Clear Sky Science · en
Targeting cancer expressed EGFR with a humanized monoclonal antibody
Finding Cancer’s Hidden Flags
Cancer treatments often struggle to strike a balance between attacking tumor cells and sparing healthy tissue. This paper describes a new laboratory‑engineered antibody, called A10, that can distinguish cancerous cells carrying a particular form of a growth signal receptor from normal cells. By homing in on a shape that appears mainly when this receptor is altered or overcrowded on tumor cells, A10 opens the door to therapies that are both powerful and more selective than many current options.
Why This Growth Switch Matters
Many tumors, including aggressive brain, lung, breast, and colorectal cancers, depend on a surface protein called the epidermal growth factor receptor (EGFR) to grow and survive. Tumor cells can boost this receptor by either amplifying its gene, mutating it, or producing a shortened, always‑on version known as EGFRvIII. Standard drugs that block EGFR often hit both cancer cells and normal tissues, because they recognize parts of the receptor that are present everywhere. The team behind this study instead focused on a small loop of EGFR that becomes clearly exposed mainly on cancer cells—either because EGFR is present in abnormally high numbers or because the receptor is truncated in EGFRvIII. That loop acts like a cancer‑specific “flag” that can, in principle, be targeted much more safely.
Turning a Mouse Antibody into a Human Medicine
The researchers had previously discovered a mouse antibody, 40H3, that recognized this exposed loop on cancer‑expressed EGFR. Mouse antibodies, however, can trigger immune reactions if given repeatedly to patients. To overcome this, the team used protein‑engineering methods to graft the key binding segments of 40H3 onto human antibody frameworks, generating fourteen “humanized” candidates plus a chimeric reference molecule. When they tested these variants on cancer cells that overproduce EGFR, three versions bound especially well, and one—A10—stood out as the strongest binder while still ignoring normal cells with modest EGFR levels. Across a wide panel of tumor cell lines with different EGFR mutations and copy numbers, A10 consistently showed higher binding than the original mouse antibody and maintained a strong preference for malignant cells.
Seeing How the Antibody Grips Its Target
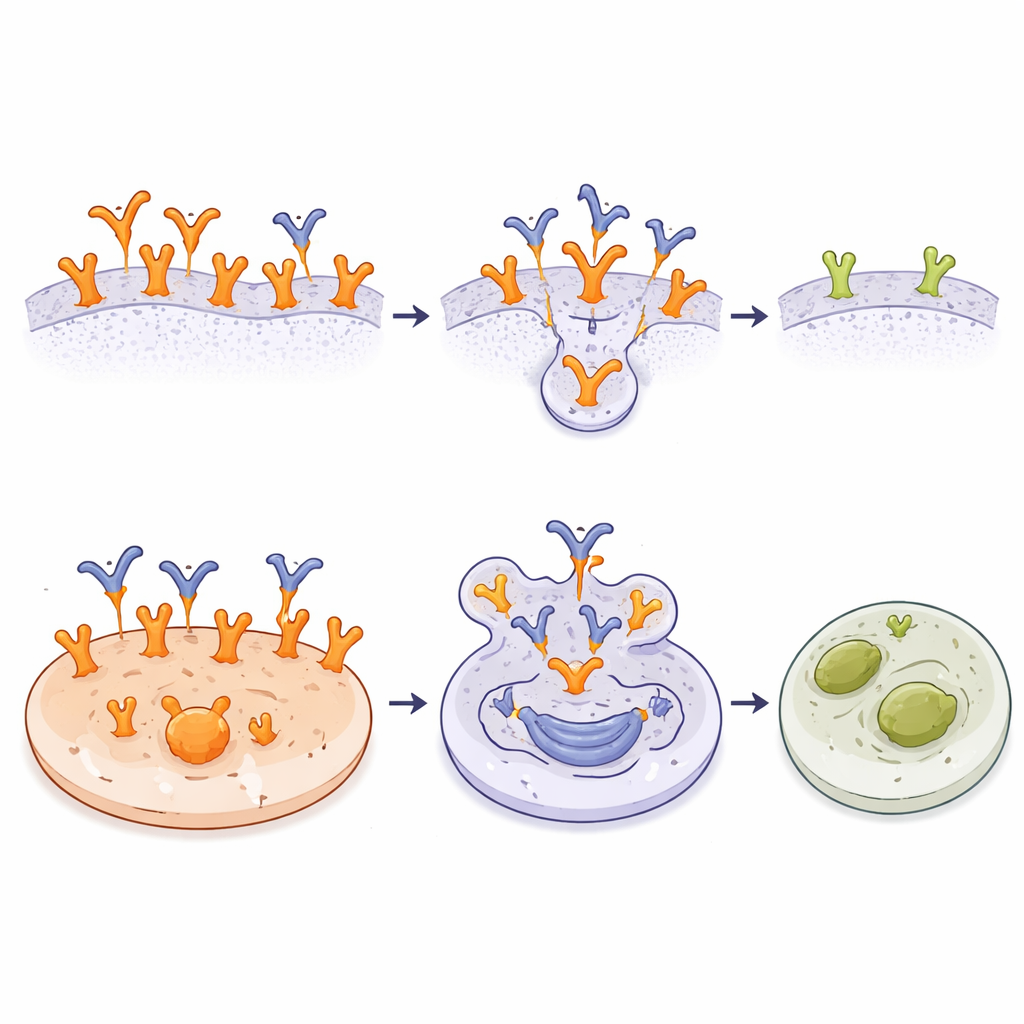
