Clear Sky Science · en
Loss of MicroRNA-29b promotes DNMT3b-mediated STING downregulation to attenuate radiotherapy-induced antitumor immunity in KRAS-mutated colorectal cancer
Why this research matters
For many people with rectal and colon cancer, radiation is a key part of treatment before surgery. Yet some tumors barely shrink, and cancer often returns or spreads. This study looks under the hood of one common genetic change in colorectal cancer—called a KRAS mutation—to explain why these tumors often resist radiotherapy and new immunotherapies, and it proposes a strategy to turn the body’s own immune system back on against them.
A common mutation that hides tumors from immune attack
Colorectal cancer frequently carries mutations in the KRAS gene, a major driver of tumor growth and spread. Clinicians had already noticed that KRAS-mutated tumors tend to respond poorly to both targeted drugs and radiotherapy. By mining large cancer databases and examining patient samples, the authors found that tumors with mutant KRAS show fewer cancer-killing CD8 T cells and weaker activity of immune-boosting interferon signals. In patients receiving standard chemoradiotherapy before rectal cancer surgery, those with normal KRAS gained many more CD8 T cells inside their tumors after treatment than those with KRAS mutations, helping explain their better outcomes.
The missing alarm system inside cancer cells
Radiotherapy not only damages tumor DNA; it can also act like a flare, causing cancer cells to release DNA fragments that trigger an internal alarm pathway called cGAS–STING. When STING is active, cells produce type I interferons and other signals that draw in and activate immune cells, including T cells and dendritic cells. The researchers showed that in colorectal cancer cells engineered to carry mutant KRAS, this alarm system is blunted: after radiation, these cells produced much less interferon and related molecules. In mice, silencing KRAS in tumors restored STING levels, boosted interferon signals, and led to more CD4 and CD8 T cells, fewer regulatory T cells, and stronger tumor control—not only in irradiated tumors but also in distant, non‑irradiated “abscopal” tumors.
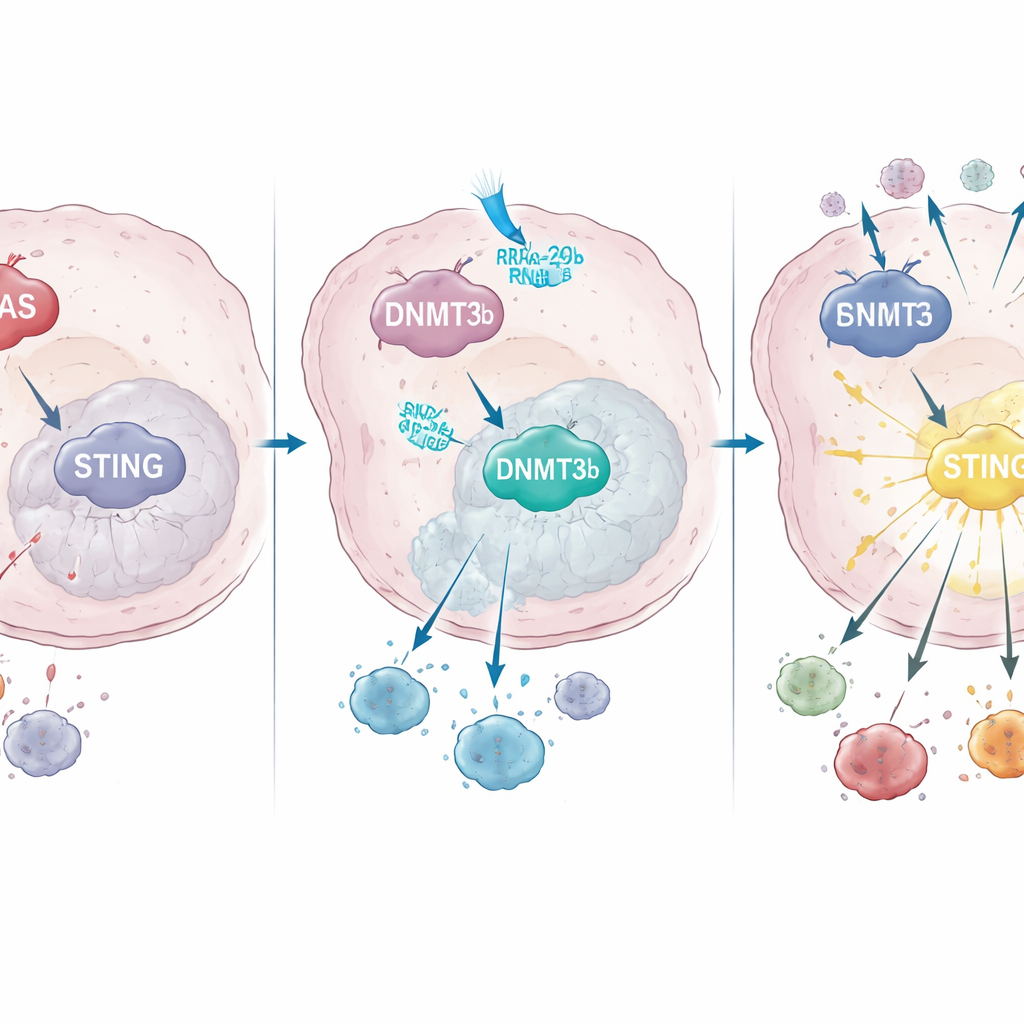
How KRAS rewires gene control to shut down STING
Diving deeper, the team discovered that mutant KRAS increases the activity of a DNA-modifying enzyme called DNMT3b, which adds methyl groups to certain DNA regions and turns nearby genes off. In both cancer cell lines and patient samples, high DNMT3b went hand in hand with low STING. A key stretch of the STING promoter—the DNA “on switch” region—was heavily methylated in KRAS-mutant tumors, but not in those with normal KRAS. Patients whose tumors could still upregulate STING after chemoradiotherapy had more CD8 T-cell infiltration and better survival, underscoring how important this pathway is for successful treatment.
