Clear Sky Science · en
M2 macrophages predict response to neoadjuvant chemotherapy in triple negative breast cancer patients
Why This Matters for Patients
For many women with an aggressive form of breast cancer called triple negative breast cancer, chemotherapy given before surgery is a major hope for shrinking or even eliminating the tumor. Yet some patients respond dramatically while others see little benefit. This study asks a simple but crucial question: can the immune cells already living around a tumor help doctors predict who will respond well to this pre-surgery treatment?
The Body’s First Responders Around the Tumor
Breast tumors do not grow in isolation; they are surrounded by blood vessels, support tissue, and many immune cells. Among these immune cells are macrophages, often described as the body’s “clean-up crew.” They can take on different personalities. One type, sometimes called M1, tends to attack threats and stir up inflammation. Another type, called M2, is more nurturing and calming in normal tissues, but inside tumors this nurturing behavior can be hijacked to help cancer grow and hide from the immune system. Earlier work suggested that when M2 cells are abundant in triple negative breast cancers, patients tend to do worse overall.
Looking Inside Tumors After Treatment
The researchers studied tissue from 42 Chinese women with stage II or III triple negative breast cancer who all received the same standard chemotherapy combination before surgery. After treatment and surgery, pathologists checked whether any invasive cancer remained. If no invasive tumor was detected in the breast or lymph nodes, this was called a pathological complete response, or pCR, which is usually a good sign for long-term outcome. Using staining methods on the removed tumors, the team measured three key features in both the tumor nests and the surrounding supporting tissue: M2-like macrophages marked by a molecule called CD163, cancer-fighting T cells marked by CD8, and a checkpoint protein called PD-L1 on tumor and immune cells.
Good and Bad Immune Neighborhoods
More than half of the patients achieved a complete response. When the researchers compared those who did and did not reach this favorable outcome, clear patterns emerged. Patients whose tumors disappeared tended to be younger and had higher levels of CD8 T cells and PD-L1 in and around the tumor. Most strikingly, they had fewer CD163-marked M2 macrophages, especially in the tissue just outside the tumor clusters. In simple terms, tumors surrounded by more active, attacking immune cells and fewer “tumor-helping” macrophages were more likely to melt away under chemotherapy, while tumors wrapped in many M2 macrophages were more resistant.
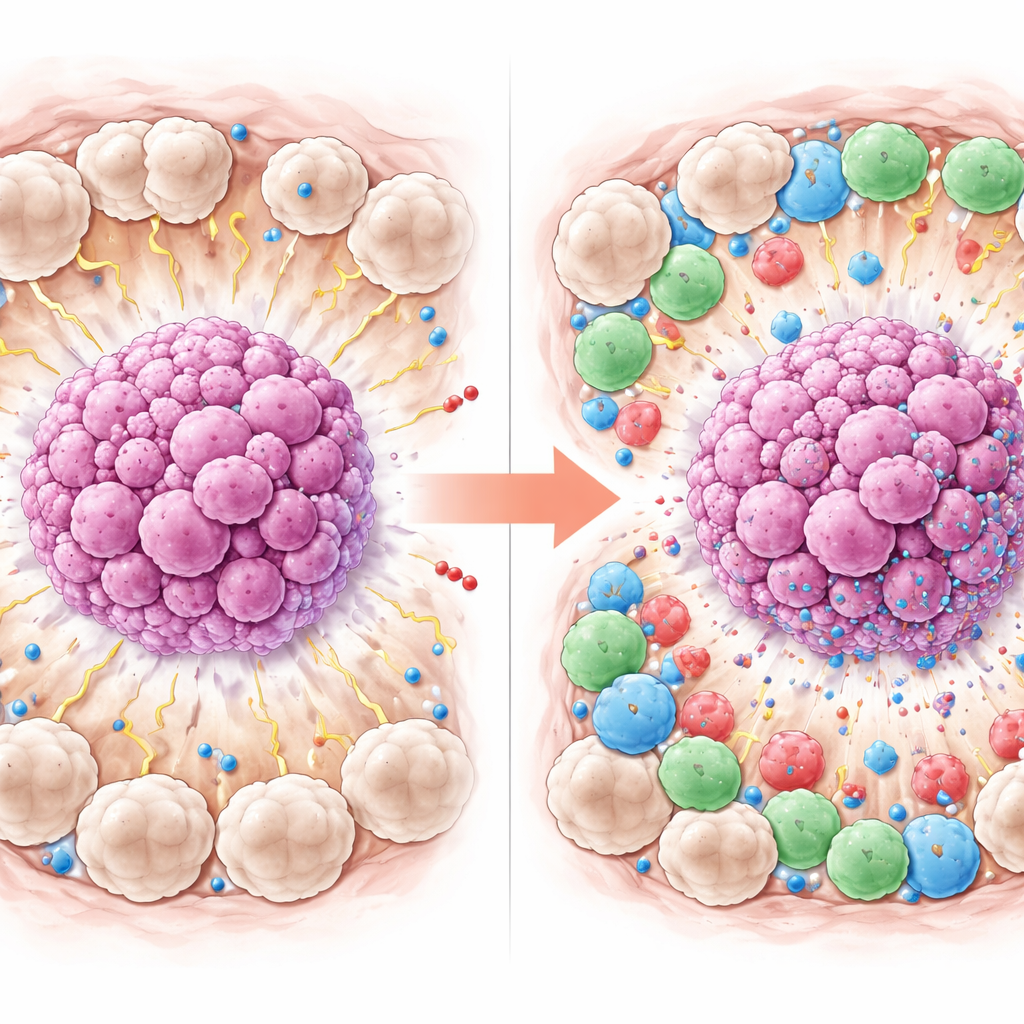
Pinpointing the Strongest Signal
To see which factors truly stood out when considered together, the team used statistical models that account for several variables at once. In this combined analysis, two features of the tumor’s neighborhood were most strongly linked to complete response: higher PD-L1 on surrounding cells and lower numbers of M2-type macrophages in the same area. Interestingly, although more CD8 T cells looked helpful when each factor was examined alone, they did not remain a strong independent predictor once the other features were included, likely because the group of 42 patients was relatively small. The work supports the idea that the mix and behavior of immune cells around the tumor can shape how well chemotherapy works.
What This Could Mean for Future Care
For patients and clinicians, the takeaway is that the immune “climate” around a triple negative breast tumor may help forecast whether standard pre-surgery chemotherapy will be highly effective. In this study, a heavy presence of M2-like macrophages marked by CD163 signaled a lower chance that chemotherapy alone would wipe out the tumor, while fewer of these cells and more active immune signals were linked to better responses. If confirmed in larger and more diverse groups, counting these cells could become part of routine testing to guide treatment choices—helping identify who might benefit from extra or different therapies, such as drugs that target macrophages or boost immune attack, and bringing more precision to the care of this hard-to-treat cancer.
Citation: Chen, Wx., Yang, M., Shao, Qf. et al. M2 macrophages predict response to neoadjuvant chemotherapy in triple negative breast cancer patients. Sci Rep 16, 10638 (2026). https://doi.org/10.1038/s41598-026-46026-7
Keywords: triple negative breast cancer, macrophages, tumor microenvironment, neoadjuvant chemotherapy, immune biomarkers