Clear Sky Science · en
Isotopically enriched 64ZN-aspartate attenuates systemic inflammation and gut dysbiosis in an LPS-induced rat model of Parkinson’s disease
Why this study matters for everyday health
Parkinson’s disease is best known for tremors and stiffness, but behind those symptoms lies a storm of whole‑body inflammation and disturbed gut bacteria. This study explores an experimental zinc-based treatment in rats that calms that storm. By dialing down inflammation in the blood and restoring a healthier mix of gut microbes, the approach hints at future therapies that might slow Parkinson’s rather than just mask its symptoms.
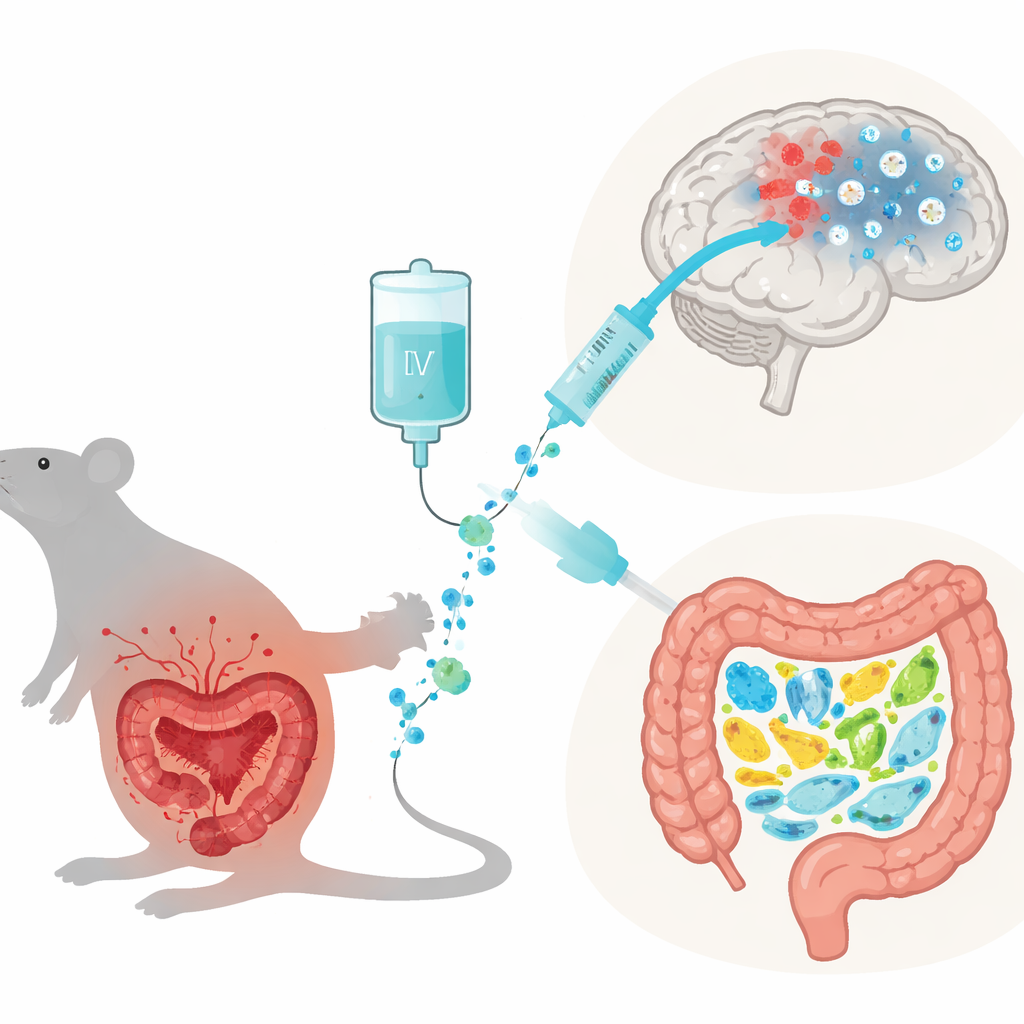
A fresh angle on Parkinson’s disease
For many years, Parkinson’s was viewed mostly as a problem of brain cells that make dopamine, a chemical needed for smooth movement. Research now shows that the immune system and gut are deeply involved. Inflammation in the brain can leak into the bloodstream, while chronic low-level inflammation in the body can, in turn, worsen damage in the brain. At the same time, people with Parkinson’s often show an imbalance of gut bacteria, and common non-motor symptoms include constipation and other digestive troubles. This two-way traffic among brain, immune system, and gut—sometimes called the gut–immune–brain axis—has become a major focus for new treatments.
