Clear Sky Science · en
Optical genome mapping as a high-resolution tool for uncovering cytogenetic complex and cryptic alterations in a cohort of patients with MDS and AML
Why hidden DNA changes in blood cancer matter
For people diagnosed with certain blood cancers, doctors increasingly rely on the patient’s DNA to decide how serious the disease is and which treatments to try. But many genetic changes are tiny, tangled, or simply too complex for standard tests to see. This study explores a newer method called optical genome mapping (OGM), which acts like a high‑resolution map of a patient’s chromosomes, to show whether it can uncover hidden changes in two related diseases: myelodysplastic neoplasms (MDS) and acute myeloid leukemia (AML).
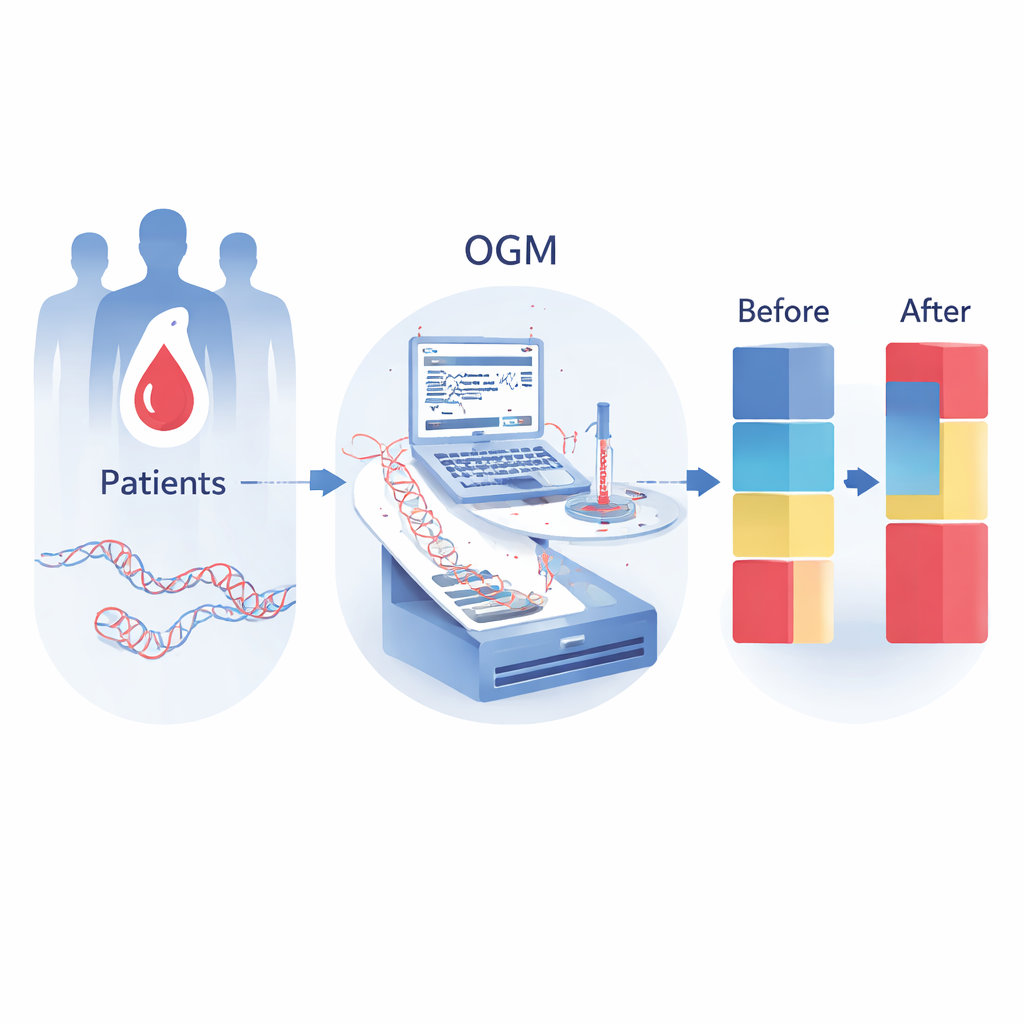
A clearer map of a complex disease
MDS and AML arise when blood‑forming stem cells in the bone marrow acquire DNA damage and begin to grow abnormally, crowding out healthy blood production. Doctors already use chromosome banding, fluorescent probes, and DNA sequencing to look for known mutations and rearrangements, which help predict prognosis and guide therapy. However, these traditional tools can miss “cryptic” changes—small or structurally complex alterations—and often struggle to interpret especially disordered genomes, known as complex karyotypes. This leaves a significant fraction of patients with only a partial picture of their disease biology.
How optical genome mapping works in practice
Optical genome mapping takes ultra‑long stretches of a patient’s DNA, labels specific sequence patterns with fluorescent tags, and then images these molecules as they are aligned to a reference genome. Rather than reading each letter, it measures large‑scale patterns and breaks in the DNA, making it particularly suited to detect structural variants and changes in copy number. In this study, the researchers applied OGM to samples from 150 adults with MDS or AML and compared the results with standard testing methods. They assessed whether OGM could match routine diagnostics and, importantly, whether it could add new information that changed how patients were classified.
