Clear Sky Science · en
Deciphering brain metastasis in epithelial ovarian cancer: multimodal analysis and potential biomarkers
Why this research matters
Ovarian cancer is already a dangerous disease, but in a small number of women it later turns up in a far more fragile place: the brain. These brain metastases are rare yet often deadly, and doctors still have few ways to predict who is at risk or how best to treat them. This study follows that journey from pelvis to brain at the molecular level, using modern DNA and RNA tools to understand how ovarian tumors adapt to the brain and to flag early warning signs that might one day guide more tailored care.
From a common cancer to a rare brain threat
Epithelial ovarian cancer is the most lethal gynecological cancer in developed countries. Most patients respond at first to surgery and chemotherapy, but about 80 percent eventually see the disease return and become resistant to drugs. Recurrences usually stay within the abdomen; only a small fraction of women develop distant spread such as to the brain. As treatments improve and women live longer, however, there is more time for stray cancer cells to seed the brain, and doctors are seeing these metastases more often. Once brain metastases appear, typical survival drops to under a year, underscoring the need to understand how and why this happens.
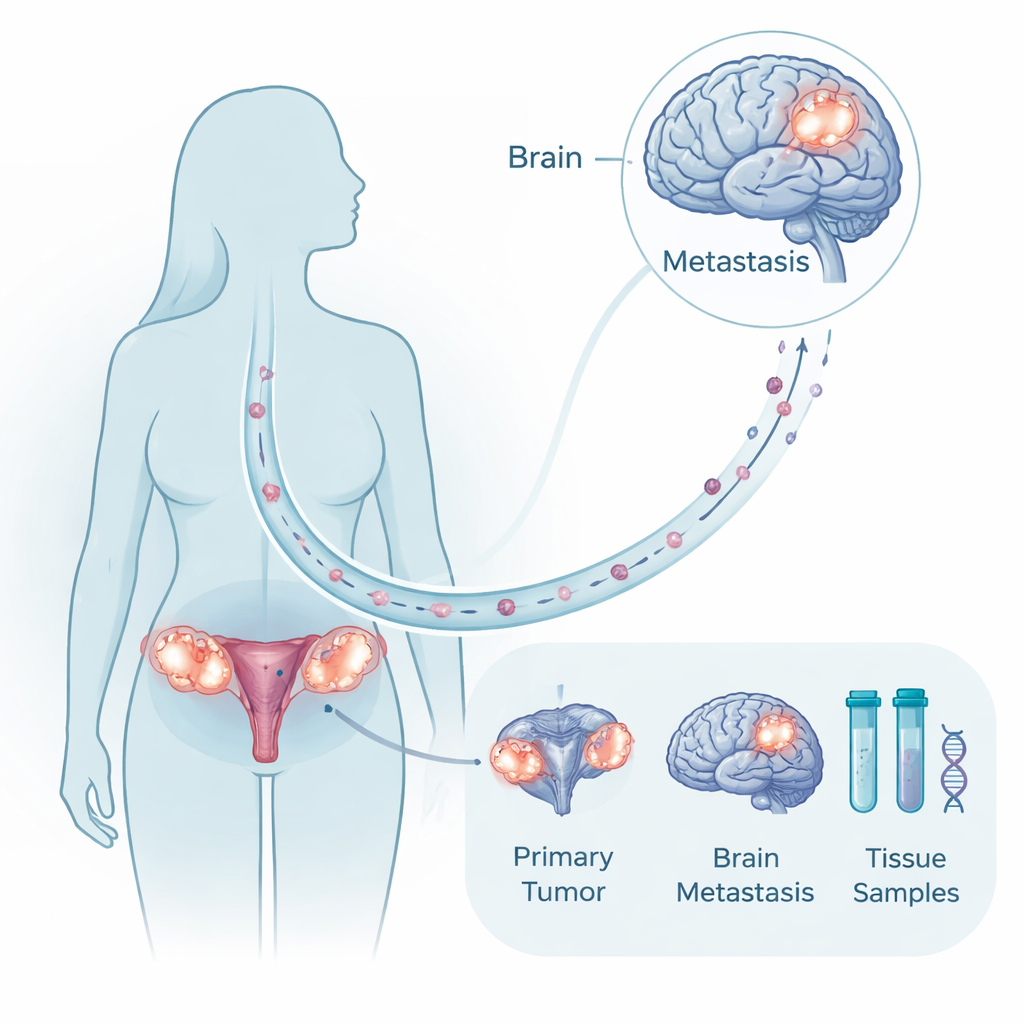
A rare but powerful tissue collection
Because brain surgery is not possible or appropriate for many patients, it is unusual to have tumor samples from both the original ovarian mass and its matching brain deposit in the same woman. The researchers assembled exactly this kind of paired material from ten patients treated over two decades, along with comparison samples from healthy brain and ovary and from other, non-brain metastases. They analyzed tumor DNA to look for mutations and large chromosomal changes, and they profiled RNA to see which genes were turned on or off. This multimodal approach allowed them to ask whether brain tumors are completely new offshoots or closely related cousins of the primary cancer, and which molecular programs change as cells take root in the brain.
