Clear Sky Science · en
Computational pathology features of immune architecture predict clinically relevant outcomes in small-cell lung cancer (SCLC)
Why the body’s defenders matter in lung cancer
Small-cell lung cancer is one of the most aggressive forms of lung cancer, often spreading quickly and returning after treatment. Doctors can see tumors under the microscope, but the fine-grained patterns of how cancer cells and immune cells sit next to each other are too complex to judge by eye. This study introduces a computer-based method called PhenopyCell that reads those hidden patterns from routine biopsy slides and uses them to predict how long patients are likely to live and how well they may respond to chemotherapy. For patients and families, such insight could one day help tailor treatment instead of relying on a one‑size‑fits‑all plan.
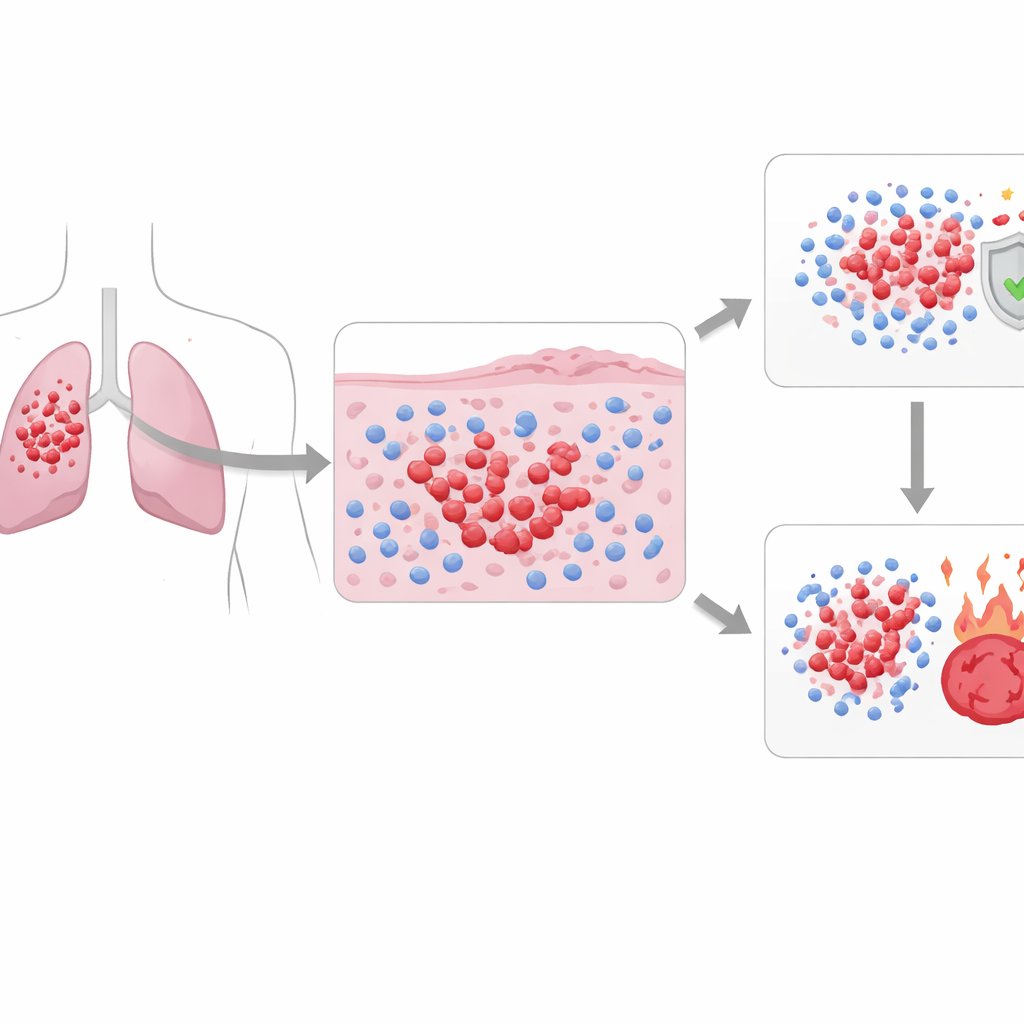
Looking closer at a deadly lung cancer
Small-cell lung cancer makes up about 15% of lung cancers but causes a large share of deaths because it grows and spreads rapidly. It is usually divided into “limited” disease, which can fit in one radiation field, and “extensive” disease, which has spread more widely. Standard first treatment involves powerful platinum-based chemotherapy, sometimes combined with immunotherapy. Many tumors seem to melt away at first, but the cancer often returns within a year, and fewer than one in ten patients survive long term. Current tests based on tumor size, spread, and basic microscopic appearance do not capture why some patients do better than others on the same drugs.
Teaching computers to read tumor neighborhoods
The researchers built PhenopyCell to turn standard stained tissue slides into a detailed map of cell “neighborhoods.” Using existing deep-learning tools, they first taught a computer to find and mark individual tumor cells and immune cells on digital images of biopsies from 281 patients treated at three U.S. centers. They then divided each slide into tumor, nearby tissue, and distant non-tumor regions and calculated over one hundred numerical features. These features describe, for example, how densely immune cells pack around tumor clusters, how far immune cells sit from cancer cells, how diverse the mix of cells is, and how regularly cells are arranged across the slide.
