Clear Sky Science · en
Enhanced NO2-driven multiphase formation of particulate nitrate and sulfate under high-humidity conditions
Why humid air can still be dirty
Many people imagine that damp, foggy weather helps clean the air, washing pollution out of the sky. But in southern China, the opposite often happens: when warm, wet ocean air meets cooler inland air, thick haze builds up even though traditional atmospheric cleaners are weak. This study explains why, revealing how a common traffic pollutant, nitrogen dioxide, quietly fuels the rapid formation of harmful fine particles when the air is very humid.
Sticky air over a coastal city
The researchers focused on Xiamen, a coastal city in southern China, during two weeks in early spring 2024. At this time of year, warm, moist ocean air frequently collides with cooler, drier continental air and stalls, creating quasi-stationary weather fronts. During these high-humidity events, the air becomes stagnant, wind speeds drop, and a shallow blanket of air sits over the city. Measurements from a rooftop monitoring station showed that fine particle levels (PM2.5) rose, visibility worsened, and fog often formed as humidity climbed. Within these hazy periods, the chemical make-up of particles changed in two clear stages: first nitrate dominated, then sulfate surged.
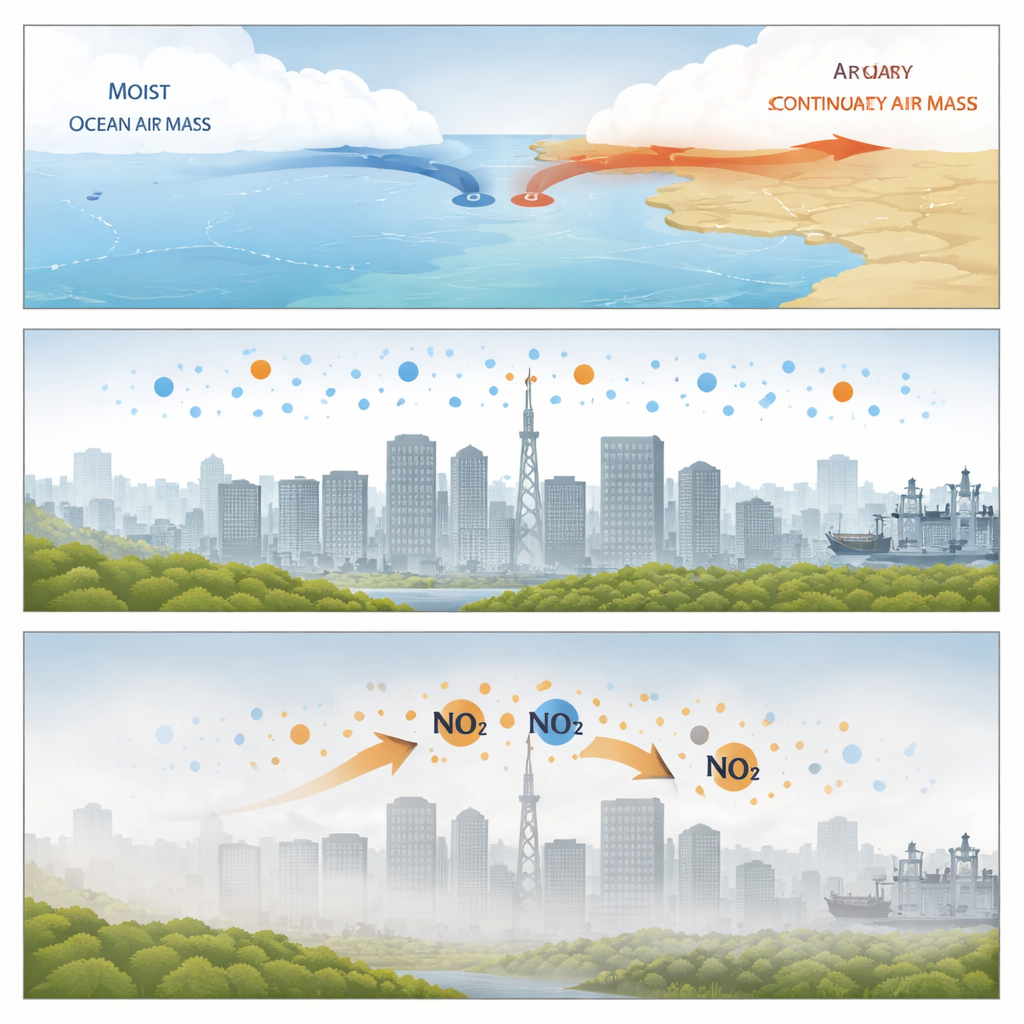
Two phases of hidden chemistry
In the early, very humid hours of these events, particles became rich in nitrate, even though the usual sunlight-driven chemistry was weak and ozone levels were low. The team found that the best explanation was that nitrogen dioxide from traffic and other sources was being taken up directly by wet particle surfaces and thin films of water on the ground. In these concentrated droplets, nitrogen dioxide reacts more quickly than in ordinary water, producing both nitrate and another reactive nitrogen form that can escape back into the air. The close link between nitrogen dioxide, aerosol water, particle surface area, and rising nitrate suggested that these surface processes dominated nitrate production at night.
When fog turns nitrogen into sulfur particles
As humidity climbed further and fog formed, the chemistry shifted. Sulfate, formed from sulfur dioxide, began to grow rapidly and could even overtake nitrate. Here again nitrogen dioxide played a central role, but in a different way. Inside fog and large droplets, nitrogen dioxide and its reaction products oxidized dissolved sulfur in a series of water-based reactions. One key intermediate, a reactive nitrogen species that shares a balance with nitrous acid, was found to stay longer inside bigger, less acidic droplets than in tiny, more acidic particles. This longer residence time allowed it to repeatedly attack dissolved sulfur, driving fast sulfate production precisely when dense fog and haze were present.
