Clear Sky Science · en
An integrated single-cell lung cancer atlas reveals distinct fibroblast phenotypes between adenocarcinoma and squamous cell carcinomas
Why the Neighborhood Around a Tumor Matters
Lung cancer is not made of cancer cells alone. It is more like a crowded city where cancer cells live alongside blood vessels, immune cells, and support cells. This study asks a surprisingly simple question with big implications: do the two major forms of non–small cell lung cancer—adenocarcinoma and squamous cell carcinoma—build different “neighborhoods” inside the lung, and could these differences help explain why patients do better or worse? Using powerful single-cell DNA-reading tools, the researchers map hundreds of thousands of individual cells to reveal how one key group of support cells, called fibroblasts, behave very differently in these two cancers.
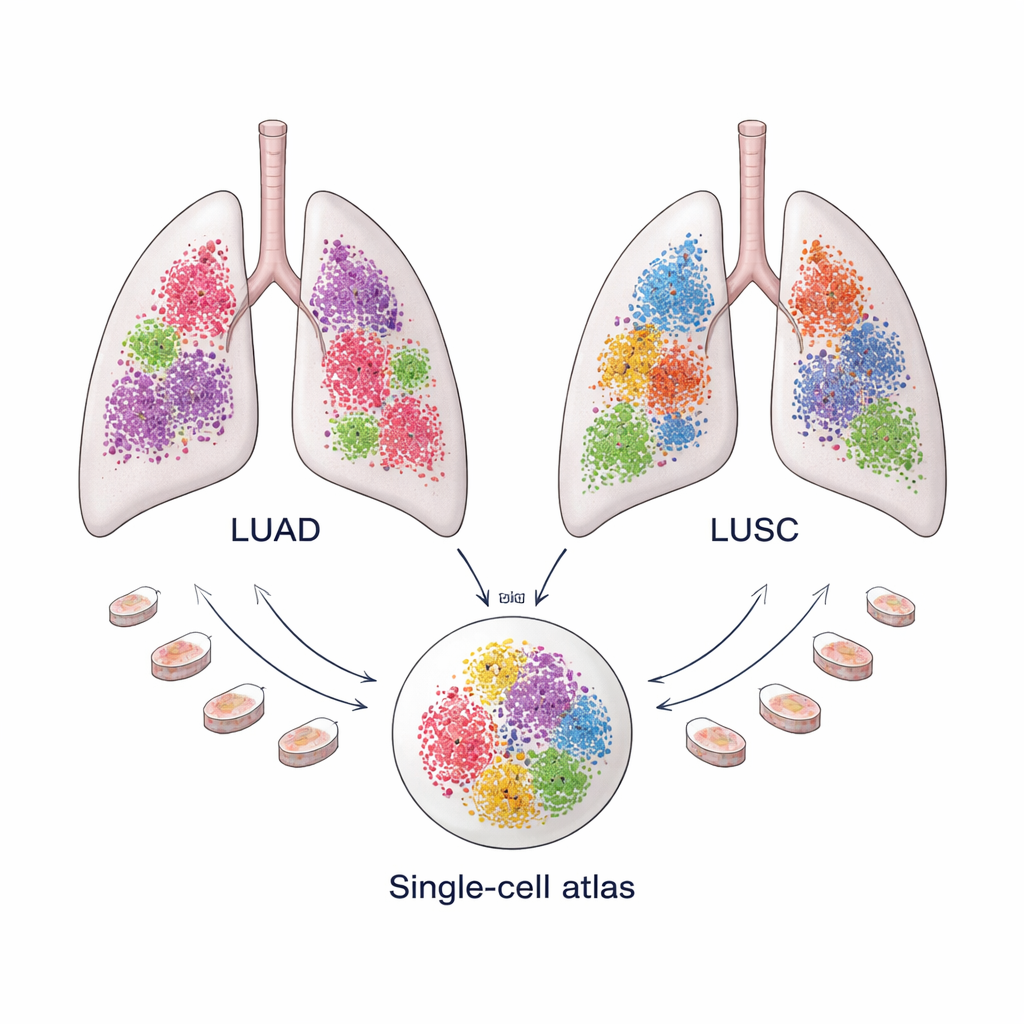
Two Common Lung Cancers, Two Distinct Ecosystems
Non–small cell lung cancer makes up about 85 percent of lung cancers and is dominated by two types: lung adenocarcinoma (LUAD), which often arises in the outer parts of the lung, and lung squamous cell carcinoma (LUSC), which tends to occur more centrally. Although both are treated under the same broad label today, they respond differently to therapy and have distinct long-term outcomes. Growing evidence suggests that these differences are not only due to the cancer cells themselves, but also to the “tumor microenvironment”—the mix of immune cells, blood vessels, and structural cells that surround and interact with the tumor.
