Clear Sky Science · en
Mechanical tunability of oriented and random electrospun poly(ε-caprolactone) scaffolds via concentration, molecular weight, and environment
Building Better Supports for Healing Tissues
When doctors try to repair a damaged heart, muscle, or bone, they often rely on tiny fiber “scaffolds” that give cells something to cling to as new tissue grows. For these supports to work well, they must be not too soft and not too stiff, and they must survive in the body long enough to do their job. This study shows how to dial in the strength and stretchiness of a popular medical plastic, poly(ε‑caprolactone) or PCL, by carefully controlling how its fibers are spun, blended, and exposed to different environments.
Why Fiber Arrangement Matters
Using a technique called electrospinning, the researchers created sheets of hair‑thin PCL fibers in two main styles: neatly aligned in one direction, and randomly tangled. They then pulled on bundles of these fibers to see how they behaved under stress. The difference was striking. Aligned fibers were much stiffer and stronger, with resistance to stretching similar to tougher soft tissues, whereas random fibers were far more stretchy but much softer. In other words, lining the fibers up turned the scaffold into a load‑bearing material, while a jumbled web produced a flexible, elastic mat. This makes fiber orientation a powerful design knob for matching scaffolds to specific tissues that may need either strength, stretch, or a balance of both.
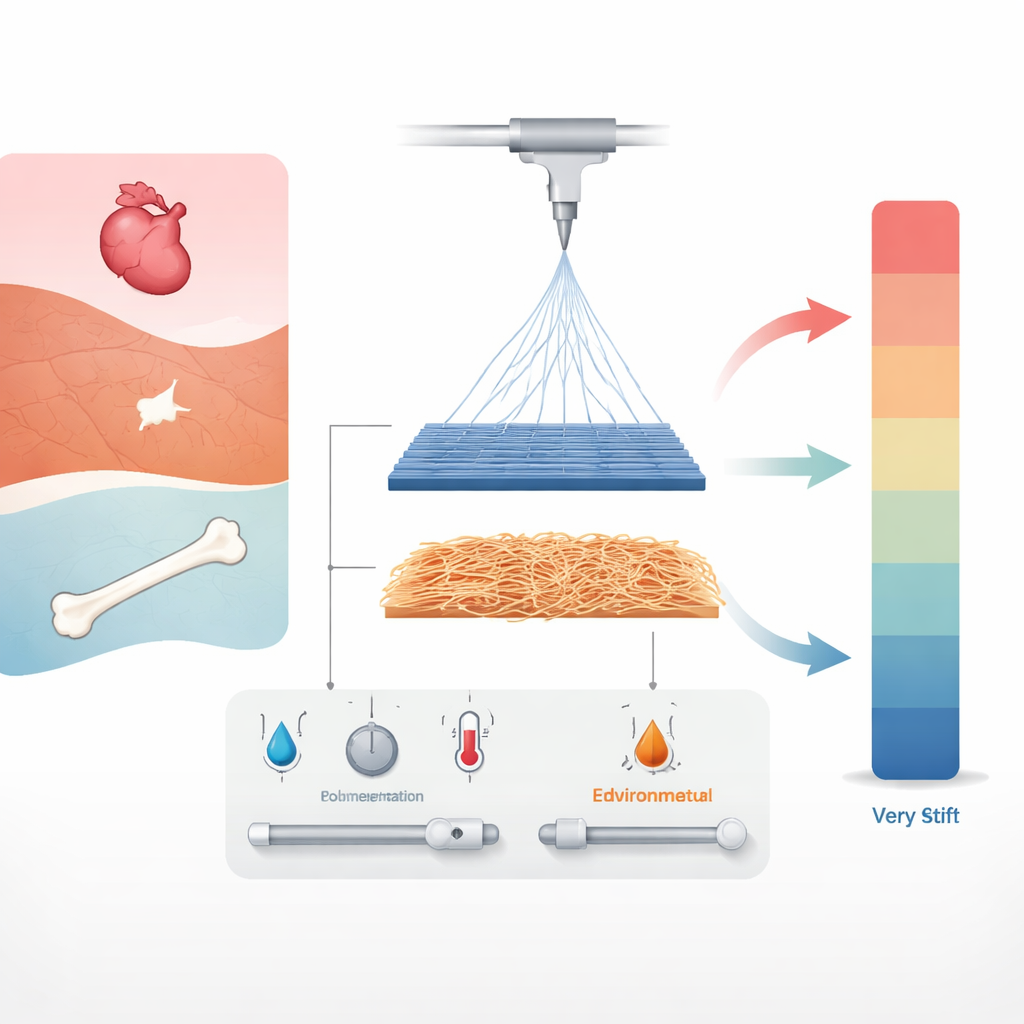
Tuning Fibers by Recipe and Thickness
The team also explored how the “recipe” of the spinning solution affects fiber structure. By increasing the amount of PCL dissolved in the solvent, they could shift from very thin to thicker fibers. For aligned fibers, the best stiffness appeared at intermediate solution levels that produced relatively thin strands. Pushing the concentration higher led to thicker fibers and a drop in stiffness. Random mats, which tended to have much larger fibers overall, needed higher concentrations to gain even moderate firmness, and still remained much softer than their aligned counterparts. These findings show that fiber diameter and alignment work together: thin, well‑ordered fibers carry load efficiently, while thick, disordered ones trade strength for stretch.
