Clear Sky Science · en
Single nuclei and spatial profiling of sacrococcygeal teratomas reveals cellular composition and X inactivation heterogeneity
Why baby spine tumors matter
Sacrococcygeal teratomas are rare tumors that grow at the base of a baby’s spine, sometimes even before birth. They can contain many different tissues—nerve, gut, skin, muscle—all mixed together. Doctors can usually remove them, but some grow dangerously fast, and they appear about three times more often in babies with two X chromosomes than in those with one. This study uses cutting‑edge genetic tools to build a detailed "cellular map" of these tumors and to ask whether unusual behavior of the X chromosome might help explain how the tumors form and why they are so strongly biased toward girls.
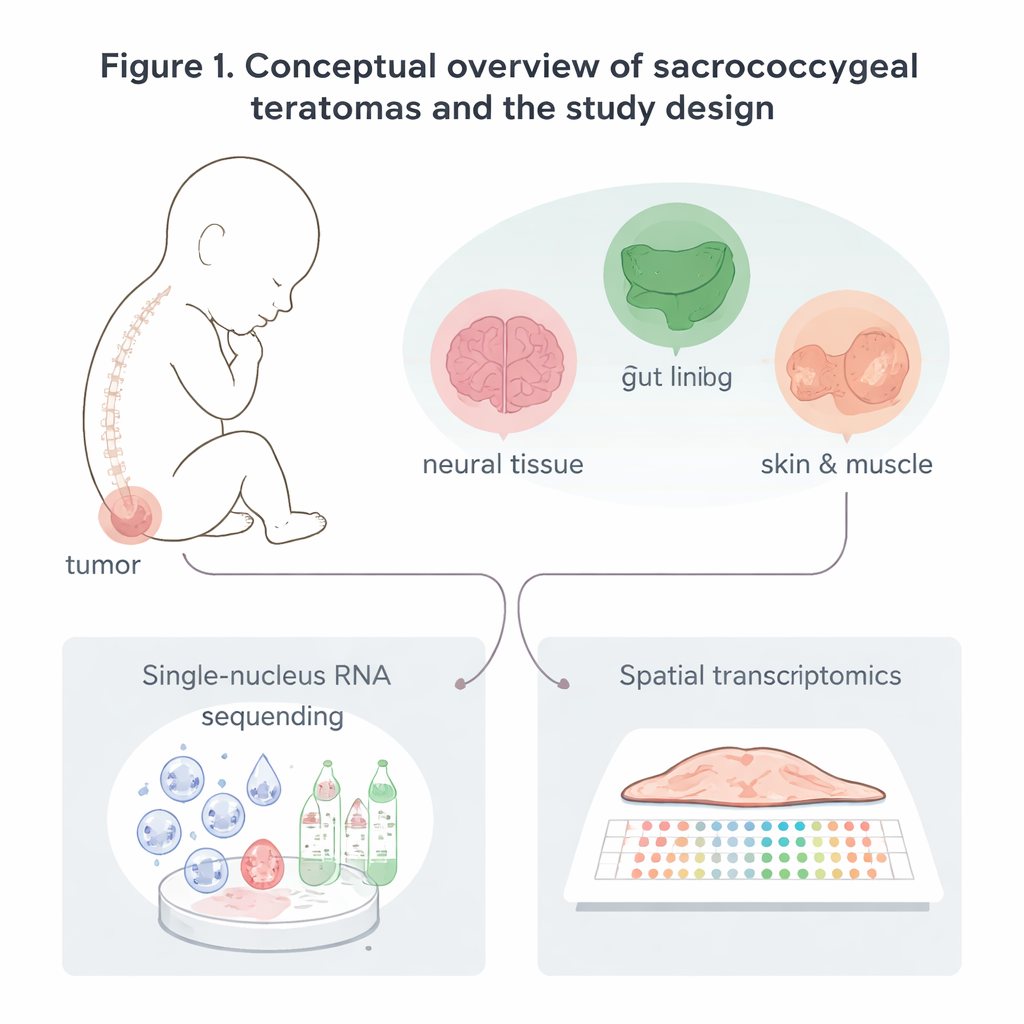
Building a detailed map of tumor cells
The researchers collected eight sacrococcygeal teratomas: six removed after birth and two removed before birth during high‑risk pregnancies. Instead of looking at the tumors under a microscope alone, they read out which genes were active in tens of thousands of individual cell nuclei. They also used spatial transcriptomics, a method that measures gene activity in thin slices of tissue while preserving where each signal came from in the tumor. From these data, they identified five major families of cells: nerve‑related cells, lining (epithelial) cells, support (stromal) cells such as muscle and cartilage, blood vessel cells, and immune cells like macrophages and T cells. Each family contained multiple subtypes, revealing that these tumors are mosaics of many recognizable, body‑like tissues rather than a single uniform mass.