Genetic stability, but new behavior
The DNA analysis showed that the original ovarian tumors and their matching brain metastases were remarkably similar. Both carried many of the same cancer-driving mutations, especially in a gene called TP53, and shared an overall “monoclonal” origin, meaning the brain lesions seemed to arise from the same ancestral cancer cell. Large DNA gains and losses were somewhat more common in brain lesions but not overwhelming. This suggests that it is not the acquisition of an entirely new set of mutations that enables brain spread. Instead, the critical shift appears at the level of gene activity: which instructions are being read from the DNA and used by the cancer cells as they adapt to the brain’s unique environment.
How ovarian tumors learn to live in the brain
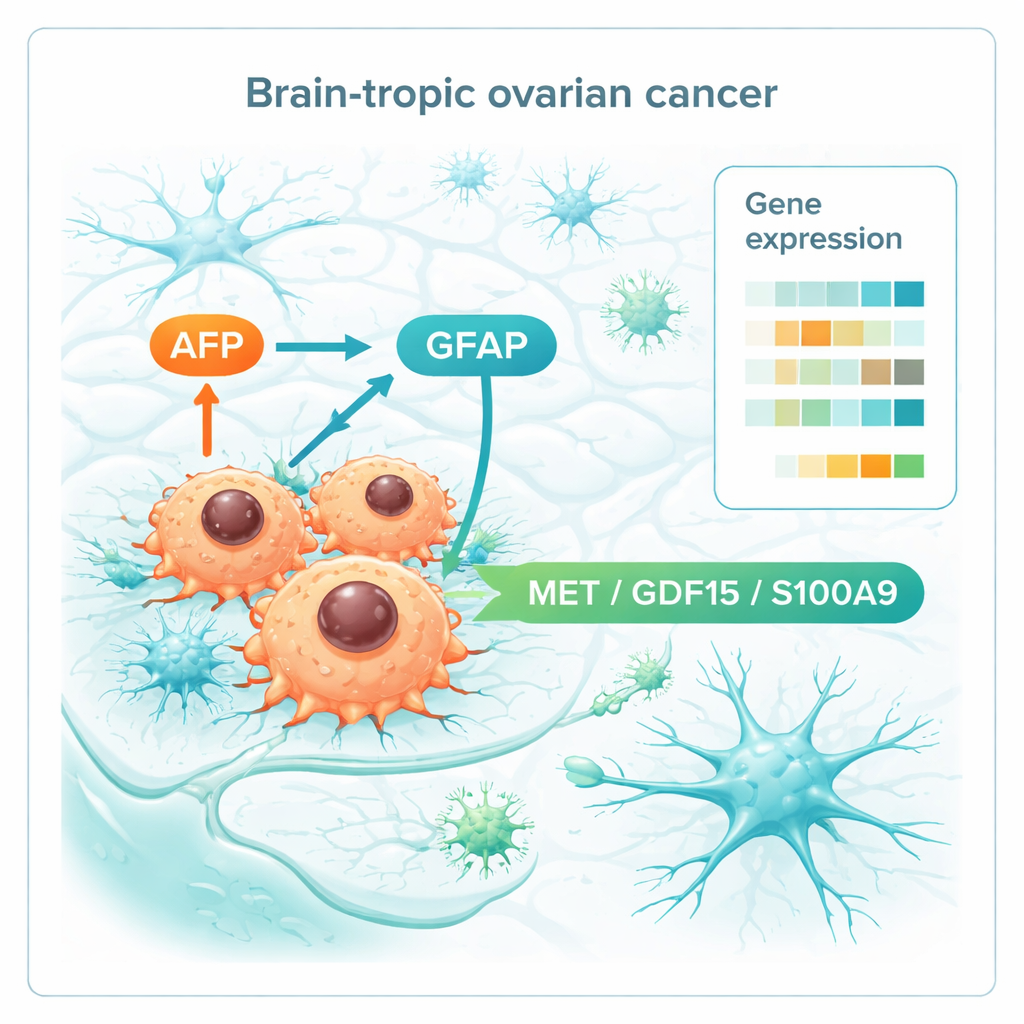
When the team compared gene activity across all the tissues, a distinct brain-metastatic signature emerged. Brain lesions showed signs of aggressive growth, with activation of cell-cycle programs driven by well-known cancer regulators such as MYC and E2F. They also altered the proteins that form and remodel the tissue scaffold, helping cells invade and anchor within brain matter. Strikingly, some genes linked to nerve and glial cells were turned on, hinting that ovarian cancer cells partly mimic brain cells to fit in. Among the most notable were AFP, usually active in the fetus and certain tumors, and GFAP, a hallmark protein of support cells in the brain. The authors also identified a small set of genes already elevated in the primary ovarian tumors of women who later developed brain metastases but not in long-term survivors without brain involvement. This “premetastatic” pattern involved immune modulation, communication with nerve-like signals, and changes in metabolism.