To understand why A10 is so selective, the team solved a high‑resolution 3D structure of the active part of the antibody bound to the EGFR loop. The loop folds into a tight hairpin stabilized by a disulfide bond, and A10 cradles this shape in a groove formed between its two arms, with many charged contacts locking it in place. Comparing this structure with known shapes of the full EGFR molecule revealed a key insight: in both the inactive “tethered” and active dimer forms of normal EGFR, the loop is largely buried or partially shielded by neighboring domains. This means A10 cannot easily reach its binding site on typical, modestly expressed receptors. In contrast, in the cancer‑specific EGFRvIII variant, the front part of the receptor is missing, leaving the loop highly exposed and accessible. The data suggest that A10 mainly recognizes EGFR during certain transitional shapes and in overcrowded or truncated forms that occur preferentially on tumor cells.
Arming the Antibody to Kill Tumor Cells
Recognizing a tumor‑specific flag is useful on its own, but the researchers went further by turning A10 into an antibody‑drug conjugate (ADC). They chemically attached a potent cell‑killing compound, monomethyl auristatin E (MMAE), to A10 using a linker that can be cut inside cells. This A10‑MMAE ADC bound cancer cells just as well as the unconjugated antibody. In lab tests across many cell lines, the ADC was most effective against tumors with very high EGFR levels or EGFRvIII, where A10 had abundant binding sites. In these cells, tiny amounts of the conjugate were enough to trigger cell death. Tumor lines with fewer A10 targets or inherent resistance to MMAE were less affected, and normal fibroblast cells with wild‑type EGFR remained unharmed despite being sensitive to free MMAE. The results show that the combination of selective binding and internal drug release can sharply focus toxicity on cancer cells.
What This Could Mean for Future Cancer Care
In simple terms, this work shows that it is possible to build an antibody that “sees” EGFR only when it looks and behaves like a cancer‑driving receptor, while mostly ignoring its everyday form on healthy tissues. By mapping exactly how A10 grips a hidden loop on EGFR and demonstrating that an A10‑based drug conjugate can efficiently kill receptor‑rich tumor cells yet spare normal cells, the study provides a strong foundation for developing safer, highly targeted therapies. If these findings translate to patients, future treatments based on A10 could deliver powerful drugs directly to EGFR‑dependent cancers while leaving the rest of the body largely untouched.
Citation: Costa, T.G.F., Sarnovsky, R., Zhan, J. et al. Targeting cancer expressed EGFR with a humanized monoclonal antibody. Sci Rep 16, 10814 (2026). https://doi.org/10.1038/s41598-026-46245-y
Keywords: EGFR, antibody drug conjugate, targeted cancer therapy, glioblastoma, EGFRvIII