Clear Sky Science · en
PiggyBac-mediated transgenesis and CRISPR–Cas9 knockout in the greater wax moth, Galleria mellonella
A Tiny Caterpillar with Big Medical Potential
Imagine testing new antibiotics or studying dangerous infections without using mice or other mammals. The larvae of the greater wax moth, Galleria mellonella, are emerging as a low‑cost, ethically attractive stand‑in for such experiments. This study shows how scientists have taken a major step forward by giving these caterpillars the same kind of genetic tools that made fruit flies and zebrafish so powerful for biomedical research.
Why This Insect Matters for Human Health
Galleria mellonella larvae are surprisingly good mimics of how human bodies respond to germs. They can be kept at 37 °C—our body temperature—and can be infected with a wide range of medically important bacteria and fungi. When they become sick, their bodies darken through a process called melanization, giving researchers a visible readout of health. Because they are invertebrates, they avoid many of the legal and ethical hurdles of vertebrate animal work, and they are inexpensive to rear in large numbers. The larvae have even been found to break down common plastics, hinting at uses in environmental research as well.
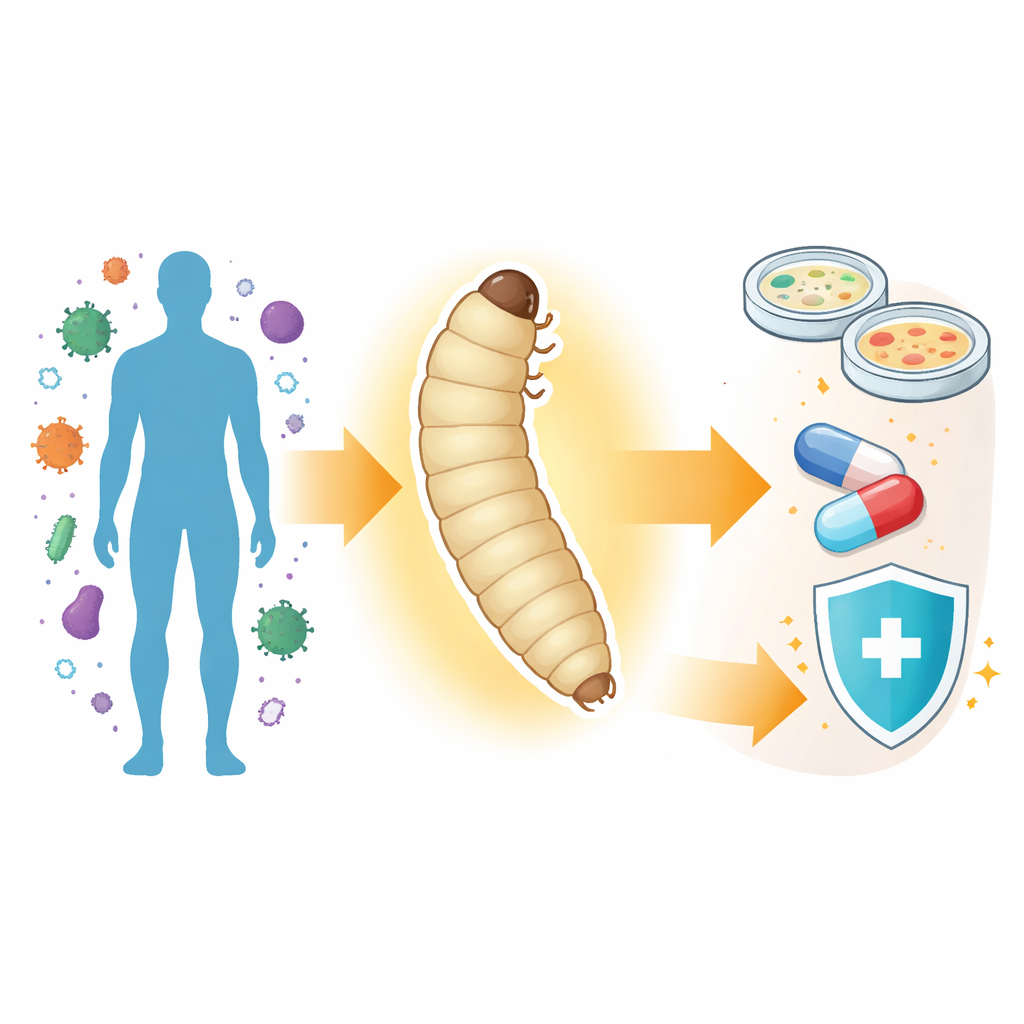
The Missing Genetic Toolkit
Despite these advantages, wax moth larvae have lacked the sophisticated genetic tricks available in classic lab animals. Over the past decade, several teams have decoded the moth’s genome and cataloged its active genes and proteins, but researchers still could not easily switch genes on or off or add useful markers. That gap has limited what could be done with the model—for example, building animals that light up when infected, or deleting specific genes to test how they affect immunity. The current paper tackles this head‑on by adapting two powerful gene‑engineering systems, known as PiggyBac and CRISPR–Cas9, for use in Galleria mellonella.
Building Glow‑in‑the‑Dark Reporter Larvae
The team first studied the timing of early moth embryo development and found a roughly six‑hour window after eggs are laid when all dividing nuclei still share a common interior. Injecting DNA during this period allows new genetic material to be carried into all later tissues, including future sperm and eggs. Using this window, the researchers introduced DNA constructs based on the PiggyBac transposon, a “cut‑and‑paste” element that inserts large pieces of DNA into the genome. After testing different helper plasmids, they identified a version carrying a hyperactive PiggyBac enzyme that successfully produced transgenic lines. These larvae expressed green and red fluorescent proteins in distinct body tissues, and DNA analysis showed that the genetic cassette landed in a harmless stretch between native genes.
