Clear Sky Science · en
Comparative analysis of ctDNA monitoring strategies in advanced NSCLC with MET exon 14 skipping mutations treated with ensartinib
Why a Blood Test Matters for Lung Cancer
For people living with advanced lung cancer, one of the biggest questions is whether a new drug is actually working—and how soon doctors can tell. This study explores a blood-based approach, using tiny fragments of tumor DNA that circulate in the bloodstream, to quickly gauge how well a targeted drug called ensartinib is helping patients whose tumors share a specific genetic glitch. The findings suggest that a simple early blood test could flag which patients are likely to enjoy longer benefit from treatment, and which may need a change in strategy.
Putting a Tumor’s Fingerprints into a Blood Tube
Cancer cells shed bits of their genetic material into the blood, known as circulating tumor DNA. In this trial, researchers focused on people with non-small cell lung cancer driven by a change in a gene called MET, specifically a defect known as exon 14 skipping. All patients received ensartinib after previous treatments had failed. Blood samples were drawn just before starting the drug and again four weeks later. Using an advanced sequencing test that scans 229 cancer-related genes, the team grouped the detected alterations into three categories: changes in MET itself, a set of well-established cancer drivers, and a broad catch-all group of any detectable mutation.
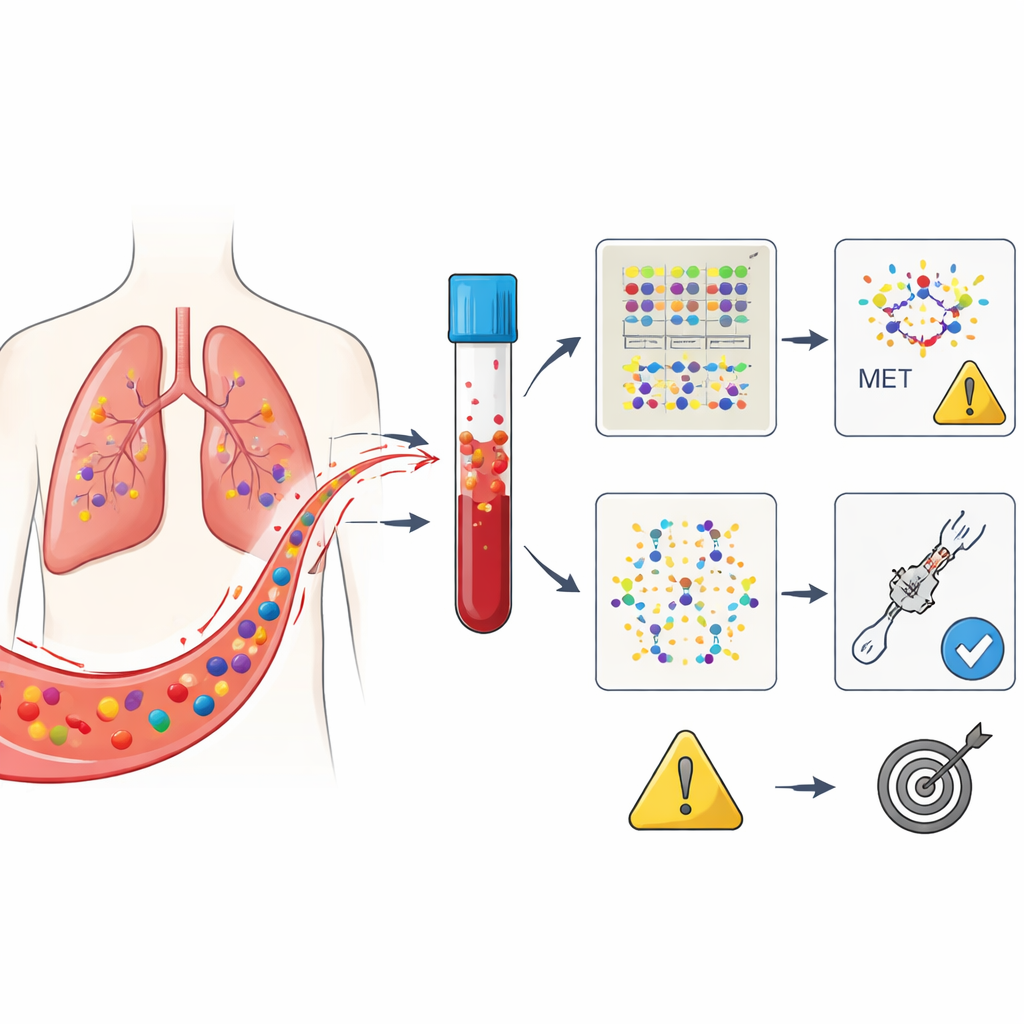
Three Ways to Read the Same Blood Signal
The central question was which of these three blood “views” best predicts who will respond to ensartinib and how long that response will last. At the start, not all patients had detectable tumor DNA in their blood, even though their tumors carried the MET defect; this reflects how unevenly cancers shed DNA. Early on, simply being positive or negative for tumor DNA at baseline did not strongly predict outcome. However, by the four-week mark, a clear pattern emerged: patients whose blood no longer showed MET-related DNA changes tended to stay free of disease worsening much longer than those in whom these signals persisted. This was true even though tumor scans at that time could still be ambiguous.