Different tumors, different mixes of tissues
Although every tumor contained cells resembling the three major embryonic layers, their mixtures varied greatly. By treating all cells from a tumor as a combined “voice,” the team compared overall gene‑expression patterns and found three broad tumor groups. One group was rich in epithelial cells that strongly expressed a gene often linked to early embryonic states (POU5F1), another group had fewer epithelial cells but more blood vessels and cartilage‑like tissue, and a third group combined epithelial and nerve‑related cells. These differences lined up with gene programs related to inflammation, blood vessel growth, and nervous system development. In rapidly growing tumors removed before birth, spatial mapping showed that stromal and nerve‑like cells dominated large regions, with clusters of actively dividing cells scattered throughout, suggesting that these fast‑growing tumors are fueled by pockets of cycling support and nerve‑related cells.
Challenging the idea of a hidden stem cell
Because teratomas contain such a variety of tissues, many scientists have suspected they arise from a rare, powerful stem‑like cell similar to the early precursors of eggs and sperm. The team specifically searched for cells that simultaneously switched on hallmark genes of pluripotent stem cells and primordial germ cells. They found scattered expression of some of these genes, but never in a way that marked a clear, unified “master” cell population. Instead, germ‑cell and pluripotency‑related genes appeared in ordinary tumor cell types, such as immune cells or particular epithelial cells. This suggests that, at least in the tumors studied here, any original stem‑like founder either disappeared or left only faint traces in more mature cell populations.
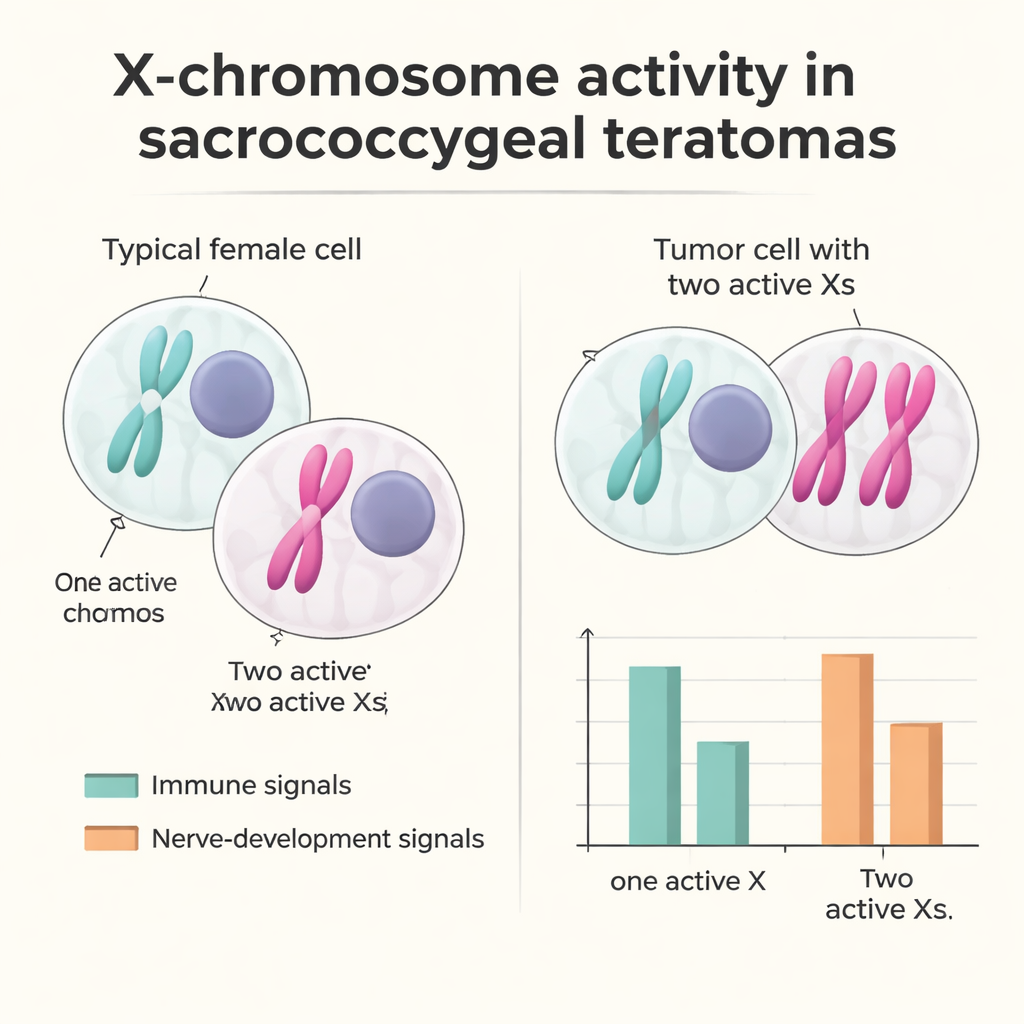
Two active X chromosomes and a quieter immune response
