Clear Sky Science · en
Multimodal single-cell profiling reveals crosstalk between macrophages and stromal cells in poor prognostic cholangiocarcinoma patients
Cancer’s Hidden Neighborhood
Cholangiocarcinoma, a cancer of the bile ducts in the liver, is rare but often deadly, with few effective treatments. Many patients do not respond to modern immunotherapy drugs that unleash the immune system against tumors. This study asks a simple but crucial question: what is happening inside the tumor’s “neighborhood” that keeps the body’s defender cells from doing their job—and why do some patients fare worse than others?
Why Immune Cells Get Stuck at the Gate
Instead of being a loose ball of cancer cells, cholangiocarcinoma is packed with supporting tissue, called stroma, and a variety of immune cells. By profiling individual cells from tumors, nearby liver tissue, and blood, the researchers show that the tumor center is poor in the very T cells that are best at killing cancer. These so-called effector memory T cells stay mostly in the surrounding liver rather than entering the tumor. In contrast, regulatory T cells—immune cells that act as brakes—pile up inside the tumor. Many of the T cells that do make it in display multiple “off switches” on their surface, signs that they are exhausted and unable to fight.
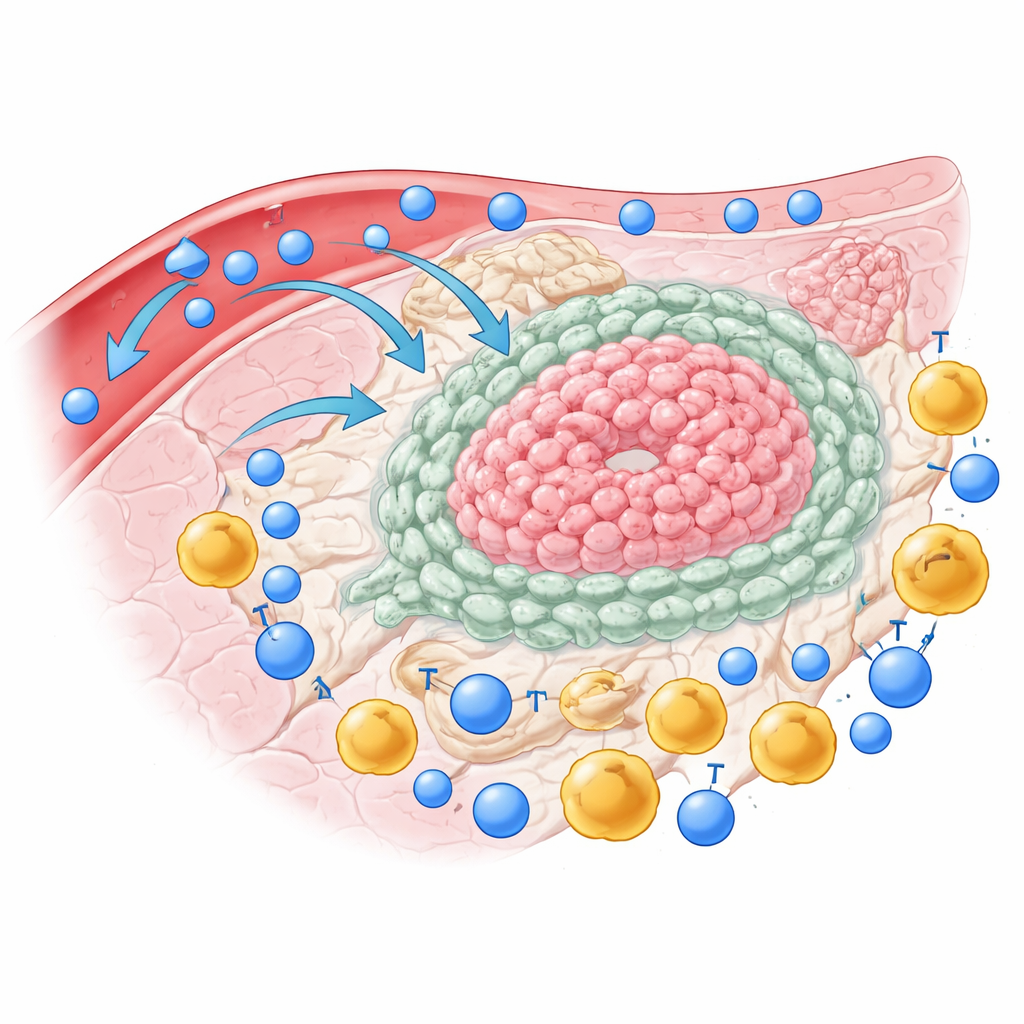
The Tumor’s Suppressive Alliance
To look beyond T cells, the team mapped thousands of single cells from patients’ tumors at the level of gene activity. They found not only several flavors of cancer cells, but also distinct groups of support cells, such as cancer-associated fibroblasts, liver stellate cells, blood-vessel lining cells, and macrophages—immune cells that can either attack or aid tumors. In patients with especially poor outcomes, a particular subset of cancer cells showed stronger communication with stromal cells and macrophages. Rather than acting independently, these cell types formed a tight network that seemed designed to keep attacking T cells out and turned off.