Early warning signals and molecular crosstalk
Digging deeper, the researchers focused on molecules that sit on the cell surface or are secreted, because these are prime candidates for blood tests or drug targets. They pinpointed three players—MET, GDF15, and S100A9—that were consistently higher both in primary tumors destined for the brain and in the brain metastases themselves. These molecules are involved in cell movement, stress and nutrient sensing, and shaping the immune environment, suggesting they help ovarian cancer cells talk to and reshape brain tissue in their favor. Together with AFP and GFAP, they form a short list of potential biomarkers that could one day flag high-risk patients or point to drugs that disrupt tumor–brain communication.
What this means for patients
For now, this work will not immediately change treatment, and the authors acknowledge that their patient group is small. Yet by showing that brain-metastatic ovarian cancers keep much of their original genetic makeup while rewiring how their genes are used, the study highlights new ways to spot trouble early. If future research confirms that markers such as AFP, GFAP, MET, GDF15, and S100A9 reliably signal a tendency to spread to the brain, doctors might monitor certain patients more closely, adjust therapy choices, or test drugs that block these pathways. In essence, the study begins to turn a frightening late complication into a process that can be measured, understood, and, eventually, intercepted.
Citation: Trozzi, R., Salvi, M., Karimi, M. et al. Deciphering brain metastasis in epithelial ovarian cancer: multimodal analysis and potential biomarkers. npj Precis. Onc. 10, 83 (2026). https://doi.org/10.1038/s41698-025-01206-y
Keywords: ovarian cancer, brain metastasis, biomarkers, gene expression, precision oncology