Clear Sky Science · en
Adipocyte-driven STAT3-ANGPT2-PTGIS axis promotes cutaneous metastasis in breast cancer and represents a targetable pathway
Why skin spread of breast cancer matters
When breast cancer spreads, most people think of the bones, liver, or lungs. Yet for many patients the cancer resurfaces in an unexpected place: the skin of the chest wall, sometimes years after the original tumor was removed. These stubborn, painful skin nodules are not just a cosmetic problem—they are a sign of aggressive disease that is hard to treat. This study digs into why some breast cancers are drawn to the skin, revealing a surprising partnership between cancer cells, nearby fat tissue, and the blood and lymph vessels that nourish tumors.
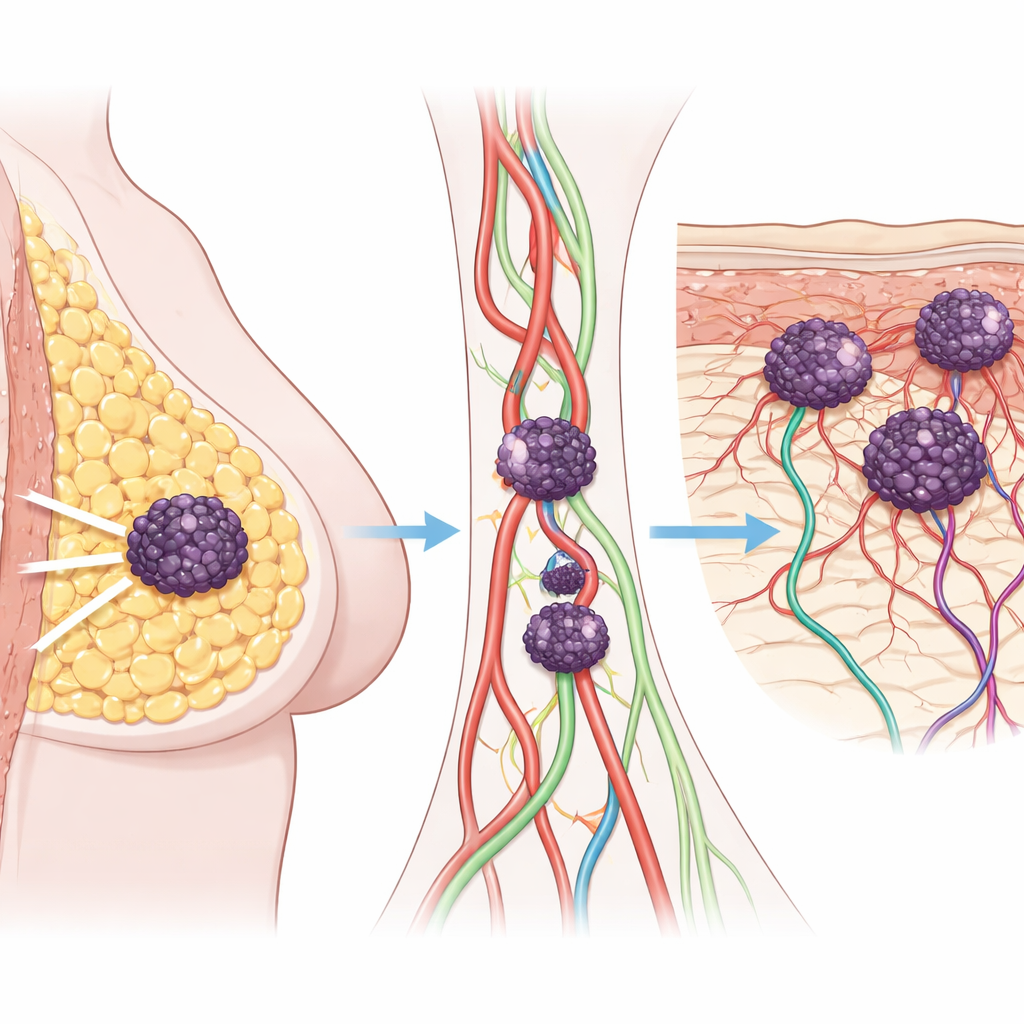
Skin tumors as a warning sign
The researchers first examined medical records from 126 people with breast cancer, comparing those who never relapsed, those whose cancer spread to organs like the lung or bone, and those who developed skin metastases. Patients whose disease came back only in the skin already had more advanced and fast-growing tumors at the time of diagnosis. Their cancers showed high cell-division rates and early invasion into blood and lymph vessels. Even though some of these patients had no visible spread to distant organs, their overall and disease-free survival were worse than patients without skin involvement, highlighting that skin metastasis marks a particularly aggressive form of breast cancer.
Blood vessels and fat shape the skin niche
To understand what makes the skin such fertile ground, the team followed one patient over several years, collecting samples from the original breast tumor and later skin nodules. Under the microscope, both breast and skin lesions were packed with newly formed blood and lymphatic vessels—tiny highways that help cancer cells travel and take root. Gene-activity profiling showed that, as the disease progressed, the tumors switched on whole networks related to blood-vessel growth, inflammation, and the handling of fats and fatty acids. In particular, pathways that break down common dietary fats, such as arachidonic and linoleic acids, became more active, suggesting that fuel from fat and the skin’s own fatty layers help drive tumor evolution.