To explore why these tumors are far more common in babies with two X chromosomes, the researchers examined how the X chromosome was regulated inside tumor cells. In typical female cells, one X is switched off to keep gene dosage balanced with males; this inactivation is marked by a molecule called XIST. In one female tumor, they discovered a substantial group of cells that lacked XIST and showed gene‑expression patterns consistent with having two active X chromosomes. These "XaXa‑like" cells most often appeared among epithelial, nerve‑related, and stromal populations. By tracking natural genetic variants, the team showed that many X‑linked genes that should be silent on the inactive X were now active from both copies, indicating failure of the usual inactivation system rather than simple duplication of an already active X. Cells with two active X chromosomes tended to express more genes involved in early development and nerve formation, while those with a normal single active X showed stronger immune and inflammation‑related signals, hinting that X‑dosage changes may subtly tilt the tumor environment away from immune attack.
What this means for patients and families
For families facing a diagnosis of sacrococcygeal teratoma, this work does not yet change day‑to‑day treatment, which still relies on surgery and careful follow‑up. But it does offer a clearer picture of what these tumors are made of and points to new ideas about why they form and why they are more common in babies with two X chromosomes. The study shows that the tumors can be grouped by the kinds of tissues and gene programs they contain, and that in at least one case, cells with two active X chromosomes gain developmental advantages while dialing down immune signals. In the future, such detailed cellular maps could help doctors better predict which tumors are likely to grow aggressively, inspire tests that track risky cell populations, and guide research into therapies that restore normal X‑chromosome balance or boost anti‑tumor immunity.
Citation: Rojas, E.J., Giannikou, K., Huang, B.J. et al. Single nuclei and spatial profiling of sacrococcygeal teratomas reveals cellular composition and X inactivation heterogeneity. npj Precis. Onc. 10, 87 (2026). https://doi.org/10.1038/s41698-025-01262-4
Keywords: sacrococcygeal teratoma, single-cell sequencing, spatial transcriptomics, X-chromosome inactivation, fetal tumors