Clear Sky Science · en
Metastatic progression of pheochromocytoma and paraganglioma occurs via parallel evolution
Why these rare tumors matter
Pheochromocytomas and paragangliomas are rare tumors that arise from hormone‑producing nerve cells deep in the body. They can trigger severe spikes in blood pressure and other dangerous symptoms, often in relatively young adults. Up to a quarter of patients eventually develop spread of the disease to distant organs, yet there are few effective drug options. This study asks a simple but crucial question: how, and when, do these tumors become metastatic—and could their DNA reveal new ways to treat them?
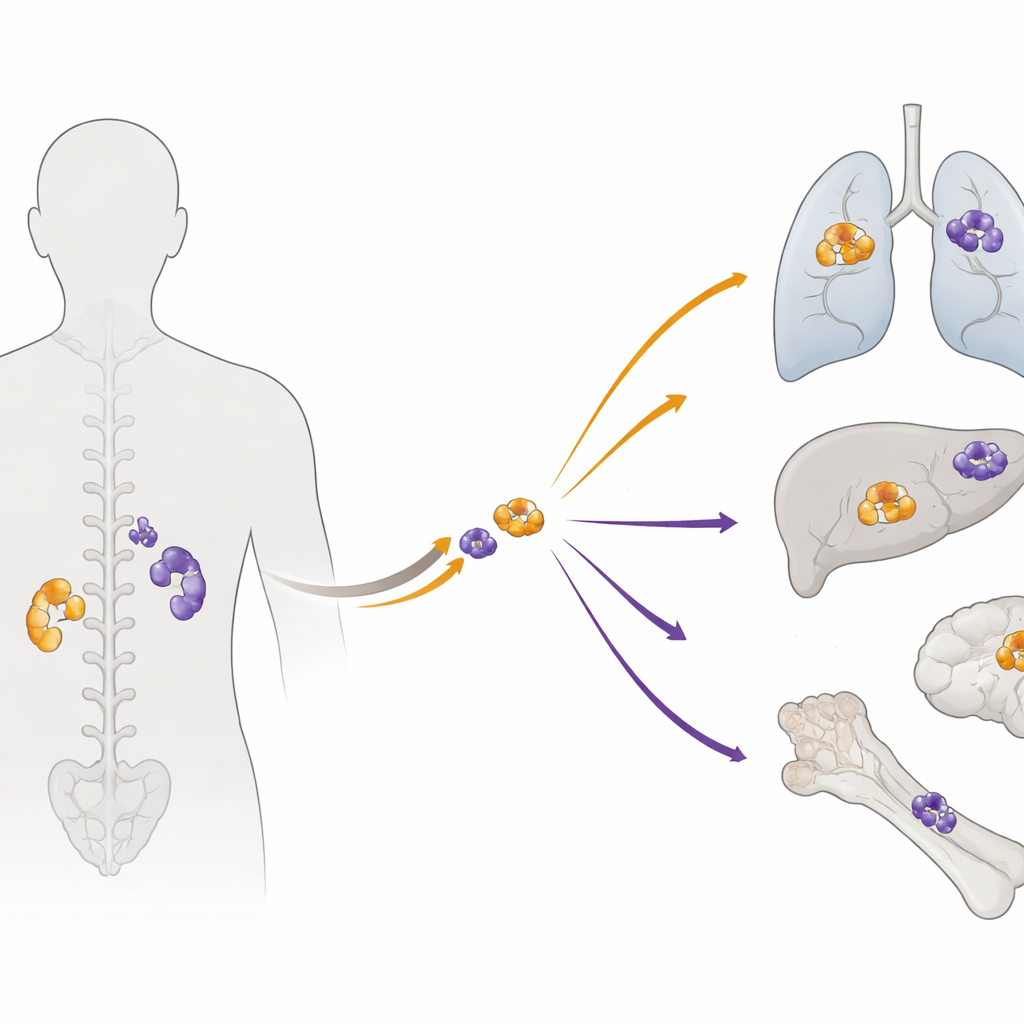
Following the trail of cancer DNA
The researchers analyzed paired tumor samples from 27 people whose disease had already spread. For many patients, they had DNA from both the original tumor and its later metastases, as well as normal tissue for comparison. Using whole‑exome sequencing—a technique that reads all protein‑coding genes—they cataloged thousands of mutations and larger DNA gains and losses. They then applied statistical tools to distinguish likely “driver” changes that push cancer forward from background noise, and to reconstruct the order in which key genetic events occurred.
Hidden weak spots in tumor defenses
One of the striking patterns was the high frequency of damage in genes that help package DNA and repair it when it breaks. Nearly 40% of tumors carried harmful changes in chromatin‑remodeling genes or DNA damage‑repair genes. These included ATRX and well‑known repair genes such as BRCA1, BRCA2, ATM, and ATR. Many patients also had inherited mutations in SDHx genes, which are already linked to these tumors. The combination of faulty metabolism from SDHx and impaired DNA repair suggests that some tumors might be unusually vulnerable to drugs that exploit such weaknesses, including PARP inhibitors and other therapies targeting the DNA repair machinery.