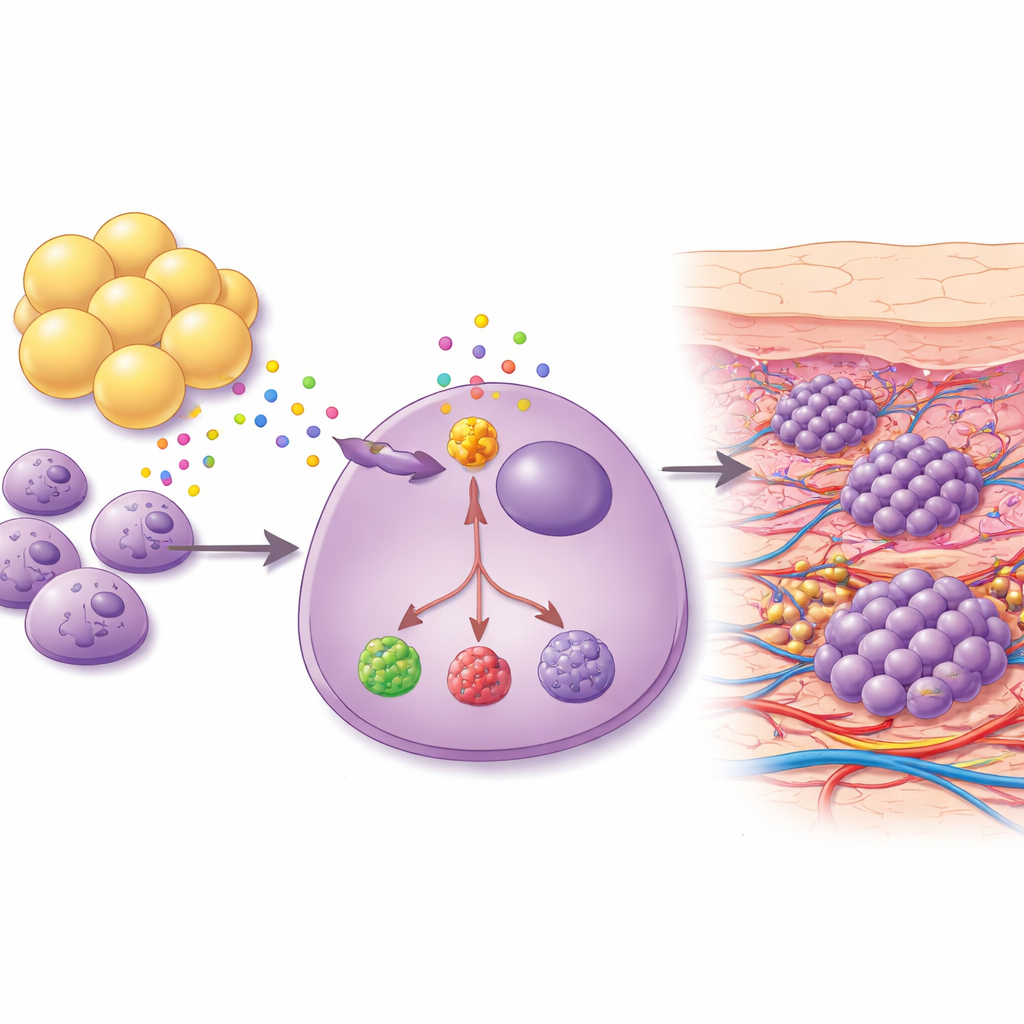
Fat cells send powerful growth signals
Because the breast and underlying chest wall are rich in fat, the scientists asked whether fat cells themselves might coax cancer cells toward the skin. They grew breast cancer cells together with adipocytes—mature fat cells—from the same patient and collected the mixture of molecules the fat cells released. This cocktail strongly boosted the ability of blood-vessel cells to form tube-like structures in the lab, a hallmark of angiogenesis. In mouse models, exposing tumors to fat-cell–derived fluid led to new cancer growth in skin-rich, fat-rich areas such as the neck and back. Cancer cells taken from these skin lesions were more stem-like, more resistant to cell death, and produced higher levels of pro-growth and pro-inflammation factors, including a protein called ANGPT2 and a metabolic enzyme named PTGIS.
A signaling switch that can be turned off
Digging deeper, the team compared ordinary tumor cells with their skin-seeking counterparts and found that a common communication route inside cells—the JAK–STAT pathway—was strongly activated, especially the protein STAT3. When switched on, STAT3 acted like a master control, boosting ANGPT2, VEGFC (a driver of lymph vessel growth), and PTGIS, all of which encourage blood-vessel formation and remodeling of the tumor environment. Blocking STAT3 with a drug called Stattic reduced skin tumor growth in mice and lowered the levels of these downstream factors. In patient tumor samples, high STAT3, ANGPT2, and PTGIS activity was linked to worse progression-free and disease-free survival, and these markers were particularly elevated in patients whose metastases were confined to the skin.
What this means for patients
Taken together, the work paints cutaneous metastatic breast cancer as a distinct and dangerous subtype fueled by a three-way interaction among cancer cells, surrounding fat, and the vessels that feed tumors. Signals from adipocytes activate STAT3 inside cancer cells, which in turn cranks up ANGPT2 and PTGIS and reshapes fatty-acid metabolism, making the skin a hospitable niche where tumors can settle and expand. For patients, this means that skin metastases are not simply “local” recurrences but a marker of aggressive biology. Encouragingly, the same pathway that drives this process may also be its weakness: drugs that target STAT3 or key metabolic enzymes like PTGIS could, in the future, help cut off the support lines that allow breast cancer to colonize the skin.
Citation: Luo, CW., Ou-Yang, F., Chang, SJ. et al. Adipocyte-driven STAT3-ANGPT2-PTGIS axis promotes cutaneous metastasis in breast cancer and represents a targetable pathway. npj Precis. Onc. 10, 111 (2026). https://doi.org/10.1038/s41698-025-01184-1
Keywords: breast cancer skin metastasis, tumor microenvironment, adipocyte signaling, STAT3 pathway, angiogenesis