Clear Sky Science · en
Neoadjuvant sintilimab, albumin-bound paclitaxel, and carboplatin for locally advanced, resectable esophageal squamous cell carcinoma: clinical study and mechanistic exploration
Turning the Tide Against a Deadly Throat Cancer
Esophageal cancer is one of the deadliest cancers worldwide, often discovered only after it has advanced deep into the chest. Many patients can still have surgery, but even with today’s best chemotherapy and radiation, the risk of the disease coming back is high. This study tests a new way to boost the body’s own defenses before surgery and looks under the microscope at how tumors and their surrounding cells change in response. The findings hint at more effective treatment and a path toward truly personalized care.
A New Three-Drug Plan Before Surgery
The researchers enrolled 24 people in China with a common and aggressive form of esophageal cancer called squamous cell carcinoma. All had tumors that were advanced but still removable by surgery. Before going to the operating room, each patient received three cycles of a combination treatment: sintilimab, an immune-activating antibody that helps T cells recognize cancer; plus two standard chemotherapy drugs, albumin-bound paclitaxel and carboplatin. After this “neoadjuvant” phase, patients underwent surgery to remove the esophagus tumor, and most then continued on sintilimab afterward.
Stronger Tumor Shrinkage and Encouraging Survival
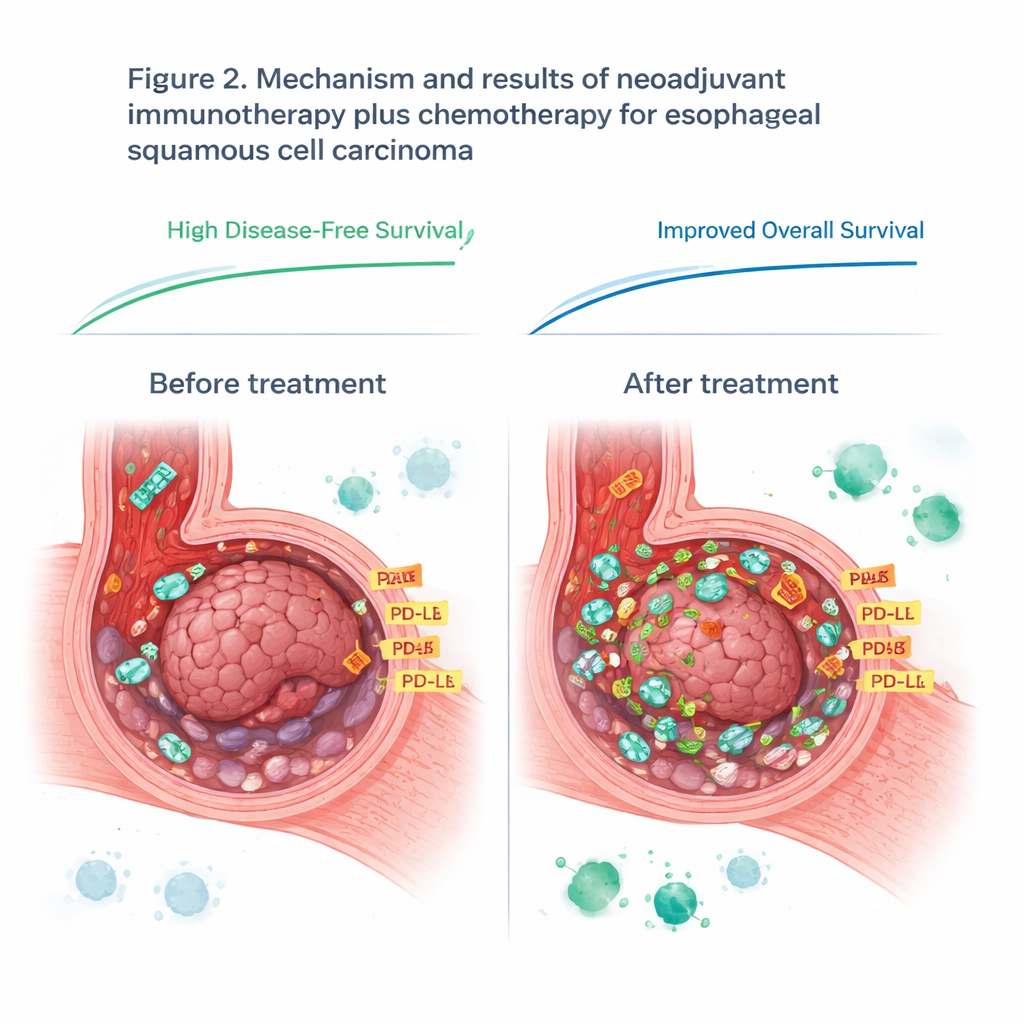
When surgeons and pathologists examined the removed tumors, they found that this pre-surgery regimen had produced deep shrinkage in many patients. About 42% showed what doctors call a major pathological response, meaning that only a small fraction of living cancer cells remained. One in three patients had no detectable cancer cells at all in the main tumor area. On scans and clinical follow-up, the results were also promising: three years after treatment, roughly three out of four patients were alive without signs of the disease returning, and nearly four out of five were still alive. Importantly, the side effects of this intensified plan were manageable, with no treatment-related deaths and surgical removal remaining safe and feasible.
How the Tumor’s Neighborhood Shapes Response
Not every patient benefited equally, so the team asked why. They focused on the tumor microenvironment—the complex neighborhood of cancer cells, immune cells, and signaling molecules. Using targeted protein measurements on carefully dissected tumor samples, they compared those who responded well with those who did not. Before treatment, they found 14 proteins that differed between these two groups. One surface protein, CD44, stood out: tumors with higher CD44 levels tended to respond better to the sintilimab-based therapy. After treatment, many proteins involved in immune activity and DNA repair were reduced in responders, suggesting that the combination therapy had successfully disrupted the cancer’s support systems and reshaped the local immune landscape.
Rethinking a Popular Immune Marker
The study also examined PD-L1, a protein often used as a compass for deciding who might benefit from immune checkpoint drugs. Before treatment, PD-L1 levels on tumor samples did not predict who would do well, challenging current practice in this cancer type. However, PD-L1 levels rose in every patient during treatment, likely because the activated immune system flooded the tumor with inflammatory signals. After therapy, higher PD-L1 levels in the remaining tumor tissue were more closely linked to how well the immunotherapy was working. This suggests that timing matters: a single snapshot of PD-L1 before treatment may miss crucial changes that unfold once the immune system is engaged.
What This Means for Patients and the Future
For people facing surgery for advanced esophageal cancer, this study offers two hopeful messages. First, combining sintilimab with chemotherapy before surgery appears to shrink tumors more deeply and may improve the chances of long-term survival, without adding dangerous side effects. Second, the work shows that the tumor and its microenvironment are not fixed; they are reshaped by treatment in ways that can be tracked through proteins like PD-L1 and CD44. In the future, such molecular fingerprints could help doctors select and adjust therapies in real time, turning more “cold” tumors that ignore the immune system into “hot” tumors that invite a powerful, lasting immune attack.
Citation: Wu, H., Jiang, Q., Li, X. et al. Neoadjuvant sintilimab, albumin-bound paclitaxel, and carboplatin for locally advanced, resectable esophageal squamous cell carcinoma: clinical study and mechanistic exploration. npj Precis. Onc. 10, 82 (2026). https://doi.org/10.1038/s41698-025-01248-2
Keywords: esophageal cancer, immunotherapy, neoadjuvant therapy, PD-1 inhibitors, tumor microenvironment