Clear Sky Science · en
Structural and mechanistic insights into α2β1 and α5β1 integrin targeting by bioengineered extracellular vesicles originating from lung cancer cells
Cancer’s Hidden Messengers
Lung cancer is often discovered late, when treatment options are limited. One promising way to fight it is to deliver drugs only to cancer cells, sparing healthy tissue. This study explores a tiny natural delivery system—extracellular vesicles, or EVs—that cancer cells already use to send messages. By reengineering these vesicles with a short targeting tag, the researchers show how they can latch onto specific docking sites on tumor cells, laying groundwork for more precise and less toxic cancer therapies.
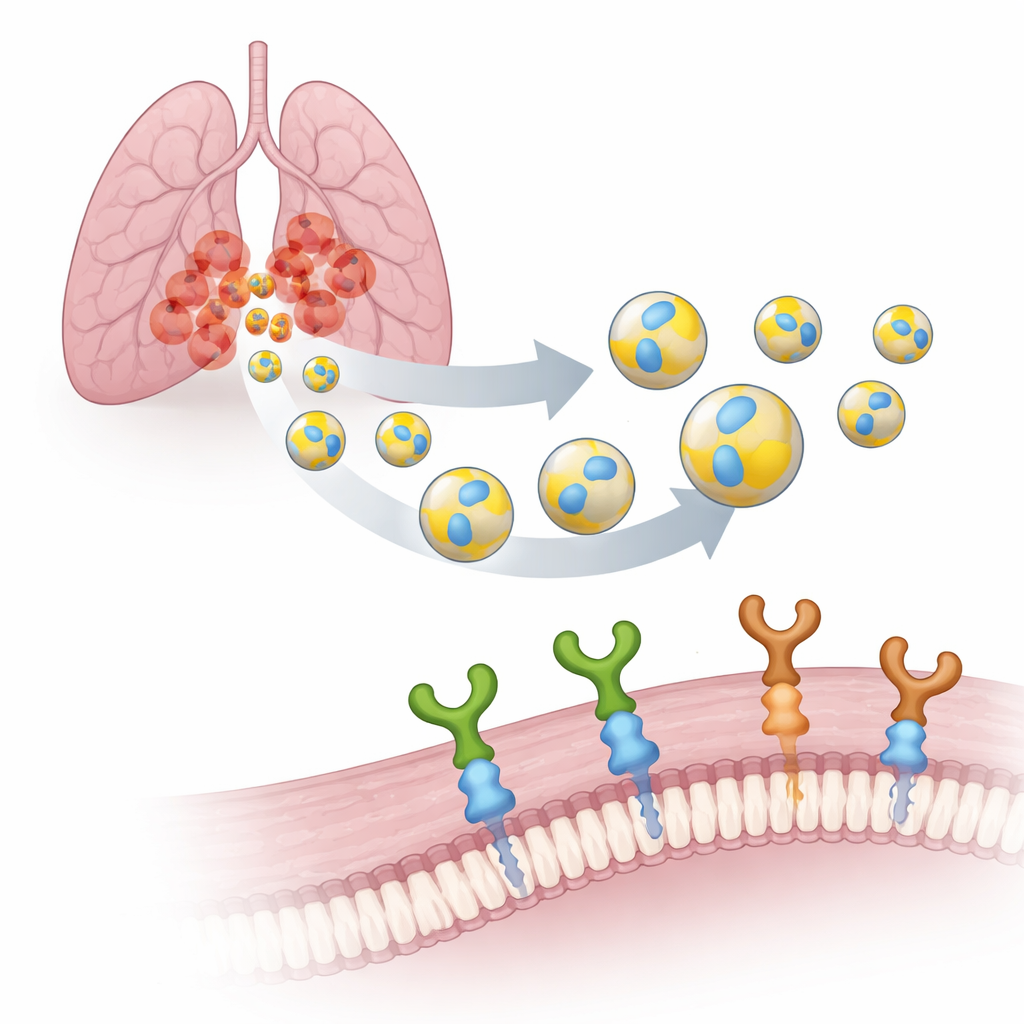
Turning Natural Packages into Smart Couriers
Every cell in our body releases EVs: nanoscale bubbles surrounded by a membrane, a bit like microscopic soap bubbles. Because they are made by our own cells, EVs are biodegradable, slip easily through biological barriers, and are less likely to provoke side effects than synthetic carriers. The team focused on EVs produced by human lung cancer (A549) cells and decorated their surface with a small seven–building block tag called PTHTRWA. Earlier experiments had shown that this tag drives EVs to home in on lung cancer cells in mice, binding much more strongly to tumor cells than to normal lung cells while remaining only weakly toxic.
Finding the Right Docking Ports on Tumor Cells
To turn these tagged vesicles into truly targeted tools, the researchers needed to identify which “ports” on cancer cells they recognize. They looked at two related surface receptors, α2β1 and α5β1 integrins. These are molecular grappling hooks that help cells stick to their surroundings and send signals inward. In lung tumors, α5β1 is strongly linked to aggressive behavior, spread, and poor survival, while α2β1 has been connected to resistance to chemotherapy. Using a sensitive technique called surface plasmon resonance, the team compared how strongly PTHTRWA-decorated EVs bind to both integrins. They found that vesicles without the tag barely interacted, whereas tagged EVs formed tight, stable complexes with both receptors under near-physiological conditions.