When MET Signals Fade, Outcomes Improve
Looking more closely at how blood results changed over time, the team found three paths. Some patients never had detectable MET DNA in their blood, some started positive and became negative, and others either stayed positive or developed new MET changes. Those who cleared MET signals by week four enjoyed the longest stretch before their cancer progressed, around nine months on average, and had the highest rate of measurable tumor shrinkage. Patients who remained positive for MET DNA did far worse, with disease progression in just over two months on average. Monitoring the broader sets of mutations also carried useful information, but was less precise because many low-level or unrelated changes could muddy the picture.
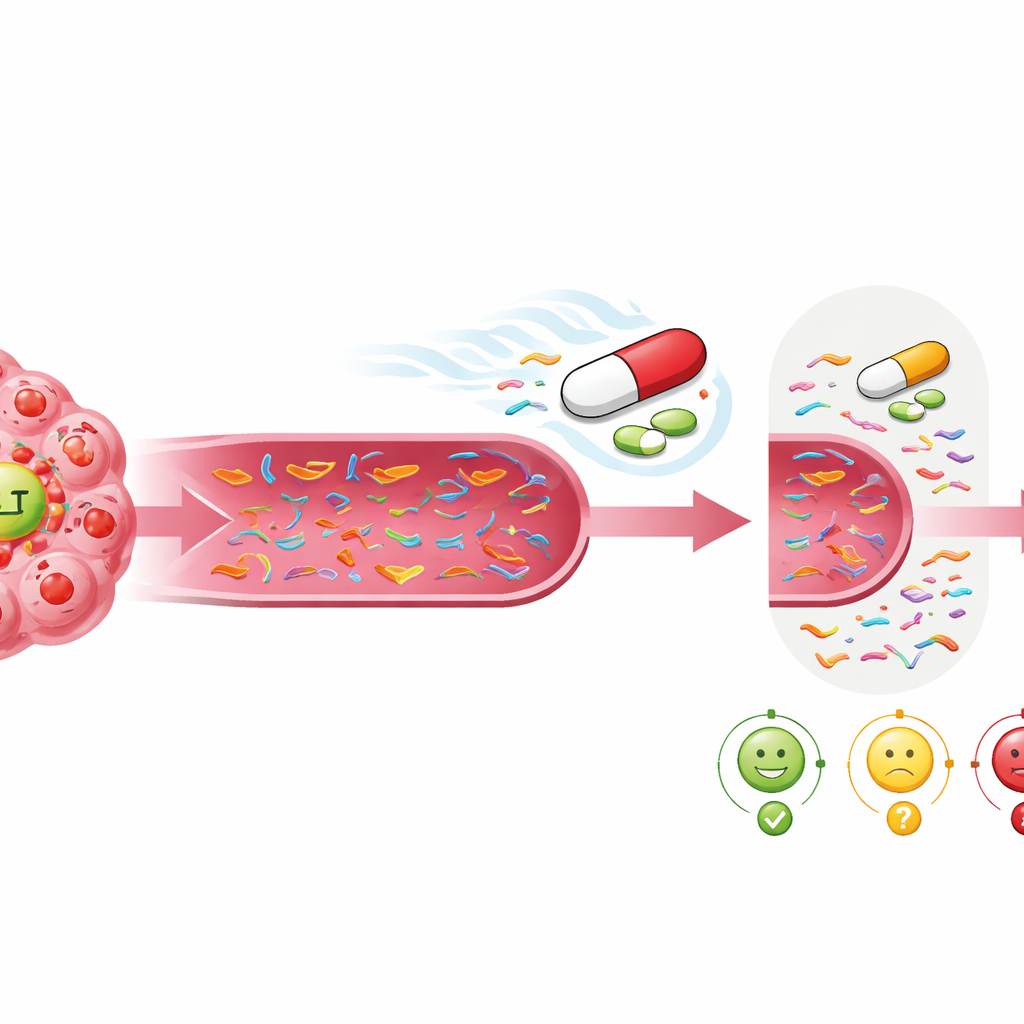
Balancing Simplicity and Deeper Insight
Using only MET-related DNA as a guide offered the sharpest and simplest snapshot of whether ensartinib was helping. This focused approach had high specificity, meaning that when MET signals disappeared, there was a strong chance the patient would do well. That makes it attractive as a practical tool: single-gene tests are cheaper, faster, and easier to interpret than large gene panels. At the same time, scanning more genes still had value, because it could uncover new mutations that might explain why a tumor later stopped responding and point to future treatment options. The study suggests that combining a narrow, MET-centered view for early treatment decisions with a broader view for tracking resistance could offer the best of both worlds.
What This Means for Patients
To a layperson, the key message is that a blood test taken just a month after starting ensartinib can already hint at the drug’s long-term payoff. If MET-related tumor DNA disappears from the bloodstream, the odds of meaningful and lasting benefit are high; if it lingers, doctors may need to watch more closely or consider other options. While this was a relatively small, single-arm study that needs confirmation in larger groups, it strengthens the idea that “liquid biopsies” can act as an early report card on targeted treatments. In the future, such monitoring could help personalize care, sparing patients from staying on ineffective drugs and guiding them more quickly toward therapies that work.
Citation: Zhou, M., Zhu, Y., Sun, X. et al. Comparative analysis of ctDNA monitoring strategies in advanced NSCLC with MET exon 14 skipping mutations treated with ensartinib. npj Precis. Onc. 10, 110 (2026). https://doi.org/10.1038/s41698-026-01284-6
Keywords: circulating tumor DNA, MET exon 14 skipping, ensartinib, non-small cell lung cancer, liquid biopsy monitoring