Blending Long and Short Chains
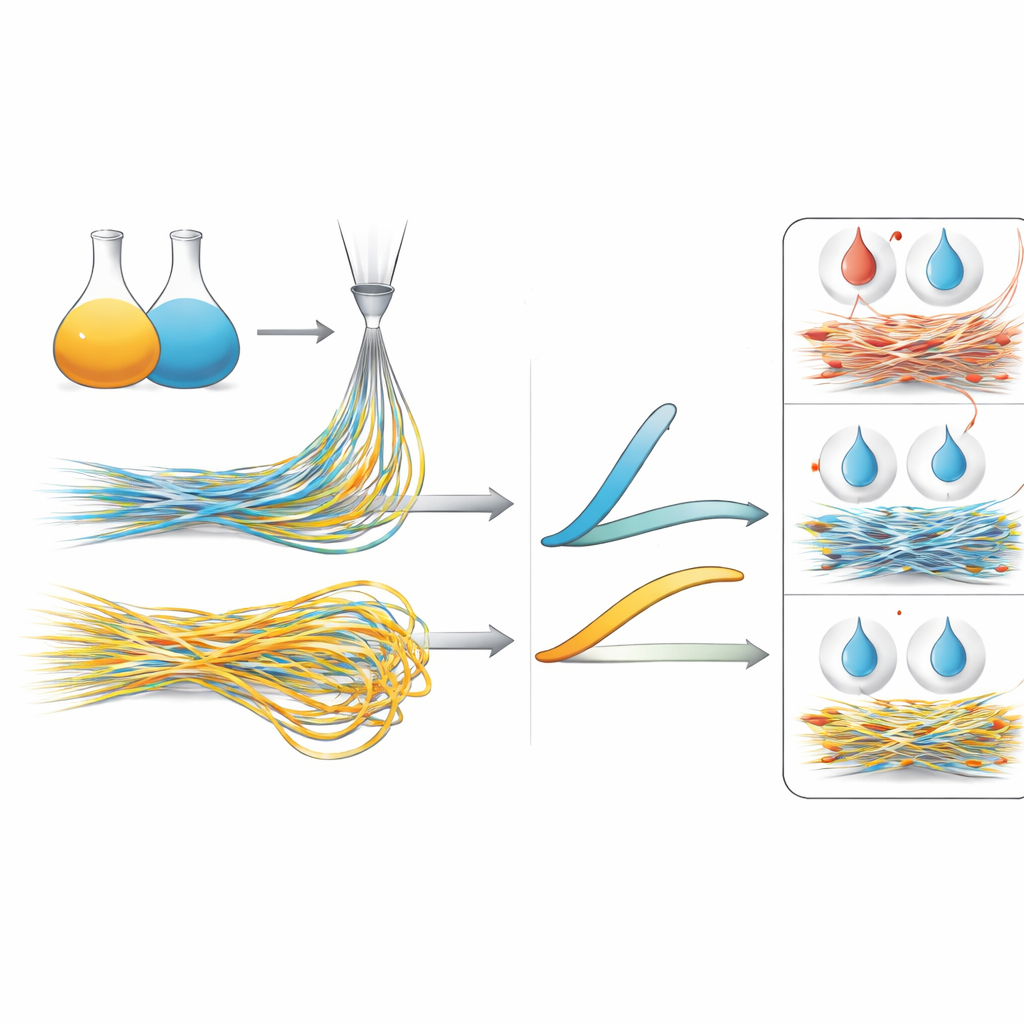
PCL is sold in versions made of long chains (high molecular weight) and shorter chains (low molecular weight). Long chains help form continuous, robust fibers but can be harder to process; short chains spin more easily but on their own form weak, unstable jets. The researchers blended the two types and discovered that mixing them offered extra control over mechanical behavior. For aligned fibers, the highest stiffness did not come from pure long‑chain PCL but from blends with roughly half long and half short chains, which produced thinner, better‑organized fibers. Random fibers, in contrast, needed a high share of long‑chain material to reach their modest stiffness, while still remaining highly stretchable. This shows that simply changing how long the polymer chains are, and how they are mixed, can fine‑tune how the scaffold responds to pulling and bending.
How Harsh and Gentle Environments Shape the Fibers
Because real implants encounter body fluids and sometimes harsh local chemistry, the team soaked the fibers in different liquids and tracked how their strength changed. In mildly acidic solutions based on acetic or formic acid, the fibers gradually softened as the acid concentration and temperature rose. At high acid levels, the fibers shrank dramatically or even dissolved, revealing how sensitive PCL can be to aggressive conditions. In a salt solution that mimics the body’s natural fluid, however, the fibers held up far better. Over one week at body temperature, scaffolds made from only long‑chain PCL lost little stiffness, while blends richer in shorter chains softened more noticeably. This suggests that both chain length and fiber alignment help the fibers resist slow breakdown in realistic, watery surroundings.
A Mechanical Menu for Future Implants
Taken together, the experiments map out a wide “mechanical window” for electrospun PCL, from very soft and stretchy to relatively stiff and strong. By choosing how aligned the fibers are, how concentrated the spinning solution is, how long the polymer chains are, and what environment the scaffold will face, designers can now select scaffolds that match the needs of different tissues—from beating heart muscle to supporting bone interfaces. For patients, this kind of tunable material could mean implants that feel and function more like the body’s own tissue, improving healing and reducing the need for repeat surgeries.
Citation: Munawar, M.A., Schubert, D.W. & Nilsson, F. Mechanical tunability of oriented and random electrospun poly(ε-caprolactone) scaffolds via concentration, molecular weight, and environment. Sci Rep 16, 10507 (2026). https://doi.org/10.1038/s41598-026-45961-9
Keywords: electrospun fibers, polycaprolactone scaffolds, tissue engineering, mechanical tunability, biodegradable polymers