Building a Single-Cell Lung Cancer Atlas
To understand these microenvironments in detail, the team assembled a large “atlas” of single-cell RNA sequencing data: 366,652 cells from 175 LUAD samples and 125,238 cells from 74 LUSC samples, drawn from ten public datasets. Each cell’s gene activity pattern served as a fingerprint that allowed the researchers to sort cells into major groups such as immune cells, blood vessel cells, cancer cells, and structural or “stromal” cells. Sophisticated computational methods then removed technical noise and aligned samples so that cells from different patients could be compared directly. This scale is crucial, because some cell types—especially fibroblasts—are rare and difficult to isolate in sufficient numbers in any one study.
Fibroblasts: The Shapers of the Tumor Landscape
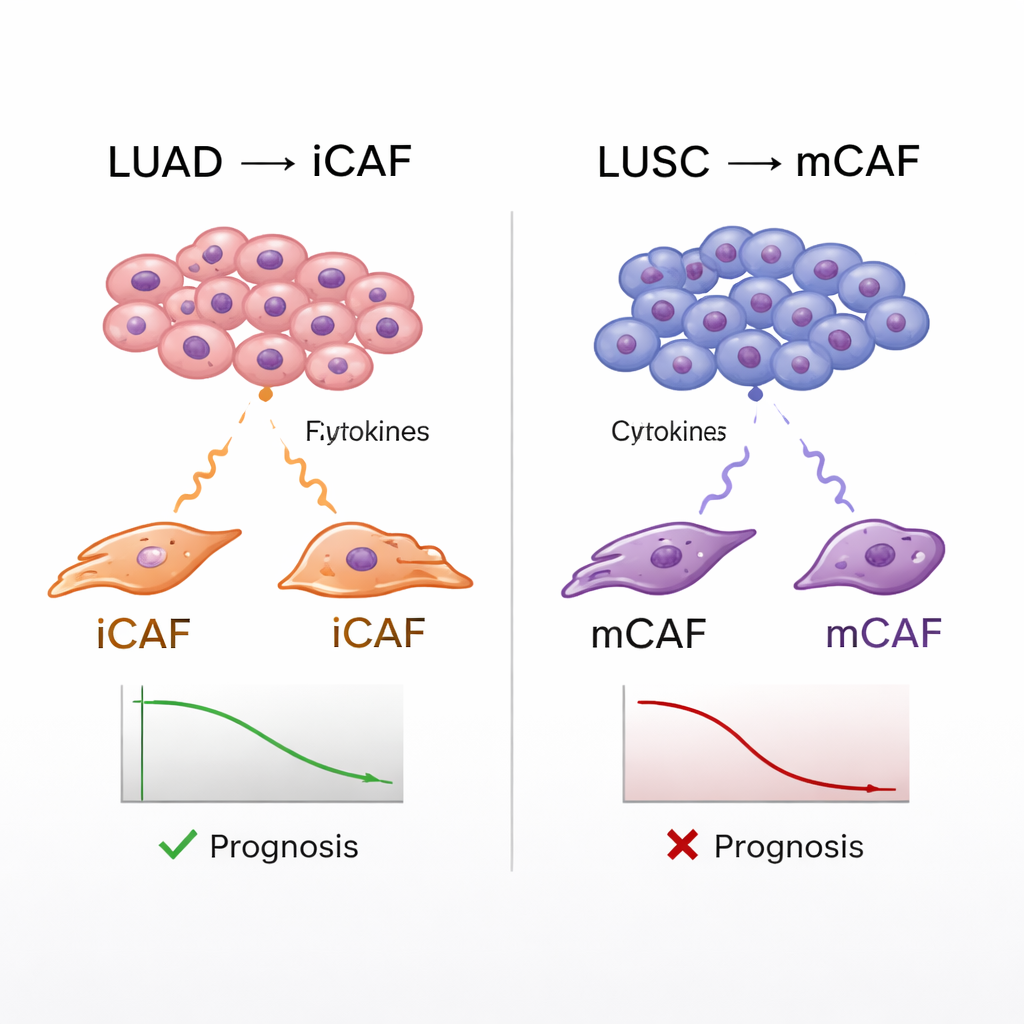
Fibroblasts are support cells that build and remodel connective tissue. In tumors, they become cancer-associated fibroblasts (CAFs), which can either restrain or promote tumor growth depending on their state. By focusing on over 8,700 fibroblasts, the authors identified five major CAF subtypes: myofibroblastic CAFs (mCAFs), inflammatory CAFs (iCAFs), vascular-associated CAFs (vCAFs), cycling CAFs (cCAFs), and antigen-presenting CAFs (apCAFs). The balance of these subtypes differed strikingly between LUAD and LUSC. LUAD tumors tended to harbor more iCAFs, which secrete high levels of inflammatory molecules, whereas LUSC tumors were richer in mCAFs, which produce stiff, fibrous tissue and help shape the tumor’s physical scaffold.
Cancer Cells Teach Fibroblasts Their Roles
To test whether cancer cells themselves instruct fibroblasts to adopt these identities, the researchers grew normal lung fibroblasts in dishes alongside either LUAD or LUSC cell lines. When exposed to LUAD cells, fibroblasts switched on genes typical of iCAFs, including well-known inflammatory signals such as IL-6 and certain chemokines. When paired with LUSC cells, the same fibroblasts instead turned on mCAF genes involved in muscle-like contraction and collagen production. Analyses of cell-to-cell communication suggested that LUAD cells use cytokines such as IL-1, LIF, and OSM to drive the inflammatory iCAF state, while LUSC cells rely more on mechanical cues and noncanonical WNT signaling to push fibroblasts toward the matrix-building mCAF state.