Early escape and long, quiet growth
When the team compared each primary tumor with its metastases, they found surprisingly little overlap in the finer‑scale mutations. On average, less than 10% of the potentially important changes were shared. Yet careful checks confirmed that each pair truly came from the same patient. Using “molecular clock” methods that infer timing from how many mutations have accumulated, the authors estimated that the founding cells of metastases often branched off very early—around 18 years before the primary tumor was diagnosed. In many cases, the actual seeding of distant sites occurred more than a decade before doctors ever found the original mass. Large‑scale DNA changes, such as whole‑chromosome gains and losses, tended to happen early and were shared, while most smaller mutations appeared later and separately in each location.
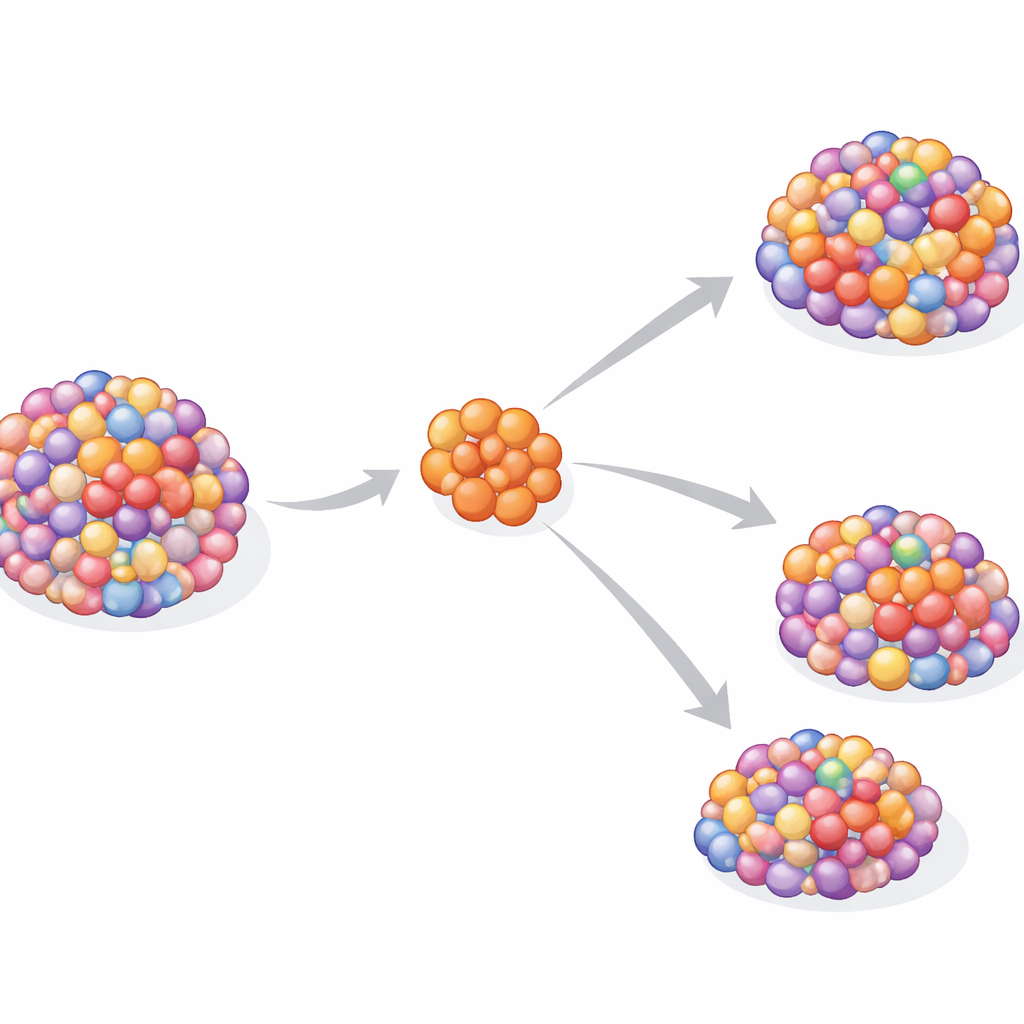
One clone, many separate journeys
Despite the genetic diversity, the data suggest that each metastasis usually began from a single ancestral cell rather than a mixture. This “monoclonal seeding” pattern contrasts with some other cancers, where multiple branches of the primary tumor can jointly colonize new sites. Here, a lone offshoot appears to leave the early tumor, settle elsewhere in the body, and then evolve largely on its own for years. The result is parallel evolution: the primary and metastatic tumors share early structural DNA changes but then diverge, each acquiring its own unique set of mutations and potential drug sensitivities over time.
What this means for patients and treatment
For people facing pheochromocytoma or paraganglioma, this work paints a sobering but informative picture. Metastases are not simply late offshoots of an advanced primary tumor; they are early travelers that can quietly grow for years before anyone knows they exist. At the same time, the study uncovers recurring damage in DNA‑repair and chromatin‑related genes, pointing to new therapeutic angles. Together, these findings argue for closer genetic study of metastatic lesions themselves and support trials of drugs that target DNA repair and related pathways. Understanding when and how these tumors spread could ultimately lead to earlier detection and more precise, personalized treatment for patients.
Citation: Pregnall, A.M., Wubbenhorst, B., D’Andrea, K. et al. Metastatic progression of pheochromocytoma and paraganglioma occurs via parallel evolution. npj Precis. Onc. 10, 101 (2026). https://doi.org/10.1038/s41698-026-01291-7
Keywords: pheochromocytoma, paraganglioma, metastasis, tumor evolution, DNA repair