Uncovering hidden rearrangements and risk
OGM produced usable results in all patients and agreed with conventional karyotyping in 97 percent of cases, confirming that it reliably captures the same major abnormalities. Yet it went substantially further: in 80 percent of patients it revealed extra details or new findings, such as precise breakpoints, partners in chromosome swaps, and small gains or losses that earlier tests had missed. Many of these involved genes already known to influence blood cancers, including MECOM, KMT2A, RUNX1, NUP98, and TP53. As a result, 33 patients were reclassified—for example, from apparently normal chromosomes to clearly abnormal or complex ones—sometimes shifting them into higher‑risk categories under current clinical scoring systems. OGM also solved cases where standard methods could not interpret the karyotype at all, turning previously “unreadable” results into actionable information.
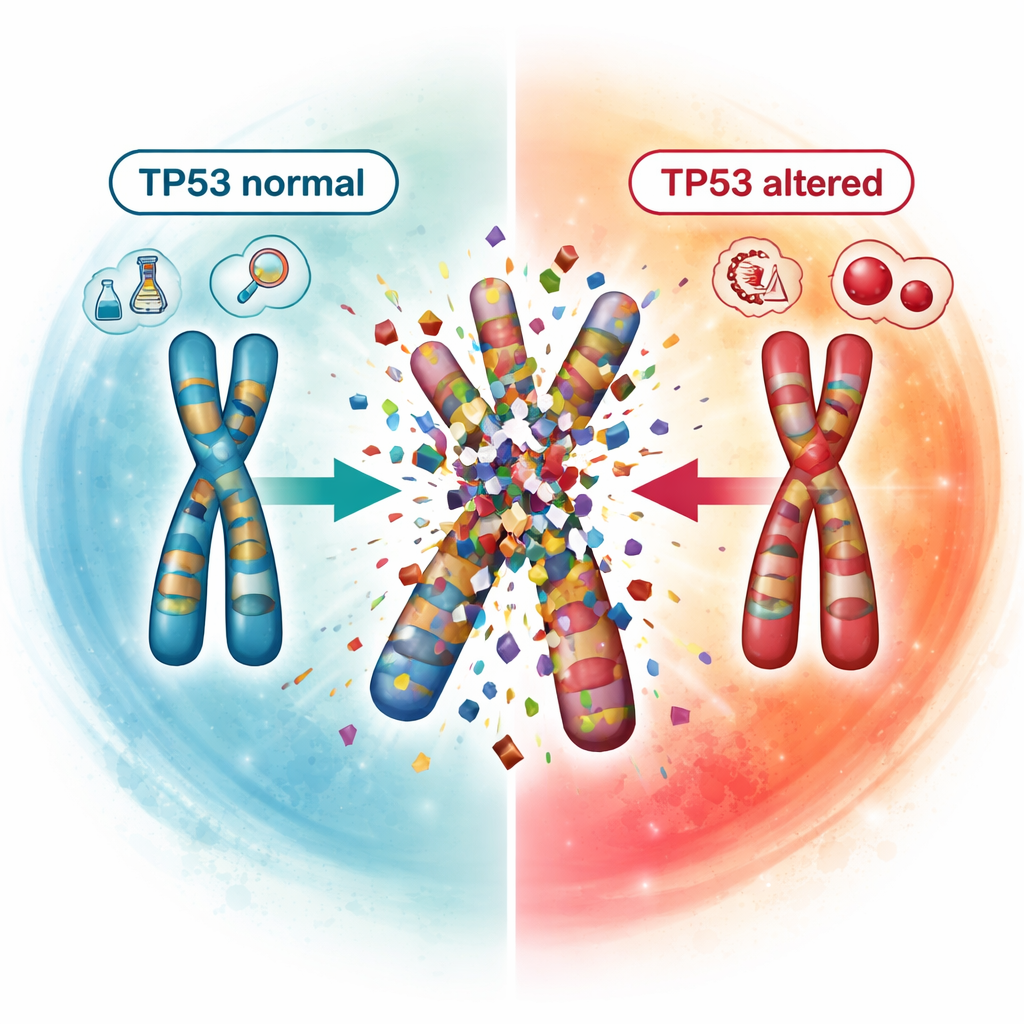
Seeing catastrophic chromosome events
A striking strength of OGM was its ability to expose catastrophic chromosome events, collectively called chromoanagenesis. In these events, one or more chromosomes shatter and are stitched back together in a chaotic fashion, producing numerous gains, losses, and rearrangements in a single cellular crisis. The team identified such events in 17 patients and showed a strong link with damage to the TP53 gene, a key guardian of genome stability. None of the patients with normal TP53 showed these catastrophic patterns, whereas they were common in those with one or both TP53 copies altered, especially when both were affected. This connection supports the idea that disabling TP53 helps drive extreme genome instability and may be one reason these patients fare poorly.
What this means for patients
For patients, the message is that a more detailed view of their cancer’s DNA can change how doctors understand and manage the disease. Optical genome mapping does not replace all other tests, and it has limits, such as difficulty detecting very small changes or very rare cell populations. But by capturing subtle and complex chromosome damage in a single assay, it can refine risk estimates, clarify puzzling results, and highlight genes and pathways that might be targeted by future treatments. As OGM becomes more widely available and integrated with sequencing, it may help move MDS and AML care closer to truly personalized medicine, where therapy is shaped by the full, high‑resolution genetic map of each patient’s disease.
Citation: Torres-Hernández, N., Mora, E., García-Ruiz, C. et al. Optical genome mapping as a high-resolution tool for uncovering cytogenetic complex and cryptic alterations in a cohort of patients with MDS and AML. npj Precis. Onc. 10, 85 (2026). https://doi.org/10.1038/s41698-025-01258-0
Keywords: optical genome mapping, acute myeloid leukemia, myelodysplastic syndromes, chromosome rearrangements, TP53