Clear Sky Science · en
Algorithm-assisted individualized therapy design improves survival in a mouse model of triple-negative breast cancer
Why Smarter Chemotherapy Matters
Chemotherapy saves lives, but it is still mostly given in a “one size fits all” way: everyone with a similar cancer often receives the same drug dose on the same schedule. This can mean harsh side effects for some people and not enough benefit for others. The study described here asks a simple but powerful question: what if we could use computer algorithms to tune chemotherapy to each individual, in real time, the way a thermostat adjusts heating in a house? Using an advanced mouse model of an aggressive breast cancer, the researchers show that such tailored dosing can extend survival and limit the development of drug resistance.
The Problem with Routine Cancer Drug Schedules
Today, many patients receive the highest dose of chemotherapy they can tolerate, given at fixed intervals. This so-called maximum tolerated dose protocol does not account for how fast a particular tumor grows, how a patient’s body handles the drug, or how that tumor responds over time. As a result, tumors may shrink at first but then return as more drug-resistant versions of themselves, while patients bear the brunt of strong side effects. Earlier attempts to improve this, such as “metronomic” chemotherapy using smaller, more frequent doses, have produced mixed results in clinical trials and have not clearly defined how best to choose doses and timing. There is a clear need for a more rational, individualized way to schedule chemotherapy.
Using Algorithms to Tailor Treatment
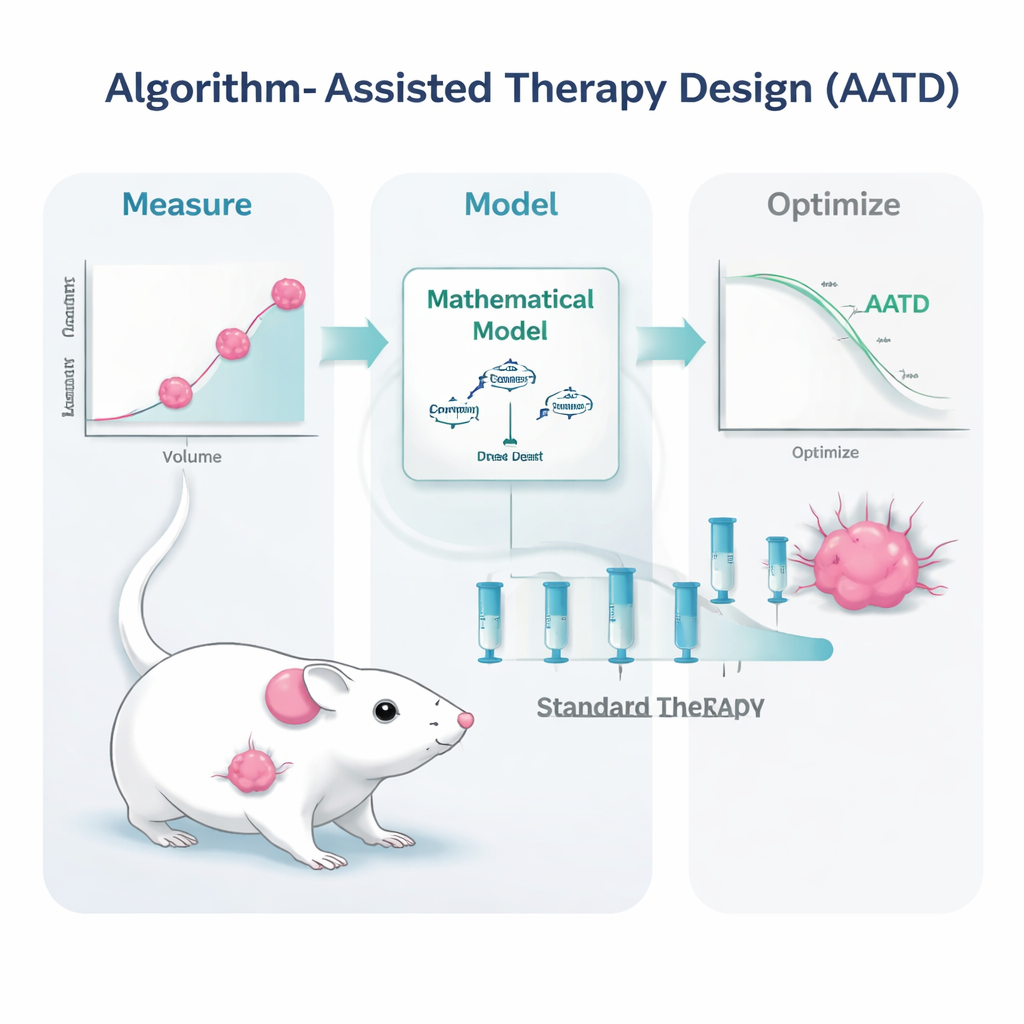
The research team tackled this challenge in mice carrying triple-negative breast tumors, a form of breast cancer that in humans is especially hard to treat and still relies almost entirely on chemotherapy. The mice were treated with a widely used drug formulation, pegylated liposomal doxorubicin. Instead of repeating high-dose treatment only when tumors regrew to a set size, the scientists built mathematical models that describe how the tumor grows, dies back under treatment, and how the drug moves through the bloodstream. They then fed the models simple, clinically realistic measurements: repeated tumor size readings and blood levels of the drug. From these inputs, a computer algorithm generated personalized treatment plans for each mouse.
Two Ways to Let the Computer Help
The study tested two flavors of algorithm-assisted therapy design. In one approach, called PDPK, the computer used early data to design a 30-day schedule of relatively small, repeated doses intended to keep the drug level in the blood steady but low—high enough to control the tumor, but low enough to limit toxicity. In the second approach, called model predictive control, the algorithm updated the plan every day based on the latest tumor measurement, adjusting dose sizes to push the tumor steadily downward. Both methods relied on the same basic idea: use a realistic “digital twin” of the tumor-bearing mouse to simulate many possible dosing patterns ahead of time and pick the ones most likely to work best under safety constraints.
Longer Survival and Less Drug Resistance
When the team compared these algorithm-guided regimens with the standard high-dose, tumor-triggered protocol, the differences were striking. Under conventional therapy, tumors often shrank sharply at first but eventually came back, and repeated large doses encouraged the emergence of drug-resistant disease. In contrast, most algorithm-designed treatments kept tumors under much tighter control, extended the time before relapse, and, in many cases, prevented clear signs of resistance during the study period. Across dozens of mice, both PDPK and model predictive control groups lived substantially longer than animals on the standard schedule, with several algorithm-based strategies showing strong, statistically significant survival gains.
What This Could Mean for Future Patients
Although this work was done in mice, its key ingredients—measuring tumor size, monitoring drug levels in blood, and running computer models—are already possible in human patients using modern imaging and laboratory tools. The study suggests that instead of asking, “What is the standard dose?”, doctors could one day ask, “Given this patient’s own tumor behavior and drug handling, what dose and timing will work best right now?” Practical hurdles remain, such as the need for more frequent measurements and the logistics of flexible dosing, but the message is clear: chemotherapy does not have to be blunt and uniform. With the help of algorithms, it can be turned into a more precise, adaptable tool that offers patients a better chance at long-term control of aggressive cancers.
Citation: Gombos, B., Léner, V., Drexler, D.A. et al. Algorithm-assisted individualized therapy design improves survival in a mouse model of triple-negative breast cancer. npj Precis. Onc. 10, 84 (2026). https://doi.org/10.1038/s41698-025-01245-5
Keywords: personalized chemotherapy, triple-negative breast cancer, algorithm-assisted therapy, mathematical tumor modeling, doxorubicin dosing