Clear Sky Science · en
Design and evaluation of a custom circulating tumour DNA assay to detect endometrial cancer recurrence
Why this blood test matters
For many people treated for womb (endometrial) cancer, the biggest worry is whether the disease will come back, and how soon doctors would notice. Today, checking for a return often relies on scans and internal examinations, which can be uncomfortable, expensive, and sometimes miss early signs. This study describes a new, highly focused blood test that looks for tiny fragments of tumor DNA circulating in the bloodstream, with the goal of spotting a returning cancer earlier and more simply, and even hinting at which medicines might work best.
From tissue to a tailored blood test
The researchers set out to build a test made specifically for endometrial cancer rather than a broad "one size fits all" cancer panel. They combed through large international datasets of tumor genetics, alongside detailed DNA sequencing of 42 tumors from their own patients. From hundreds of endometrial cancers, they pulled out the changes in DNA that appeared again and again, especially in genes already known to drive this disease. They then designed a compact test panel that homes in on 1,848 of these hotspots plus several markers linked to a feature called microsatellite instability, all packed into 635 short DNA regions that can be read in a single run on an automated sequencing machine. 
Putting the panel through its paces
Before trying the new test on patients’ blood, the team checked whether it could reliably find known mutations at very low levels. Using commercial reference samples that mimic the DNA fragments seen in cancer patients, they showed the panel could detect almost all known changes even when they made up less than 1% of the DNA present. In one test, the panel picked up a signal when the altered DNA represented just 0.74% of the sample, which is close to the limit of what most current technologies can see. They also confirmed that the same key mutations seen in tumor tissue were detected by the panel in over 95% of primary tumors, showing that the target regions chosen were well matched to real endometrial cancers.
Testing real patients after treatment
The crucial step was to see how the blood test performed in women who had already been treated for endometrial cancer. The study followed 39 such patients: 14 whose cancer did eventually return and 25 who remained free of disease during long-term follow-up. Each woman gave a blood sample around a clinic visit, and the team searched for tumor-linked DNA fragments using their panel. Among those whose cancer recurred, the test correctly found tumor DNA in 10 out of 14 cases. Among those who stayed well, only one of 25 had a suspicious result, giving an overall sensitivity of about 71% and specificity of 96%. Importantly, in eight of the ten true-positive cases the pattern of mutations suggested specific drugs or targeted treatments that might help guide care at recurrence. 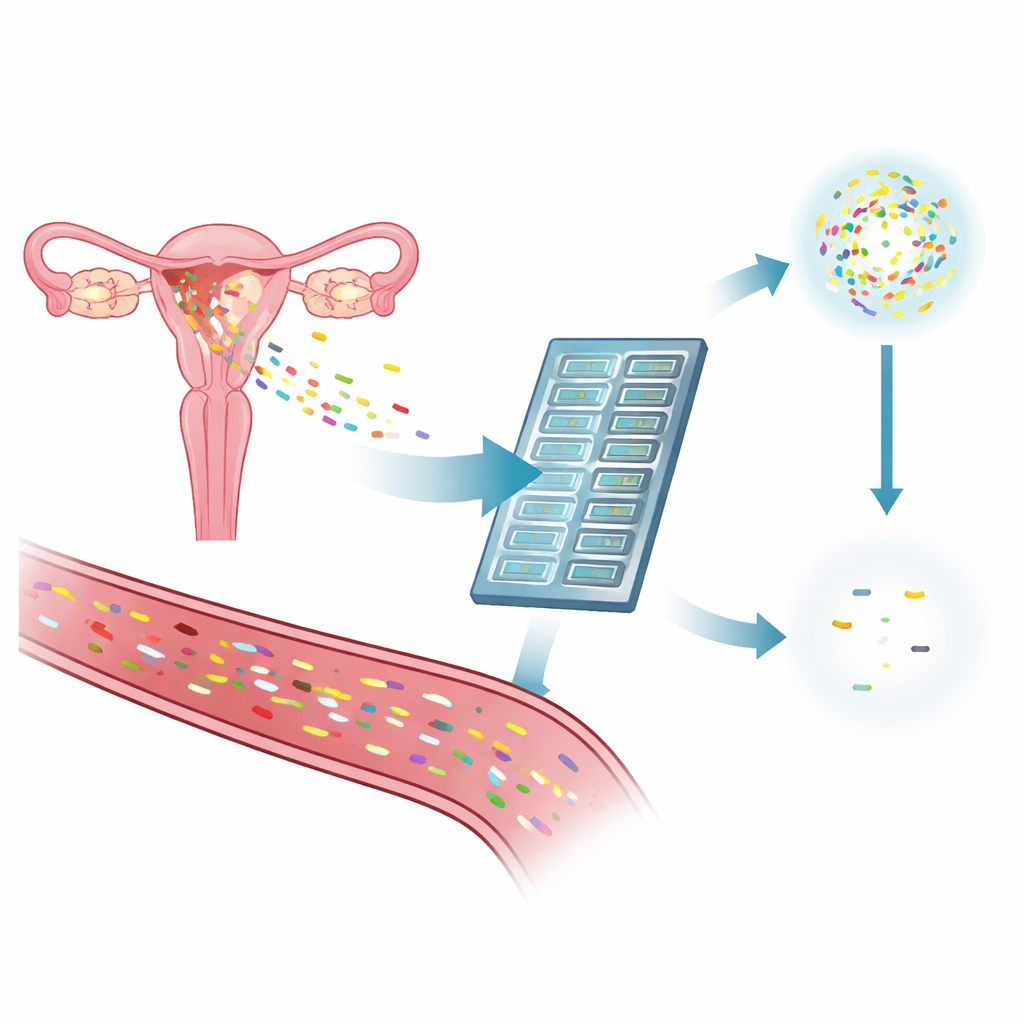
What the test can and cannot see
Not every recurrence showed up in the blood. In four women with returning cancer, no clear tumor DNA signal was detected. In several of these, very low-level hints of relevant mutations were visible but did not cross strict quality cutoffs, suggesting that extremely small or slow-growing recurrences may still slip under the radar. The panel also struggled to read some of the more technically tricky DNA regions used to measure microsatellite instability, indicating that future design tweaks are needed. However, the test worked well for the vast majority of its targets, and in three women who actually had a different new cancer (such as breast cancer) instead of endometrial cancer recurrence, the panel generally stayed negative or produced results that fit with the second tumor rather than the original one, supporting its disease focus.
What this could mean for follow-up care
The study shows that a focused, endometrial-cancer–specific blood test can be run in a routine diagnostic lab, delivering results in about 48 hours at a cost projected to be lower than repeated scans and invasive biopsies. Used alongside other clinical information, such a test could reassure many patients when no tumor DNA is found, prompt earlier imaging or treatment when it is detected, and offer clues to targeted therapies based on the mutations present. Although the work was done in a relatively small group and will need to be repeated in larger studies, the message for patients is clear: a simple blood draw may soon play a central role in watching for the return of womb cancer, cutting down on uncomfortable exams and helping doctors act quickly and precisely when problems arise.
Citation: Wadsley, M., Guttery, D., Cowley, C. et al. Design and evaluation of a custom circulating tumour DNA assay to detect endometrial cancer recurrence. npj Precis. Onc. 10, 127 (2026). https://doi.org/10.1038/s41698-025-01246-4
Keywords: endometrial cancer, liquid biopsy, circulating tumor DNA, cancer recurrence, precision oncology