Clear Sky Science · en
Integrated experimental and computational evaluation of Anagallis foemina derived terpenoids against carbapenem resistant Acinetobacter baumannii
Why a garden weed matters for hospital infections
Hospitals around the world are struggling with infections caused by Acinetobacter baumannii, a tough microbe that shrugs off many of our strongest antibiotics. Some strains now resist carbapenems, drugs once reserved as the last line of defense. This study looks to an unassuming wildflower, the blue pimpernel (Anagallis foemina), to see whether its natural chemicals could help disarm these dangerous bacteria and point the way to new medicines.
A rising threat in intensive care
Acinetobacter baumannii thrives on dry hospital surfaces and equipment, and easily infects vulnerable patients with wounds, weakened immune systems, or breathing tubes. Many strains have become multidrug resistant, and some are now extremely or even pan-drug resistant, meaning that almost no available antibiotic works. In particular, this bacterium produces special enzymes called beta-lactamases that destroy carbapenem antibiotics. Because of this, the World Health Organization lists carbapenem-resistant A. baumannii as a top-priority threat, tied to tens of thousands of deaths each year and urgently in need of new treatment strategies.
Turning a wild plant into an experimental medicine
The researchers collected aerial parts of A. foemina from rural Pakistan and prepared an ethanolic extract, essentially drawing out its oily and aromatic components. Using gas chromatography–mass spectrometry, they identified 16 main plant chemicals, including fatty acids, vitamins, and a group of small fragrance-like molecules called terpenoids. Rather than isolating single ingredients first, the team tested the crude extract directly against three clinical A. baumannii strains that were resistant to many antibiotic classes, including carbapenems. They measured how well the extract stopped bacterial growth on plates, how much was needed to halt growth in broth, and whether it could actually kill the bacteria rather than merely slow them down.
Stopping growth, killing cells, and breaking up slime
In petri dish tests, the A. foemina extract created clear rings around the wells where bacteria could not grow, with zones up to about 20 millimeters wide at higher doses—larger than those produced by the reference carbapenem drug under the test conditions. In liquid culture, the minimum amount needed to stop visible growth (MIC) was 1.25 mg/mL, and doubling that dose completely killed 99.9% of the bacterial cells (MBC), giving an MBC/MIC ratio of 2, a hallmark of a truly bactericidal effect. Importantly, the extract also strongly reduced formation of biofilms—sticky, protective layers of cells and slime that coat medical devices and help bacteria survive. At the MIC, biofilm formation fell by about 80–90%, and even at lower, non-lethal doses, biofilms were cut by more than half, suggesting the extract interferes with the way the bacteria organize themselves on surfaces.