A special form of zinc enters the picture
Zinc is a trace mineral crucial for immunity, antioxidant defenses, and brain function. People with Parkinson’s tend to have lower zinc levels in the blood, and animal studies suggest that extra zinc can protect nerves. The compound tested here is a laboratory-made form called 64Zn‑aspartate, in which zinc is paired with the amino acid aspartate and enriched in a naturally light zinc isotope. Earlier work from the same group showed that this form of zinc reduced inflammation and memory problems in an Alzheimer’s-like rat model and eased brain inflammation in a Parkinson’s model. In the current study, the researchers asked whether 64Zn‑aspartate could also calm down inflammation throughout the body and protect the gut ecosystem in rats with an inflammation-driven form of Parkinson’s.
Putting the treatment to the test in rats
To mimic key features of Parkinson’s, the scientists injected a bacterial fragment called LPS directly into a brain region that controls movement. This triggered lasting loss of dopamine-producing cells, movement problems, and heightened anxiety in the animals, along with strong signs of body‑wide inflammation. Some of these Parkinsonian rats then received daily intravenous doses of 64Zn‑aspartate for ten days. Compared with untreated animals, treated rats moved more freely in behavioral tests, showed less anxious behavior, and had fewer abnormal turning movements triggered by a drug that reveals dopamine damage. These changes point to partial protection of the brain’s movement circuitry.
Quieting the immune storm
The team then looked at the rats’ blood and immune cells. In the Parkinsonian animals, classic markers of inflammation were elevated: C‑reactive protein in the blood, an excess of granulocytes and platelets, fewer lymphocytes, and a suite of combined blood-cell ratios that doctors increasingly use to gauge inflammatory burden. Immune cells called monocytes and neutrophils were primed in a highly reactive, pro‑oxidant state, churning out large amounts of damaging reactive oxygen species. After 64Zn‑aspartate treatment, these measures largely returned to levels seen in healthy or sham-operated rats. The thymus—a key organ for maturing immune cells—also regained a more normal size and cell makeup. Overall, the drug pushed immune cells away from a destructive, overactivated profile toward a calmer, more balanced one.
