Clear Sky Science · en
Validation of a methylation-based, tissue-free MRD assay in colorectal cancer patients from the GALAXY study
Why a Blood Test After Cancer Surgery Matters
For people treated for colorectal cancer, one of the biggest worries after surgery is, “Will my cancer come back?” Today, doctors mostly rely on scans and routine checkups to look for signs of relapse, but these can miss very small amounts of remaining cancer. This study tests a new kind of blood test that looks for tiny traces of tumor DNA left in the body—long before a scan would show anything—so treatment can be better tailored to each patient’s real risk.
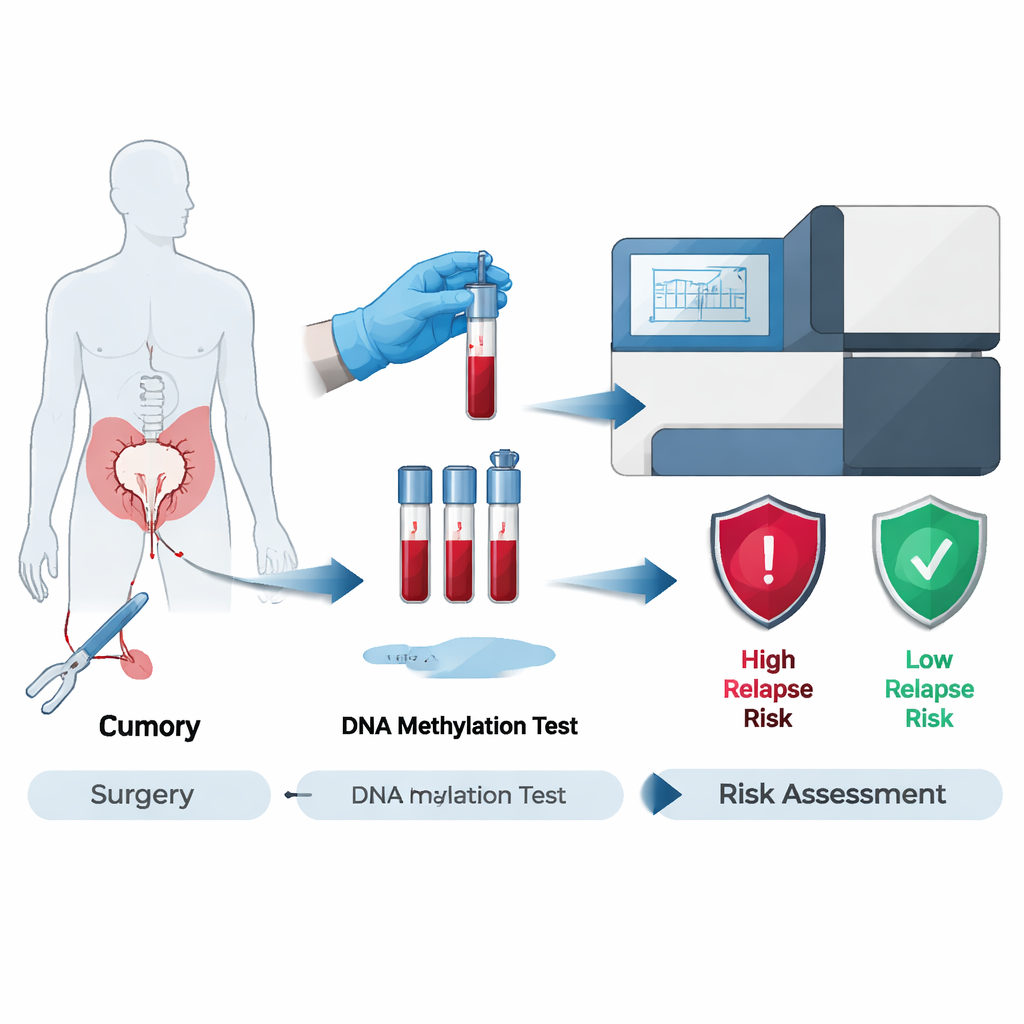
A Simple Blood Test With a Sophisticated Twist
The researchers evaluated a test called the Latitude assay, which searches blood for bits of DNA shed by colorectal tumors. Rather than needing a piece of the original tumor, this test is “tissue-free”: it looks for cancer-specific chemical tags on DNA, known as methylation patterns, that are common in colorectal cancer. Because it doesn’t require tumor tissue, the test can be used even when tissue samples are missing, poor quality, or take too long to process—common hurdles in real-world care.
Following Patients Through Surgery and Beyond
The team examined 195 patients from the larger GALAXY study in Japan, all of whom had surgery intended to remove their colorectal cancer completely. Blood samples were taken a few weeks after surgery, and then repeatedly over the next months to years. The researchers focused on two key time frames. The first, called the minimal residual disease (MRD) window, covered blood tests done 2–10 weeks after surgery. The second, the post-treatment surveillance window, followed patients after they had finished any chemotherapy or, if they did not receive it, after surgery alone. Across these periods, 1,230 blood samples were analyzed to see whether tumor DNA was detectable and how that related to who later relapsed.
