Clear Sky Science · en
The fluoroquinoline compound exerts anti-erythroleukemic effects by dual-targeting GLUT1 and the PI3K/AKT signaling pathway
A New Angle on a Dangerous Blood Cancer
Erythroleukemia is a rare but highly aggressive form of blood cancer with few treatment options beyond stem cell transplantation. For many mostly older patients, this means harsh therapy, high costs, and grim odds. This study explores a laboratory-designed compound called FKL-137 that tackles erythroleukemia cells in an unexpected way: by cutting off their favorite fuel, sugar, and triggering their self-destruction. The work suggests a possible new class of targeted drugs that exploit cancer cells’ dependence on altered energy use while sparing more of the healthy body.
When Blood Cells Go Rogue
In erythroleukemia, immature red blood cell precursors grow out of control in the bone marrow and spleen. Patients can develop severe anemia, swollen spleens that are at risk of rupture, and a rapid decline in health. Current therapies are limited, and many people are too frail for aggressive treatment. At the same time, scientists have learned that many blood cancers rely on reprogrammed sugar use: they pull in glucose at high rates and rapidly convert it to energy and building blocks, helping them multiply and evade the immune system. Targeting this metabolic “addiction” has become an attractive strategy for new drugs.
Starving Cancer Cells of Sugar
The researchers focused on a small synthetic molecule, FKL-137, derived from a chemical family already known for anticancer activity. In lab experiments, they exposed two human erythroleukemia cell lines to increasing doses of FKL-137. The compound sharply slowed cell growth over time and pushed the cells into programmed death, as seen by classic changes in cell shape and activation of internal death machinery. Importantly, FKL-137 was much more potent against leukemia cells than against normal liver cells, and measures of drug safety suggested relatively low toxicity at effective doses.
Hitting the Cancer’s Sugar Gateways
Diving deeper, the team asked how FKL-137 interferes with cancer metabolism. They found that treated leukemia cells took up less glucose and released less lactate, a by-product of intense sugar burning. Key proteins that normally help convert glucose to energy and building blocks were dialed down. Using computational modeling, thermal stability tests, and imaging, the researchers showed that FKL-137 binds to GLUT1, a major “gate” on the cell surface that brings glucose inside. When GLUT1 levels were reduced by genetic tricks, leukemia cells grew more slowly and used less sugar, and they became even more sensitive to FKL-137. This confirmed that GLUT1 is both a driver of erythroleukemia cell growth and an important target of the drug.
Disrupting a Growth-Signaling Loop
The study also uncovered effects beyond the glucose gate itself. Erythroleukemia cells normally use a signaling route known as PI3K/AKT to promote survival, growth, and high sugar use, in part by supporting GLUT1. FKL-137 treatment lowered the active forms of key proteins in this pathway, breaking a reinforcing loop between sugar transport and growth signals. In mice infected with a virus that reliably causes erythroleukemia, FKL-137 treatment improved red blood cell levels, shrank enlarged spleens, reduced cancer cell invasion into organs, and corrected abnormal sugar metabolism in spleen tissue—all without obvious damage to the heart, liver, lungs, or kidneys.
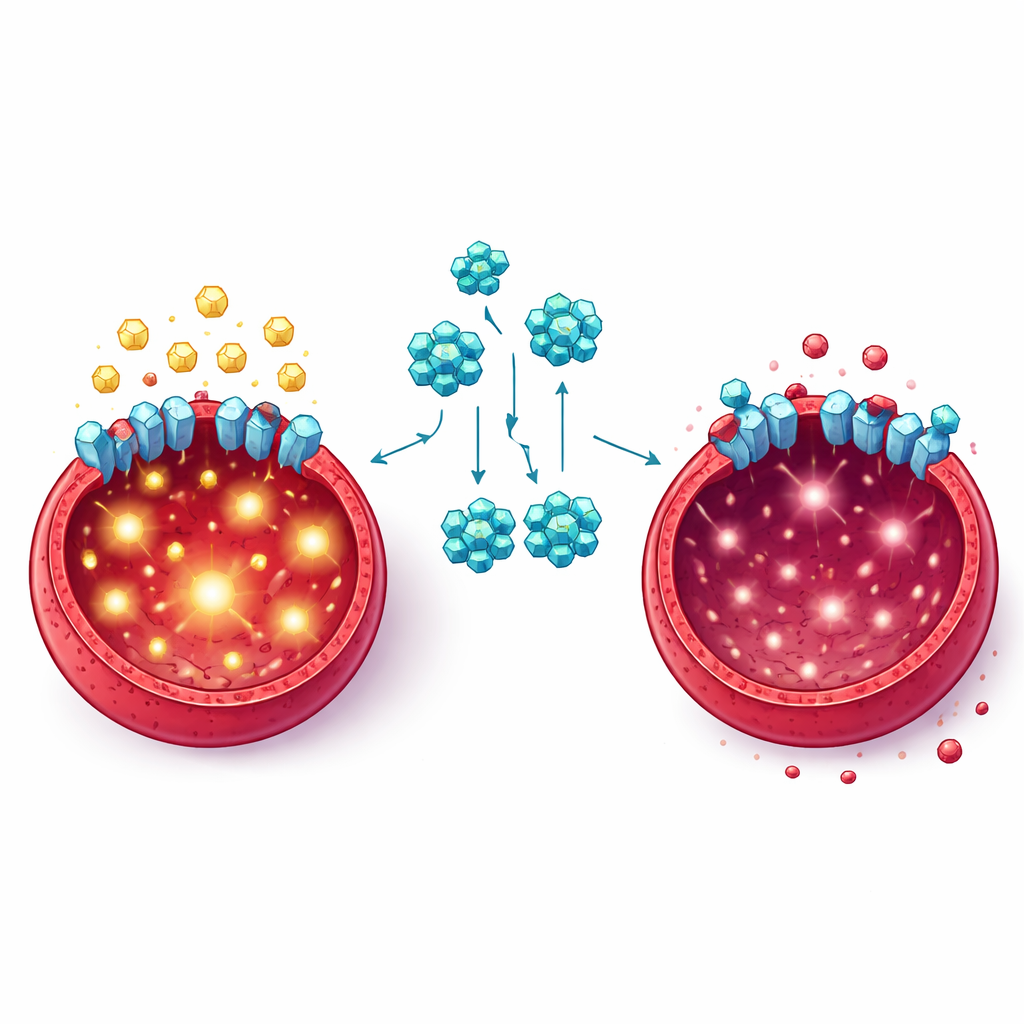
Promise, Limits, and Next Steps
Taken together, the work presents FKL-137 as a dual-action candidate drug that both blocks the main sugar gate of erythroleukemia cells and dampens a powerful growth pathway that depends on that fuel. By starving cancer cells first and then pushing them toward death, the compound offers a conceptually elegant way to exploit a weakness that normal cells do not share to the same degree. The authors caution, however, that FKL-137 still needs refinement: it has a relatively narrow dose window, cancer cells may find ways to compensate by rerouting their metabolism, and findings from cell lines and mouse models must be confirmed in human tissues. Even so, the study strengthens the idea that precisely targeting how cancer cells eat and grow could open a new front in treating this otherwise hard-to-manage blood cancer.
Citation: Cheng, S., Zhao, W., Yu, J. et al. The fluoroquinoline compound exerts anti-erythroleukemic effects by dual-targeting GLUT1 and the PI3K/AKT signaling pathway. Sci Rep 16, 10916 (2026). https://doi.org/10.1038/s41598-026-45597-9
Keywords: erythroleukemia, cancer metabolism, GLUT1 inhibitor, PI3K AKT pathway, targeted therapy