Clear Sky Science · en
TheraMind: a multi-LLM ensemble for accelerating drug repurposing in lung cancer via case report mining
Why this matters for patients and doctors
For people with advanced lung cancer, time is precious and treatment options often run out quickly. Developing brand-new drugs can take more than a decade, but scattered clues about helpful therapies may already be hidden in published patient stories called case reports. This article introduces TheraMind, an artificial intelligence system that combs through thousands of these reports to uncover existing medicines that might be safely reused to help people with hard-to-treat lung cancer.
The challenge of finding new uses for old drugs
Non-small cell lung cancer is the world’s deadliest cancer, with survival rates remaining very low once the disease has spread. Discovering and testing new drugs from scratch is slow, costly, and risky. In contrast, “repurposed” drugs are already approved for other conditions, so their safety is much better known. Oncologists are interested in such drugs, especially for patients who have exhausted standard treatments, but they need real-world evidence that a drug has actually helped someone with lung cancer—not just promising lab data. These clinical hints often appear in individual case reports buried deep in the medical literature, which are difficult and time-consuming to find by hand.
A digital reader for thousands of patient stories
The researchers built TheraMind to act as a tireless, careful reader of case reports. Starting from 18 drug candidates suggested by earlier genomic and laboratory studies, the system automatically pulled 10,023 case reports from PubMed, the largest medical article database. It then fed the text of each report to three separate language models—different AI “readers” trained to understand medical writing. For every report, these models answered the same four simple questions: Is the patient described as having non-small cell lung cancer? Was the study drug given specifically to treat it? Was treatment stopped early? And did the patient experience a good clinical outcome?
How the AI team reaches a joint decision
Once TheraMind turns each report into a set of yes-or-no answers, several layers of decision-making come into play. One method uses a hand-crafted decision tree that requires all four conditions to be satisfied before calling a case relevant for drug repurposing. Another lets a single model weigh the evidence more flexibly, considering both the yes-or-no answers and the short explanations it produced. The most powerful method combines three separate classifiers, each based on a different model, and accepts a report only when at least two of the three agree it is relevant. This “majority vote” approach helps cancel out individual mistakes, much like asking several independent doctors to review the same chart.
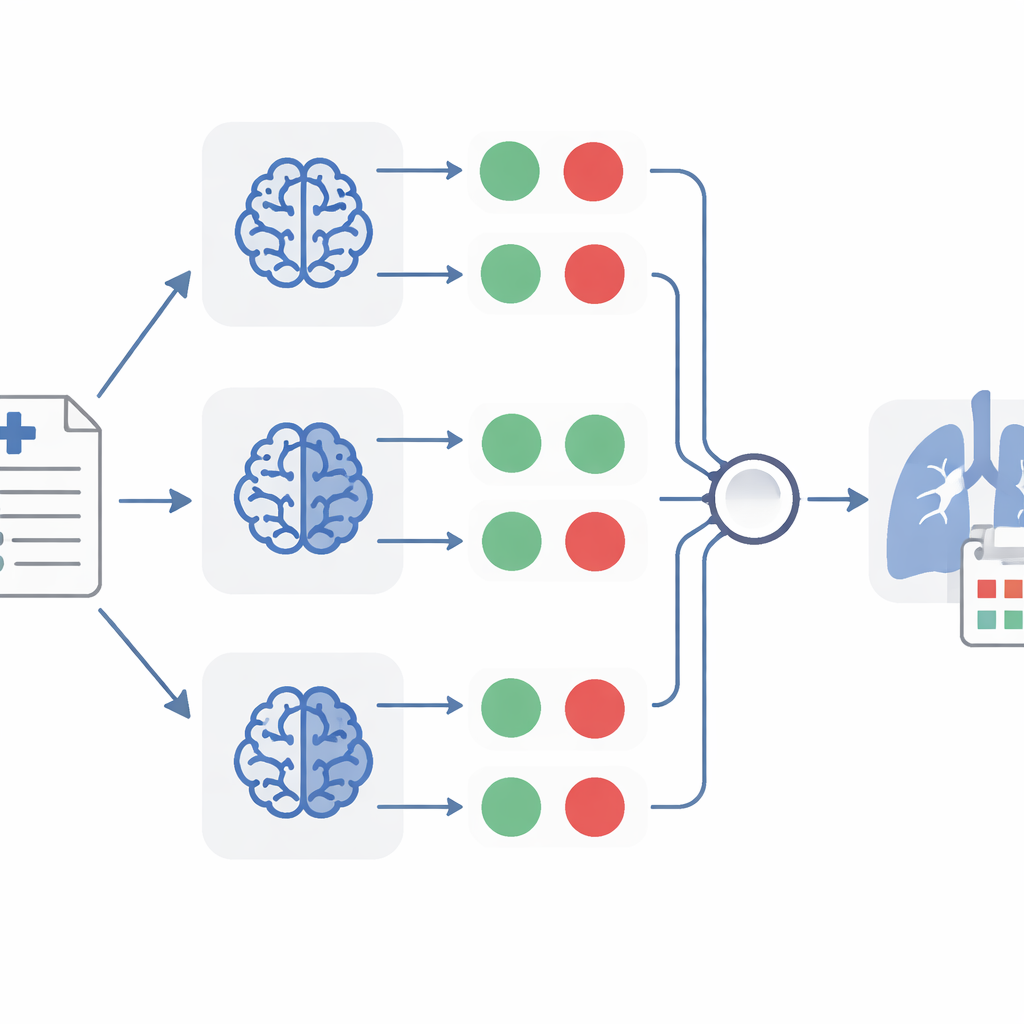
What TheraMind discovered in lung cancer reports
Applied to the large collection of case reports, TheraMind flagged 26 as strongly supporting the use of ten different drugs in non-small cell lung cancer. The ensemble method achieved a recall of about 92 percent, meaning it captured nearly all truly relevant reports, while maintaining a very high specificity of 99.7 percent, so that very few irrelevant reports slipped through. For each selected report, the system also extracted key patient details—such as age, sex, medical history, condition, and the suspected drug—into a consistent data format, and generated a short, plain summary of the case. Human reviewers with biology and computer science backgrounds checked these outputs and confirmed that one of the models in particular produced highly accurate and complete extractions.
Beyond lung cancer and into the clinic
To test how flexible the system is, the team also aimed TheraMind at breast cancer and several candidate drugs previously identified by their own research. In that setting, the system reported no matching case reports, accurately reflecting the current scientific record rather than inventing evidence. The authors argue that this kind of structured, transparent pipeline—combining rule-based logic with multiple AI readers—can help close the gap between laboratory discoveries and treatments ready for clinical trials. They highlight both the promise of applying similar methods to other cancers and the need to handle patient privacy carefully if future versions connect directly to electronic health records.
What this means for future treatments
In plain terms, TheraMind turns scattered, unstructured patient stories into organized evidence that doctors and researchers can act on. By rapidly surfacing reports in which existing drugs seem to help people with lung cancer, it can guide which repurposed medicines deserve closer study in clinical trials. While it does not replace medical judgment, this multi-model AI reader offers a way to sift through mountains of medical text and deliver a short, trustworthy list of promising options for patients who urgently need new choices.
Citation: More, V., Lu, L., Ding, Z. et al. TheraMind: a multi-LLM ensemble for accelerating drug repurposing in lung cancer via case report mining. npj Precis. Onc. 10, 95 (2026). https://doi.org/10.1038/s41698-025-01265-1
Keywords: drug repurposing, lung cancer, case reports, large language models, clinical evidence mining