Putting numbers on the invisible
To test these ideas, the authors built a detailed computer “box model” that combined gas-phase chemistry, reactions in liquid water, and exchanges between air and particles. When they allowed nitrogen dioxide to be taken up by wet particles and fog droplets, the model closely reproduced the observed rises in nitrate and sulfate. During humid events, direct nitrogen dioxide uptake accounted for nearly half of all nitrate formed, and nitrogen-based oxidants (nitrogen dioxide plus its aqueous products) produced almost two-thirds of the sulfate. At night, the nitrogen-dioxide pathway far outpaced the more familiar route involving another nighttime oxidant, dinitrogen pentoxide. The model also showed that fog droplets, with their larger size and higher pH, are especially favorable reactors for the nitrogen–sulfur chemistry that makes sulfate.
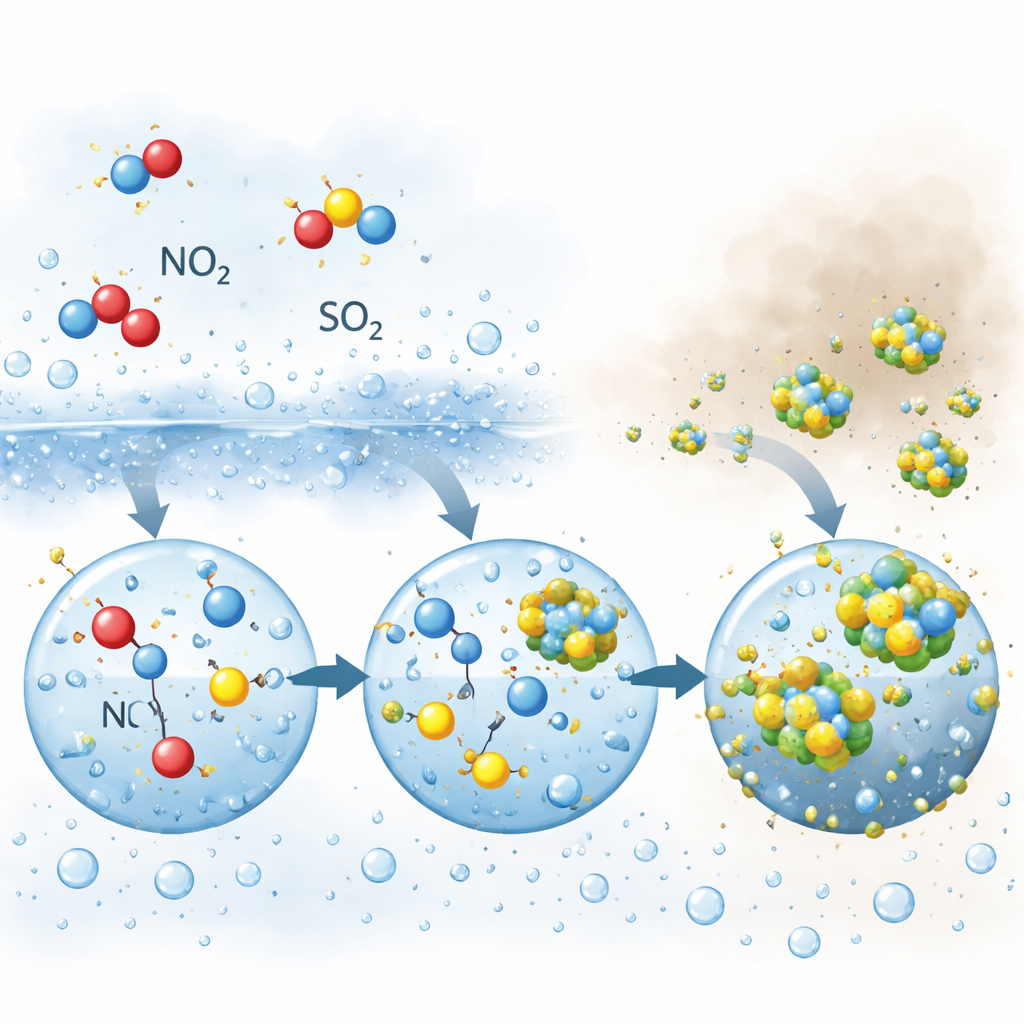
What this means for cleaner air
The study concludes that nitrogen dioxide does much more than serve as a warning gas for traffic pollution: under very humid, stagnant conditions it becomes a powerful driver of fine particle formation. By speeding up the conversion of gaseous nitrogen and sulfur into nitrate and sulfate particles, these hidden water-based reactions help explain why particle levels have not fallen as quickly as emissions of nitrogen and sulfur gases in China. The findings suggest that effective smog control in coastal and humid regions must consider not just how much nitrogen dioxide is emitted, but also how often the atmosphere enters these sticky, foggy states that turn it into a microscopic chemical factory.
Citation: Lin, Z., Ji, X., Xu, L. et al. Enhanced NO2-driven multiphase formation of particulate nitrate and sulfate under high-humidity conditions. npj Clim Atmos Sci 9, 76 (2026). https://doi.org/10.1038/s41612-026-01352-5
Keywords: air pollution, nitrogen dioxide, fine particles, high humidity, fog chemistry