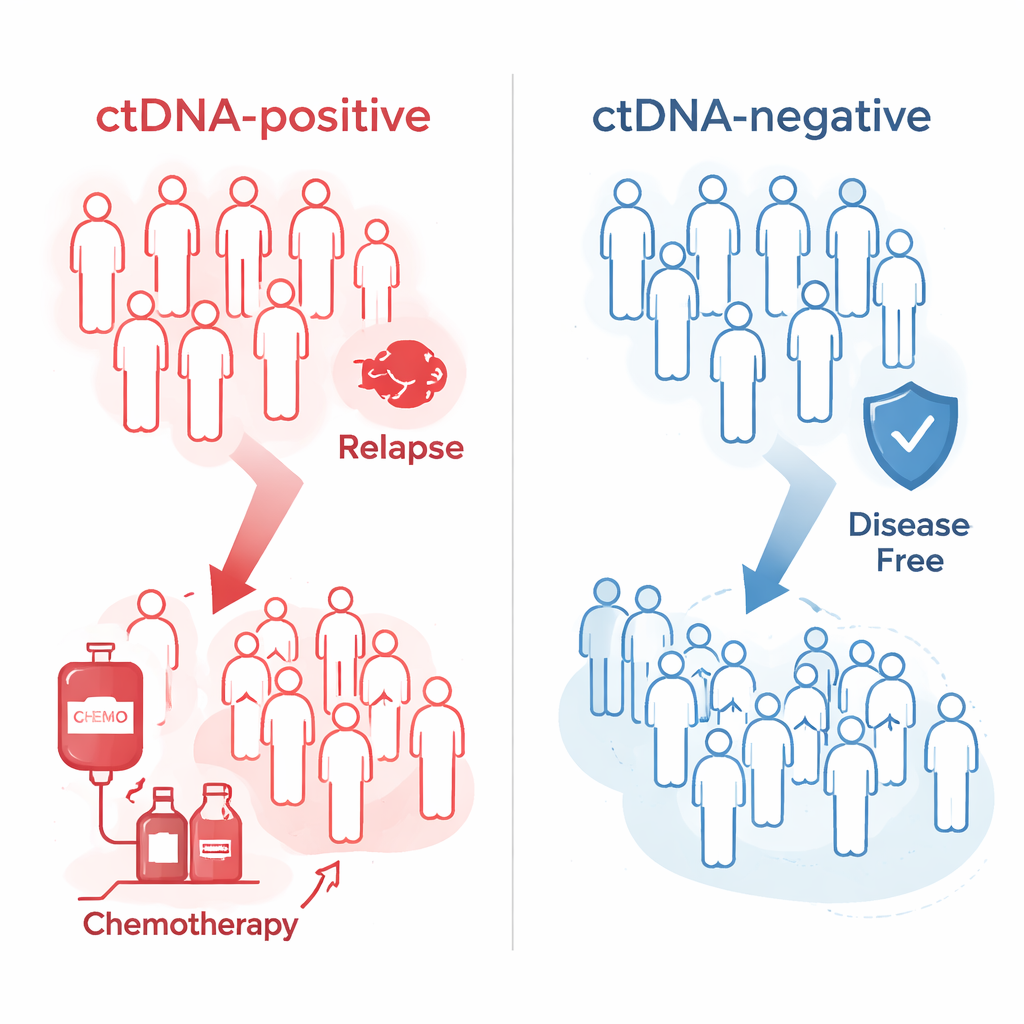
Early Warning of Who Relapses and Who Stays Well
The results were striking. In the early MRD window, about one in four patients tested positive for tumor DNA. Of those, nearly nine out of ten eventually relapsed, compared with fewer than two in ten of those who tested negative. Looking further out in time, when the researchers used repeated testing during surveillance, the blood test correctly flagged about 84% of patients who would go on to relapse, while remaining negative in over 90% of patients who stayed disease-free. On average, the test turned positive about four and a half months before the cancer was confirmed by scans or other clinical evidence, giving a meaningful head start for doctors and patients.
Guiding Who Truly Needs Chemotherapy
The study also asked whether this blood test could help decide who benefits from chemotherapy after surgery. Among patients with higher-risk stage II or stage III disease, those who were tumor-DNA positive clearly gained from receiving chemotherapy: their risk of the cancer coming back dropped dramatically compared with similar patients who did not get chemotherapy. In contrast, patients whose blood was tumor-DNA negative did not show a meaningful benefit from chemotherapy. When the researchers tracked patients already on chemotherapy, those whose blood stayed positive—or briefly turned negative and then positive again—had a much higher chance of relapse than those whose blood remained or became negative during treatment.
What This Means for Patients
To a non-specialist, the takeaway is straightforward: this methylation-based blood test can act like an ultra-sensitive smoke detector for lingering colorectal cancer after surgery. A positive result signals a high chance that cancer cells remain, often months before they can be seen on scans, and identifies patients who are much more likely to benefit from extra treatment. A negative result suggests a low risk of relapse and may spare some patients from the side effects of chemotherapy they are unlikely to need. While more trials are underway to refine how best to use this approach, the study supports a future in which post-surgery care for colorectal cancer is guided not just by what doctors see under the microscope, but by what they can detect in a simple vial of blood.
Citation: Nakamura, Y., Reiter, J.G., Natarajan, P. et al. Validation of a methylation-based, tissue-free MRD assay in colorectal cancer patients from the GALAXY study. npj Precis. Onc. 10, 70 (2026). https://doi.org/10.1038/s41698-026-01277-5
Keywords: colorectal cancer, circulating tumor DNA, minimal residual disease, liquid biopsy, adjuvant chemotherapy