A small RNA that can reawaken the alarm
The authors then asked what makes DNMT3b so abundant in KRAS-mutant cancer. Using microRNA sequencing, they identified a tiny regulatory RNA, microRNA‑29b‑3p, that normally keeps DNMT3b in check but is strongly suppressed by mutant KRAS. Restoring this microRNA in lab-grown KRAS-mutant cells lowered DNMT3b, revived STING, and enhanced interferon production after radiation; blocking it had the opposite effect. In tumor samples, KRAS-mutant patients tended to have low microRNA‑29b‑3p, high DNMT3b, and low STING, and those with higher microRNA‑29b‑3p lived longer, linking this molecular chain directly to clinical outcomes.
A targeted gene delivery approach that boosts radiotherapy and immunotherapy
To translate these findings into a potential treatment, the team engineered a harmless virus (AAV) that delivers microRNA‑29b‑3p under the control of a promoter active mainly in colorectal cancer cells. In mouse models bearing KRAS‑mutant colorectal tumors, giving this AAV plus localized radiotherapy dramatically shrank both irradiated tumors and distant tumors that did not receive radiation. Tumors showed less DNMT3b, more STING, more interferon signals, and heavy infiltration by T cells and other immune cells. When the same microRNA‑29b‑3p treatment was combined with an immune checkpoint inhibitor targeting PD‑1, survival improved even further, and this benefit depended on CD8 T cells, confirming that the immune system was doing the heavy lifting.
What this could mean for patients
In simple terms, this study reveals how KRAS-mutated colorectal cancers “cut the wire” to an internal alarm system that radiation normally sets off, allowing them to dodge immune attack. By restoring a single small RNA, microRNA‑29b‑3p, the researchers could lower DNMT3b, reawaken STING, and turn immunologically “cold” tumors into “hot” ones that respond better to both radiotherapy and checkpoint blockade. While this work is still at the preclinical stage, it points toward future treatments that pair radiation with tumor-targeted gene therapy to overcome resistance in KRAS‑mutant colorectal cancer, potentially making existing therapies work for many more patients.
Citation: Chang, HY., Chen, JY., Ke, TW. et al. Loss of MicroRNA-29b promotes DNMT3b-mediated STING downregulation to attenuate radiotherapy-induced antitumor immunity in KRAS-mutated colorectal cancer. npj Precis. Onc. 10, 93 (2026). https://doi.org/10.1038/s41698-026-01290-8
Keywords: KRAS-mutated colorectal cancer, radiotherapy resistance, STING pathway, microRNA-29b, tumor immunology