Zooming In on the Molecular Embrace
To understand what happens atom by atom when the tag meets the integrins, the researchers turned to computer simulations. They built three-dimensional models of the tag bound to key regions of the integrins and let these systems evolve in virtual water for hundreds of nanoseconds. The calculations showed that PTHTRWA fits snugly into a shallow groove on the α2β1 receptor, forming a network of hydrogen bonds and hydrophobic contacts that stabilizes the complex. Crucially, binding favored a more “open” shape of the receptor—its high-affinity, signal-ready form—over the “closed” resting state. When they tested slightly modified versions of the tag, they saw that small chemical tweaks at one end can subtly reshape how the peptide lies in the groove and how strongly it prefers one integrin over the other.
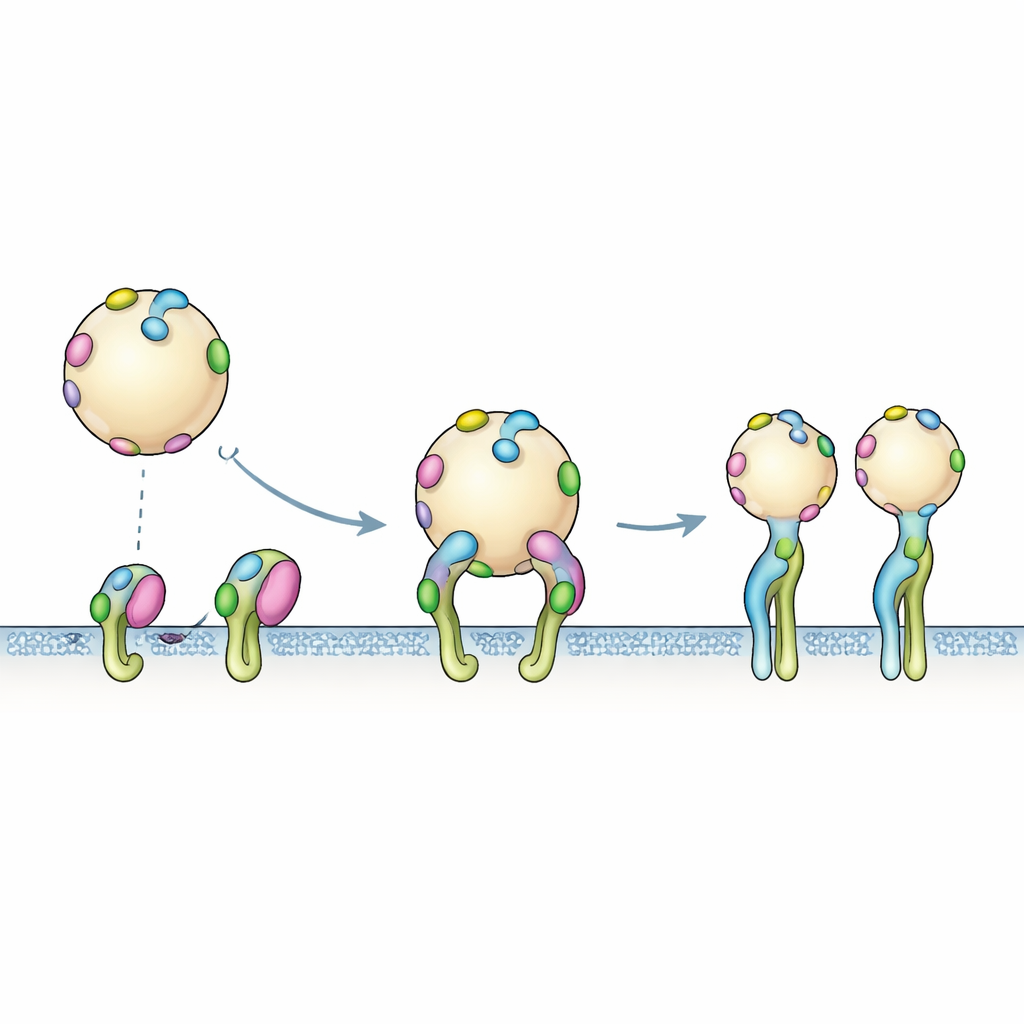
From Single Tags to Multivalent Grips
Computer models capture one tag touching one receptor at a time, but the real EV surface bristles with many copies of PTHTRWA. The binding experiments reflected this multivalent reality: once a tagged vesicle approaches a patch of integrins, multiple tags can engage at once. The sensor data fit best to a two-step process. First, the vesicle briefly docks through fast, reversible encounters. Then, a slower rearrangement occurs in which the integrins shift into their open shape and the overall grip tightens. This cooperative, many-handed clasp explains why the apparent binding strength of whole vesicles is higher than what would be predicted from a single tag alone.
What This Could Mean for Future Treatments
Put simply, the study shows that EVs armed with the PTHTRWA tag can selectively recognize integrins that are overactive in lung cancer, and that they do so by nudging these receptors into an open, tightly bound form. By mapping these interactions in detail, the researchers provide design rules for next-generation vesicle-based carriers that could deliver drugs, imaging agents, or other payloads directly to malignant cells while largely ignoring healthy tissue. Although more work in animals and humans will be needed, this integrin-targeting strategy brings the vision of personalized, EV-based cancer medicine a step closer to reality.
Citation: Nowicka, A.M., Żołek, T., Kowalczyk, A. et al. Structural and mechanistic insights into α2β1 and α5β1 integrin targeting by bioengineered extracellular vesicles originating from lung cancer cells. Sci Rep 16, 10833 (2026). https://doi.org/10.1038/s41598-026-46071-2
Keywords: lung cancer, extracellular vesicles, integrins, targeted drug delivery, molecular simulations