Silent Signals That Shut Down Defenses
Diving into these conversations, the scientists identified key molecular “handshakes” passed from one cell to another. Cancer cells produced versican, a sticky matrix protein that interacts with sensors on macrophages and growth receptors on stromal cells, reshaping the tissue in ways that favor tumor growth and dampen immune attack. Macrophages produced TGF-beta, a powerful signal known to both generate more regulatory T cells and weaken cancer-killing T cells. Another pathway involved GAS6 binding to the AXL receptor on macrophages, fibroblasts, and cancer cells, a combination linked in other cancers to aggressive behavior and immune escape. Together, these signals create a feedback loop: the stroma and macrophages remodel the environment, reinforce immune brakes, and help cancer cells thrive.
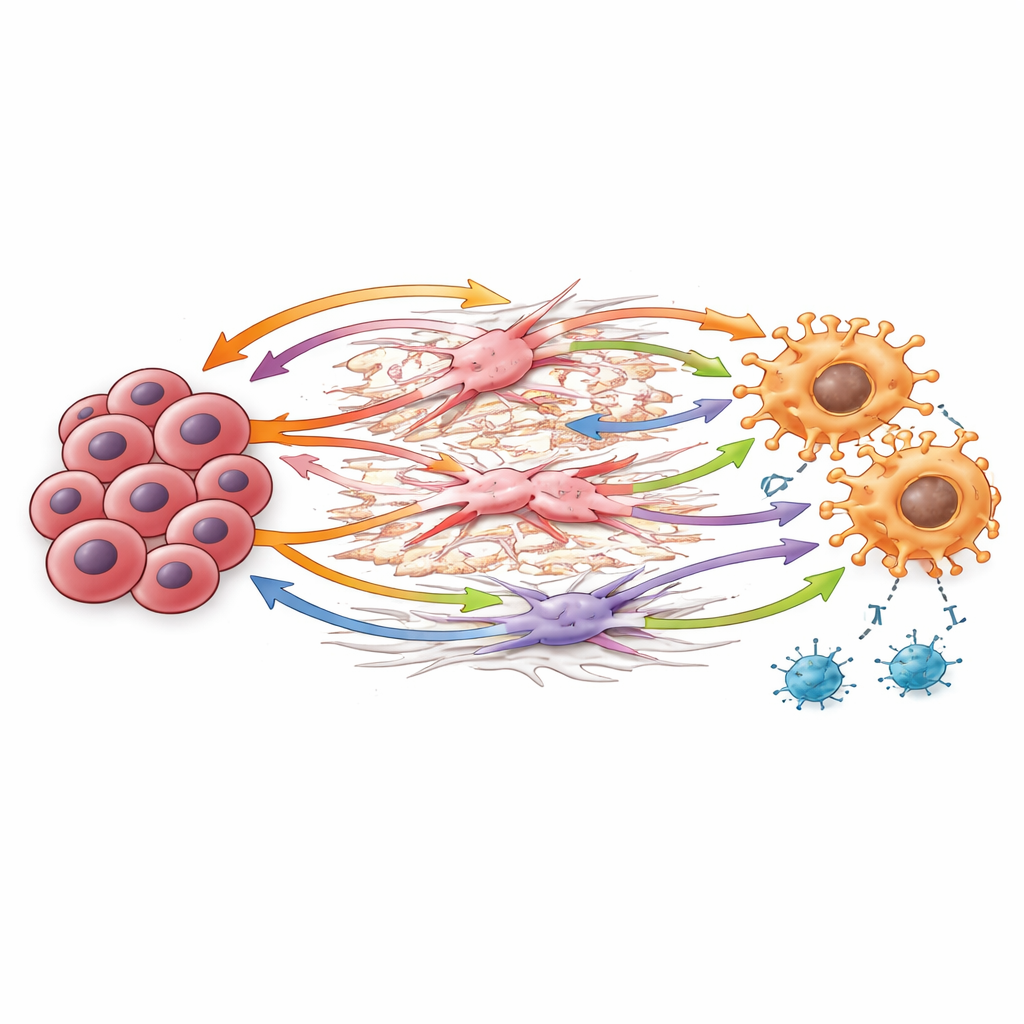
A Pattern That Extends Beyond a Small Group
Although the single-cell work was done in a modest number of patients, the team checked their findings against a much larger public dataset of extrahepatic cholangiocarcinoma samples. By mathematically estimating which cell types were present in each tumor, they discovered a sizeable group of patients whose tumors were rich in stromal cells and macrophages and expressed high levels of the same key molecules—versican, GAS6, AXL, and the growth receptor EGFR. This suggests that the suppressive network uncovered in the small study is not a rare curiosity but may define a clinically important subgroup of patients with particularly aggressive disease biology.
What This Means for Future Treatment
For people with cholangiocarcinoma, these findings help explain why standard immune checkpoint drugs often fall short: the problem is not just a single brake on T cells, but an entire neighborhood of cells collaborating to keep the immune system in check. The study highlights several signaling routes—such as the GAS6–AXL, versican–TLR2, and TGF-beta–EGFR axes—that could be targeted alongside checkpoint inhibitors to weaken the tumor’s protective shield. It also points to the need for tests that look not only at tumor cells but also at surrounding liver tissue to see whether aggressive T cells are present yet blocked from entry. In the long run, decoding this complex cellular crosstalk may lead to more precise ways to select patients for immunotherapy and to design combination treatments that give their immune system a real chance to fight back.
Citation: Heij, L., Hayat, S., Reichel, K. et al. Multimodal single-cell profiling reveals crosstalk between macrophages and stromal cells in poor prognostic cholangiocarcinoma patients. npj Precis. Onc. 10, 105 (2026). https://doi.org/10.1038/s41698-026-01292-6
Keywords: cholangiocarcinoma, tumor microenvironment, single-cell profiling, immune exhaustion, tumor-associated macrophages