Linking Fibroblast Types to Patient Outcomes
The atlas also allowed the team to connect CAF subtypes to real-world patient data. Using large cancer databases, they estimated how strongly each patient’s tumor expressed gene signatures for the different fibroblast types, then compared these scores with survival. Across both LUAD and LUSC, tumors enriched in mCAFs were linked to poorer outcomes, consistent with the idea that a dense, fibrous capsule around tumors can aid cancer progression and block immune attack. iCAFs, however, showed a split personality: in LUSC, high iCAF signatures also predicted worse survival, whereas in LUAD they were associated with better outcomes. Further analyses suggested that in LUSC, iCAFs help attract neutrophils—immune cells that, in this context, appear to suppress helpful T cells—creating an especially hostile environment for effective immune responses.
What This Means for Future Lung Cancer Care
For a non-specialist, the key message is that not all support cells in lung tumors are alike, and the same fibroblast subtype can have opposite meanings depending on the cancer context. LUAD and LUSC do not just differ in the DNA of their cancer cells; they build different microscopic ecosystems that shape how tumors grow and how patients fare. By charting these ecosystems cell by cell, this study identifies specific fibroblast populations that could serve as markers of prognosis or as targets for future therapies—ideally allowing treatments to be tailored not only to the cancer type, but also to the mix of cells that surround and sustain it.
Citation: Hirano, Y., Suzuki, H., Nakayama, J. et al. An integrated single-cell lung cancer atlas reveals distinct fibroblast phenotypes between adenocarcinoma and squamous cell carcinomas. npj Precis. Onc. 10, 72 (2026). https://doi.org/10.1038/s41698-026-01279-3
Keywords: lung cancer, tumor microenvironment, fibroblasts, single-cell RNA sequencing, cancer prognosis