Watching Cells and Knocking Out Genes
Next, the authors created more specialized reporter lines. One construct tagged a structural protein called tubulin with green fluorescence to highlight the cell’s internal skeleton, while another fused a red marker to a histone protein that wraps DNA in the nucleus. These lines glowed in characteristic patterns across gut, fat body, silk glands and other tissues, allowing researchers to visualize cell structures in live or fixed larvae. To go beyond adding genes and actually disable them, the team turned to CRISPR–Cas9. They injected pre‑assembled CRISPR protein–RNA complexes targeting the green fluorescent marker into embryos that already carried the reporter cassette. Many offspring showed partial or complete loss of green signal while retaining red eye fluorescence, and DNA sequencing confirmed small insertions and deletions at the intended CRISPR cut site.
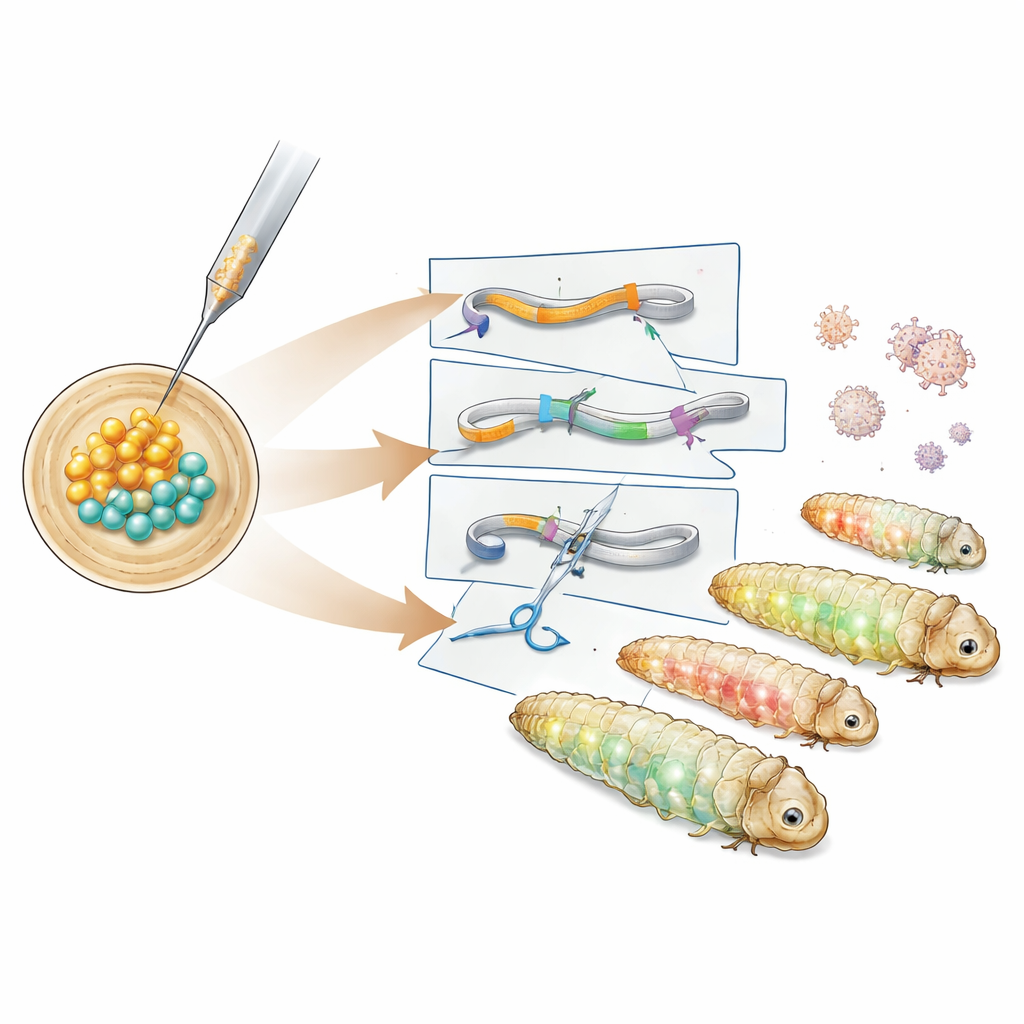
From Technical Advance to Practical Impact
Although the efficiency of both PiggyBac and CRISPR in these moths is still modest compared with some other insects, the authors outline clear paths for improvement by tuning promoters, enzyme variants and injection strategies. Even in its current form, the toolkit is powerful enough to generate stable lines that visualize key cell components or carry targeted gene knockouts. For non‑specialists, the big picture is that Galleria mellonella can now be engineered almost as flexibly as better‑known lab animals. That means researchers can build caterpillars that report on infection, model aspects of human disease, or test new drugs in ways that are quicker, cheaper and more humane, potentially reducing the reliance on mammalian models in infection biology and related fields.
Citation: Pearce, J.C., Campbell, J.S., Prior, J.L. et al. PiggyBac-mediated transgenesis and CRISPR–Cas9 knockout in the greater wax moth, Galleria mellonella. Lab Anim 55, 95–102 (2026). https://doi.org/10.1038/s41684-025-01665-7
Keywords: Galleria mellonella, invertebrate infection model, genetic engineering, PiggyBac transgenesis, CRISPR knockouts