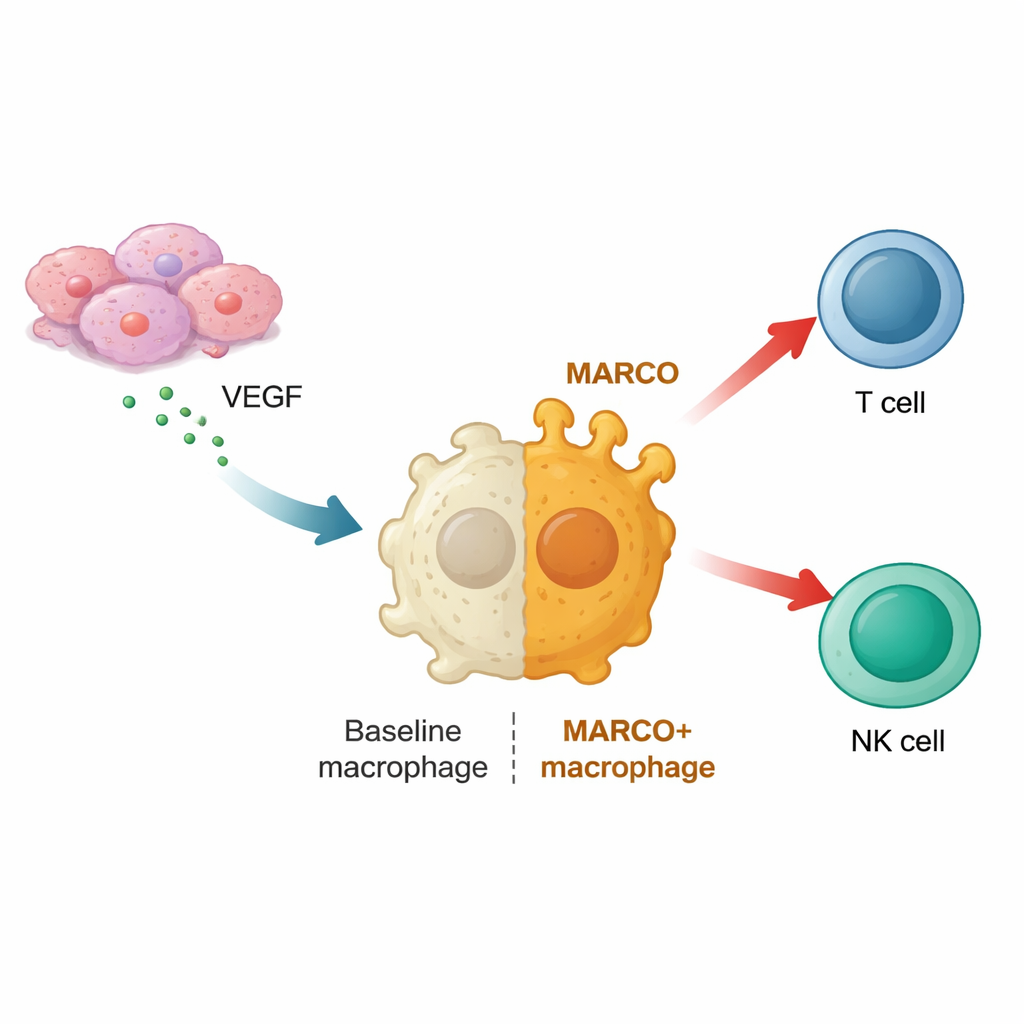
To understand how these harmful macrophages arise, the researchers grew human and mouse macrophages together with pancreatic cancer cells in the laboratory. The cancer cells drove macrophages to switch on MARCO and adopt a “M2-like” profile—a well-known, healing-oriented state that usually calms inflammation. These reprogrammed cells displayed surface markers and gene signatures associated with supporting tumor growth rather than fighting it. Further computational analyses of cell-to-cell communication pointed to VEGF, a growth factor already famous for helping tumors build blood vessels, as a key signal. When macrophages were exposed to VEGF in vitro, MARCO levels rose, suggesting that pancreatic tumors may use VEGF not only to feed themselves with new blood vessels, but also to convert arriving macrophages into loyal allies.
Switching Off MARCO Tips the Balance Back to Attack
The team then asked what would happen if MARCO were removed. In mice genetically engineered to lack the Marco gene, pancreatic tumors grew more slowly and weighed less than in normal mice. Tumors in MARCO-deficient animals contained more active NK cells and showed signs of a more energized immune microenvironment. Macrophages from these mice produced more inflammatory, tumor-fighting molecules and fewer genes linked to the suppressive, wound-healing program. In human tumor samples, sections rich in MARCO-positive macrophages consistently showed fewer NK and CD8 T cells, more regulatory T cells, and higher levels of Ki-67, a marker of rapid cancer cell growth, reinforcing the idea that MARCO-marked macrophages help create a safe haven for the tumor.
Beyond the Pancreas: A Shared Weak Spot in Many Cancers
Finally, the researchers broadened their view to include single-cell data from 17 different cancer types. In 11 of them—including skin melanoma, stomach, colon, lung, brain, and kidney cancers—tumor-associated macrophages expressed more MARCO than their counterparts in noncancerous tissue. Across these tumors, MARCO-positive macrophages repeatedly showed gene programs linked to invasion, altered metabolism, and blood vessel formation, suggesting that this harmful macrophage state is a common strategy cancers use to thrive. This raises the possibility that drugs or antibodies designed to block MARCO could have value far beyond pancreatic cancer.
What This Means for Patients
For a layperson, the message is that some of the immune cells sitting inside an aggressive tumor are not failed soldiers, but active collaborators. This work identifies MARCO as a simple flag on the surface of those collaborator cells. Because MARCO is exposed on the outside of macrophages, it could be targeted with antibodies, much like other modern cancer drugs. In animal models, removing MARCO made tumors smaller and allowed more cancer-killing cells to enter the tumor. Together, these findings suggest that therapies aimed at blocking or reprogramming MARCO-positive macrophages might help “unmask” pancreatic tumors and make existing immunotherapies more effective, and they may hold promise for several other hard-to-treat cancers as well.
Citation: Sun, H., Gao, M., Liu, Z. et al. Identification and characterization of MARCO-expressing tumor-associated macrophages in pancreatic ductal adenocarcinoma with pan-cancer relevance. npj Precis. Onc. 10, 78 (2026). https://doi.org/10.1038/s41698-026-01293-5
Keywords: pancreatic cancer, tumor microenvironment, macrophages, immunotherapy, MARCO