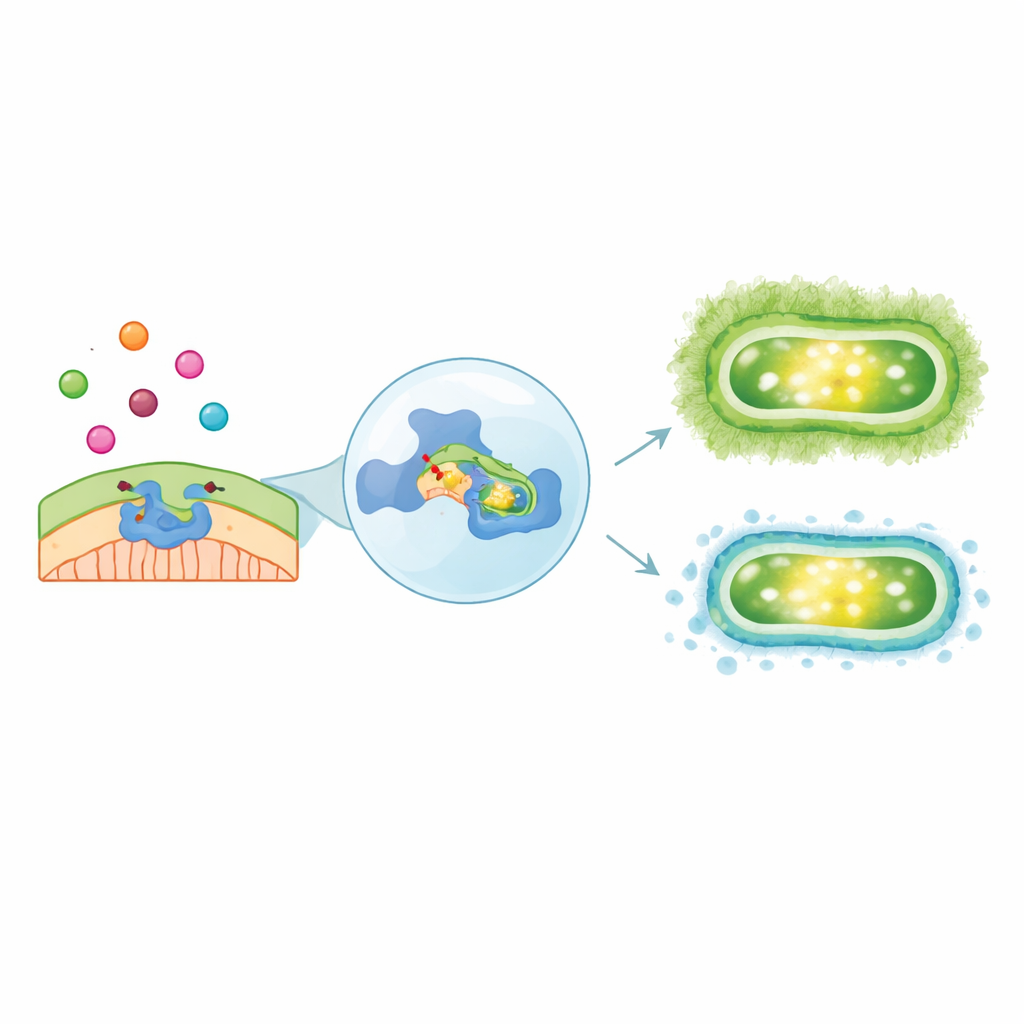
Peering into the bacteria’s defenses on a computer
To understand what might be happening at the molecular level, the team focused on two minor components of the extract, the closely related terpenoids α‑Terpinen‑7‑al and γ‑Terpinen‑7‑al. Although present at only about 1–2% each, similar compounds are known to act on microbes. Using detailed computer models, the researchers docked these molecules onto the structure of an A. baumannii beta-lactamase enzyme (OXA‑24) that helps the bacteria resist carbapenems. Simulations suggested that both molecules nestle into the enzyme’s active site and form a stabilizing contact with a key serine residue at the heart of the chemical reaction. Extended molecular dynamics runs showed that these complexes remained stable over 100 nanoseconds, and energy calculations indicated that α‑Terpinen‑7‑al in particular might bind strongly through hydrophobic contact with surrounding amino acids.
Are these plant compounds drug candidates?
Beyond binding, the study also asked whether these plant molecules look like plausible drugs. Computational checks of absorption, metabolism, and toxicity predicted that both terpenoids are small, reasonably fat‑loving molecules that should cross cell membranes, be well absorbed from the gut, and avoid major red flags such as liver damage, genetic toxicity, or interference with heart rhythm channels. The models suggest they could be given orally and might even reach the brain, though any real-world use would require extensive safety testing in animals and humans, far beyond what computer tools can guarantee.
What this work really tells us
Taken together, the findings show that an extract from the common blue pimpernel can kill highly drug-resistant A. baumannii in the lab and sharply reduce the slimy biofilms that help these bacteria endure in hospitals. Computer simulations point to two rare terpenoids in the extract as promising leads that may block a key resistance enzyme, while other fatty components could rough up bacterial membranes or weaken biofilms. The authors stress that this is an early, exploratory study: the exact mechanisms remain unproven, and no animal or human testing has been done. Still, the work offers a clear message for non-specialists: even humble wild plants may hide new chemical tricks that, with careful study, could help us catch up in the ongoing race against antibiotic-resistant superbugs.
Citation: Afzal, M., Khan, M.U., Naqvi, S.Z.H. et al. Integrated experimental and computational evaluation of Anagallis foemina derived terpenoids against carbapenem resistant Acinetobacter baumannii. Sci Rep 16, 10650 (2026). https://doi.org/10.1038/s41598-026-45012-3
Keywords: antibiotic resistance, Acinetobacter baumannii, medicinal plants, biofilm inhibition, natural product drug discovery