Hidden patterns linked to survival and drug response
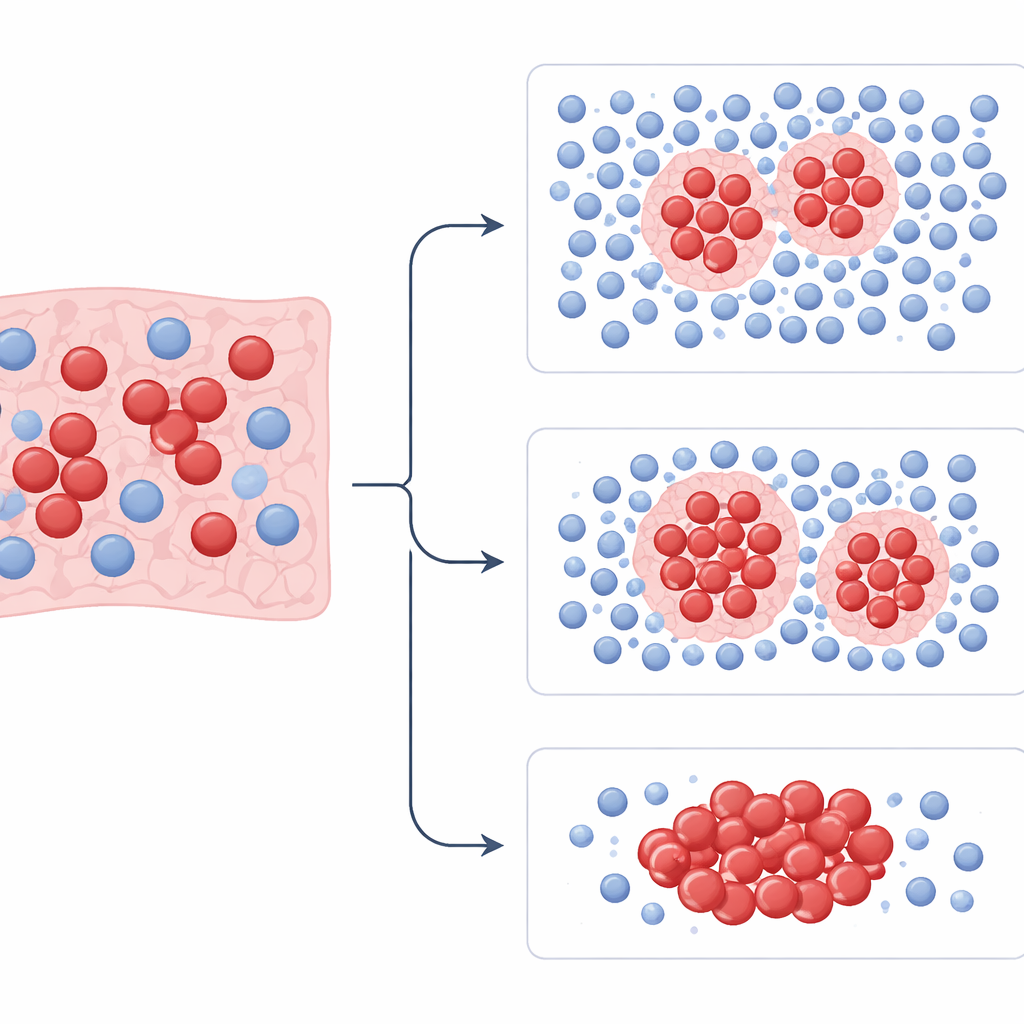
With these measurements in hand, the team trained models to separate patients into higher- and lower-risk groups and to predict who would respond to platinum chemotherapy. They trained the models on one hospital’s cases and then tested them on patients from the other two hospitals. Across all datasets, patterns captured by PhenopyCell were strongly tied to overall survival and to whether chemotherapy worked. Patients whose tumors showed many organized pockets of immune cells tightly surrounding tumor clusters tended to live longer. Those with scattered or sparse immune cells and highly uneven tumor cell distribution were more likely to have aggressive disease and poor outcomes. These links held even when accounting for age, sex, and other clinical factors, and they were seen in both limited and extensive stages of disease.
Beyond black boxes and routine microscope checks
The study also compared PhenopyCell with newer “foundation” artificial intelligence models and with traditional measures of immune cells called tumor-infiltrating lymphocytes. Some large deep-learning models reached high accuracy in the original training group but performed inconsistently when tested on outside patients, raising concerns about stability. Simple counts of immune cells, whether estimated by computer or judged by a pathologist, also did not reliably predict survival. In contrast, PhenopyCell’s focus on how immune and tumor cells are arranged in space, not just how many there are, provided more dependable and easier-to-interpret signals. The features it uses can be related directly to biological ideas such as immune exclusion, areas of dead tissue, and varying tumor shapes.
What this could mean for patients
To a non-specialist, the main message is that the “architecture” of a tumor—how cancer cells and the body’s defenders occupy the battlefield—contains important clues about how the disease will behave and how it will respond to treatment. PhenopyCell shows that these clues can be extracted from the same routine biopsy slides already taken in standard care, without extra procedures. While the study is retrospective and based mostly on patients receiving chemotherapy alone, it suggests that future tools might help doctors identify which patients are at highest risk, who might benefit from more aggressive or novel therapies, and who might avoid unnecessary side effects. Larger, forward-looking studies, especially in patients receiving modern immunotherapies, will be needed before such a system can guide everyday decisions, but this work lays a clear path toward more personalized care in small-cell lung cancer.
Citation: Barrera, C., Jain, P., Corredor, G. et al. Computational pathology features of immune architecture predict clinically relevant outcomes in small-cell lung cancer (SCLC). npj Precis. Onc. 10, 119 (2026). https://doi.org/10.1038/s41698-025-01225-9
Keywords: small-cell lung cancer, computational pathology, tumor microenvironment, immune cell architecture, treatment response prediction