Clear Sky Science · en
Proteomic analysis of papillary thyroid carcinoma in the context of Hashimoto’s thyroiditis
Why this thyroid study matters
Papillary thyroid cancer is the most common form of thyroid cancer, and Hashimoto’s thyroiditis is the most common chronic inflammation of the thyroid. Many people have one or both conditions, and doctors have long suspected a link between them. Yet when Hashimoto’s roughens and scars the gland, standard ultrasound scans struggle to show whether a lump is harmless or cancerous. This study uses a modern "protein fingerprint" approach to look inside thyroid tissue at the molecular level, searching for reliable signals that could help tell a benign nodule from a cancer in patients who already have Hashimoto’s thyroiditis.
Two thyroid problems tangled together
The researchers focused on patients whose thyroids were affected by Hashimoto’s thyroiditis, an autoimmune disease in which the immune system slowly attacks the gland. Some of these patients also had papillary thyroid cancer, while others had only benign nodules. Clinically, their hormone levels often look similar, and imaging can be confusing because Hashimoto’s makes the whole gland look patchy. That makes it hard for doctors to decide who truly needs surgery and how aggressive treatment should be. The team set out to see whether the proteins present in the tissue itself could reveal a clearer distinction between benign and malignant nodules in this setting.
Reading the thyroid’s protein fingerprints
From surgical samples preserved in wax blocks, the scientists extracted proteins and broke them into small pieces. They then used high‑precision mass spectrometry and advanced liquid chromatography to identify and measure thousands of proteins at once. Statistical analyses showed that the overall protein profiles of cancerous nodules in Hashimoto’s glands were clearly different from those of benign nodules in similar glands. In total, they found 93 proteins whose levels changed markedly: 72 were higher and 21 were lower in the cancer group. This broad shift hinted that the tissue around the tumor is being rebuilt in a distinctive way when cancer is present.
Changes in the tissue scaffolding
When the team mapped these proteins onto known biological pathways, a consistent theme emerged: many of the altered proteins were involved in the extracellular matrix, the fibrous scaffold that surrounds and supports cells. In particular, the changes pointed to a process called epithelial‑mesenchymal transition, in which orderly, sheet‑like cells loosen their attachments and adopt a more mobile, invasive behavior. Network analysis of how the proteins interact with one another highlighted several hub molecules. Among them, two stood out in the cancer samples: THBS2 and COL12A1, both associated with matrix remodeling and the physical microenvironment that can either restrain or promote tumor spread.
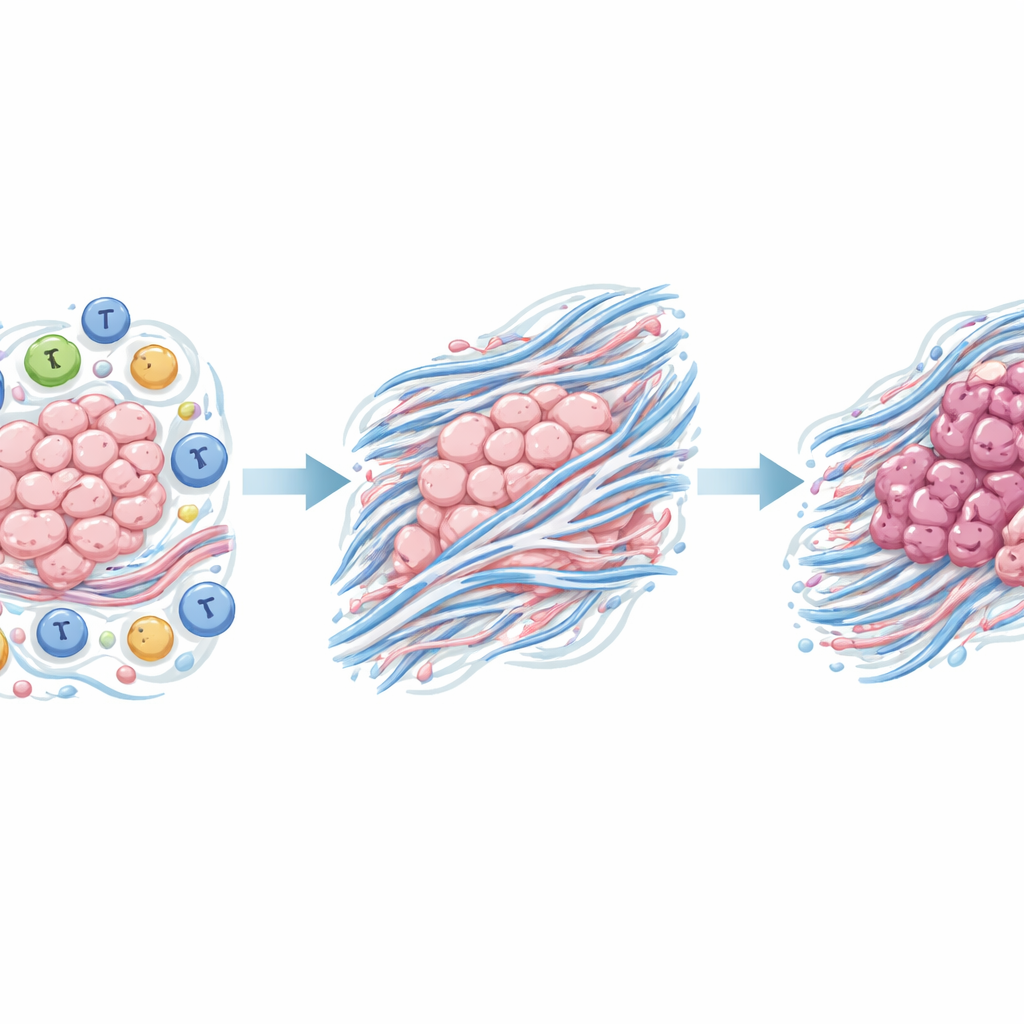
Zeroing in on a promising marker
To move from big data to practical use, the researchers tested these key proteins directly in additional tissue sections from 35 patients using standard hospital staining methods. They found that COL12A1, a form of collagen that helps organize the matrix around cells, was present much more often in papillary thyroid cancers arising in Hashimoto’s glands than in benign nodules from similar glands: roughly three‑quarters of cancerous cases showed strong staining, compared with less than one‑third of benign nodules. THBS2, by contrast, was commonly seen in both groups, likely reflecting the underlying inflammation of Hashimoto’s rather than cancer itself.
What this could mean for patients
The study suggests that papillary thyroid cancer developing in the background of Hashimoto’s thyroiditis is closely tied to a reshaping of the tissue scaffold around thyroid cells, and that COL12A1 is a visible sign of this remodeling. While the work involved relatively few patients and will need to be confirmed in larger groups, COL12A1 emerges as a promising candidate marker to help pathologists and, eventually, clinicians distinguish malignant from benign nodules in people with Hashimoto’s disease. If validated, it could become part of a more precise toolbox for diagnosing thyroid cancer when standard scans and hormone tests are not enough.
Citation: Zhou, H., Tan, G., Sun, H. et al. Proteomic analysis of papillary thyroid carcinoma in the context of Hashimoto’s thyroiditis. Sci Rep 16, 10938 (2026). https://doi.org/10.1038/s41598-026-44977-5
Keywords: thyroid cancer, Hashimoto’s thyroiditis, papillary thyroid carcinoma, biomarkers, proteomics