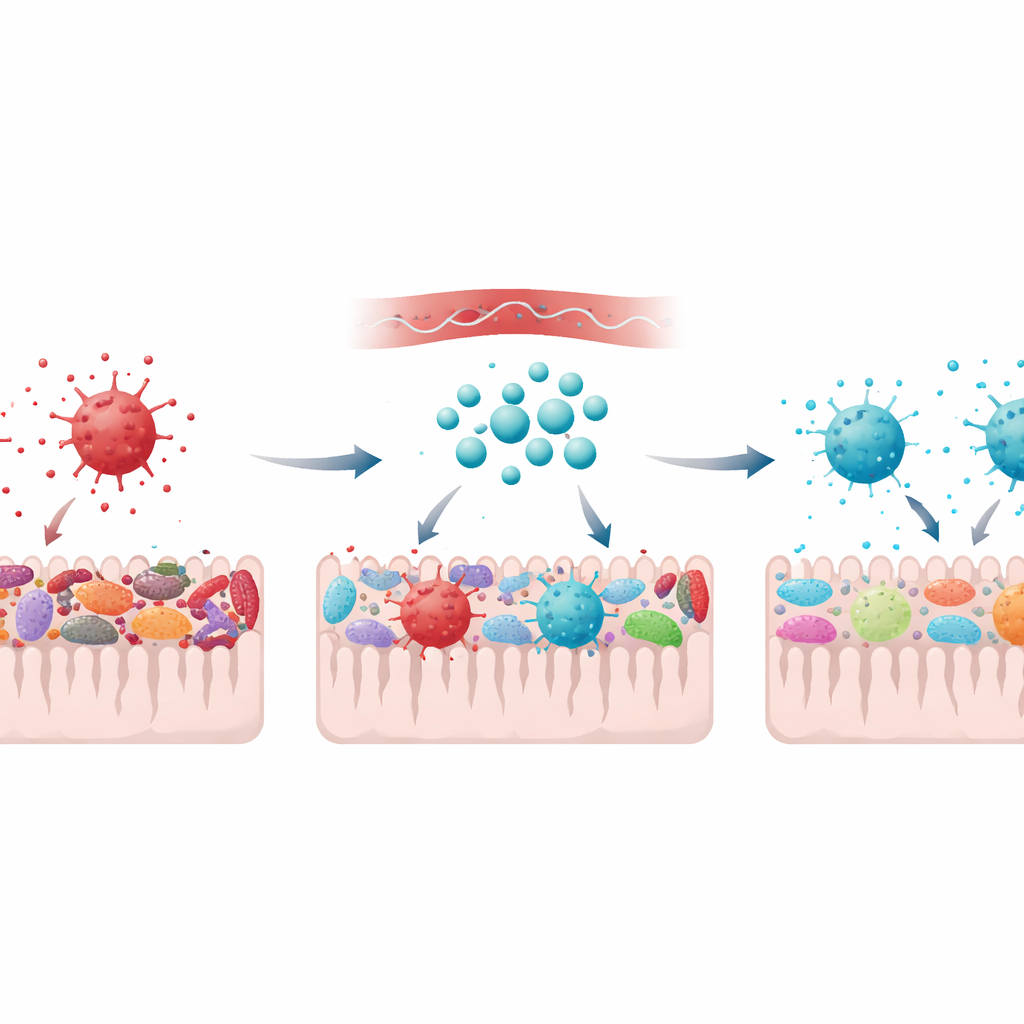
Helping the gut find its balance again
The gut told a similar story. In Parkinsonian rats, beneficial bacteria such as Bifidobacterium and Lactobacillus—which help break down food and produce molecules that talk to the nervous system—were depleted. At the same time, potentially troublesome groups such as Enterobacteriaceae and Staphylococcus expanded, especially in bacteria clinging to the intestinal wall. This shift suggests a more oxygen-rich, inflamed gut environment and echoes patterns seen in people with Parkinson’s. Treatment with 64Zn‑aspartate largely preserved friendly bacteria and held opportunistic species in check, both in the gut lining and in stool. Although the rats still produced somewhat more feces than controls, the microbial community looked noticeably healthier.
What this could mean for people
This work was done in rats, not humans, and it does not prove that 64Zn‑aspartate will work in people with Parkinson’s. But it offers a compelling proof of concept: by targeting systemic inflammation and stabilizing the gut microbiota, it may be possible to protect vulnerable brain cells and improve both movement and mood. The study positions this special zinc compound as a promising candidate in a new generation of treatments that act on the gut–immune–brain axis, aiming not just to ease symptoms but to slow the disease process itself.
Citation: Temnik, M., Rudyk, M., Balakin, A. et al. Isotopically enriched 64ZN-aspartate attenuates systemic inflammation and gut dysbiosis in an LPS-induced rat model of Parkinson’s disease. Sci Rep 16, 10907 (2026). https://doi.org/10.1038/s41598-026-45640-9
Keywords: Parkinson’s disease, systemic inflammation, gut microbiota, zinc therapy, neuroimmune gut axis