Clear Sky Science · en
Pyrometallurgical valorization of waelz, fayalite, and linz-donawitz slag mixtures
Turning Waste from Metal Plants into New Resources
Across the globe, steel and non‑ferrous metal plants generate mountains of glassy, rock‑like leftovers called slags. These heaps often end up in landfills, taking up space and slowly releasing trace metals into the environment. This study shows that three of the most common slags can instead be melted together in an electric furnace to recover valuable metals like iron, copper, and vanadium while producing safe building and ceramic materials—an example of how heavy industry can move toward a true circular economy.
Why These Industrial Piles Matter
Slags from zinc recycling (Waelz slag), steelmaking (Linz‑Donawitz, or LD slag) and copper production (fayalite slag) are produced in tens of millions of tons every year. They contain useful metals but also elements that can be harmful if they leach into soil and water. At present, only a fraction is reused in construction; much is stockpiled. The authors argue that treating slags as a combined secondary ore, rather than a waste, could both reduce the demand for freshly mined raw materials and cut the long‑term environmental burden of disposal sites.
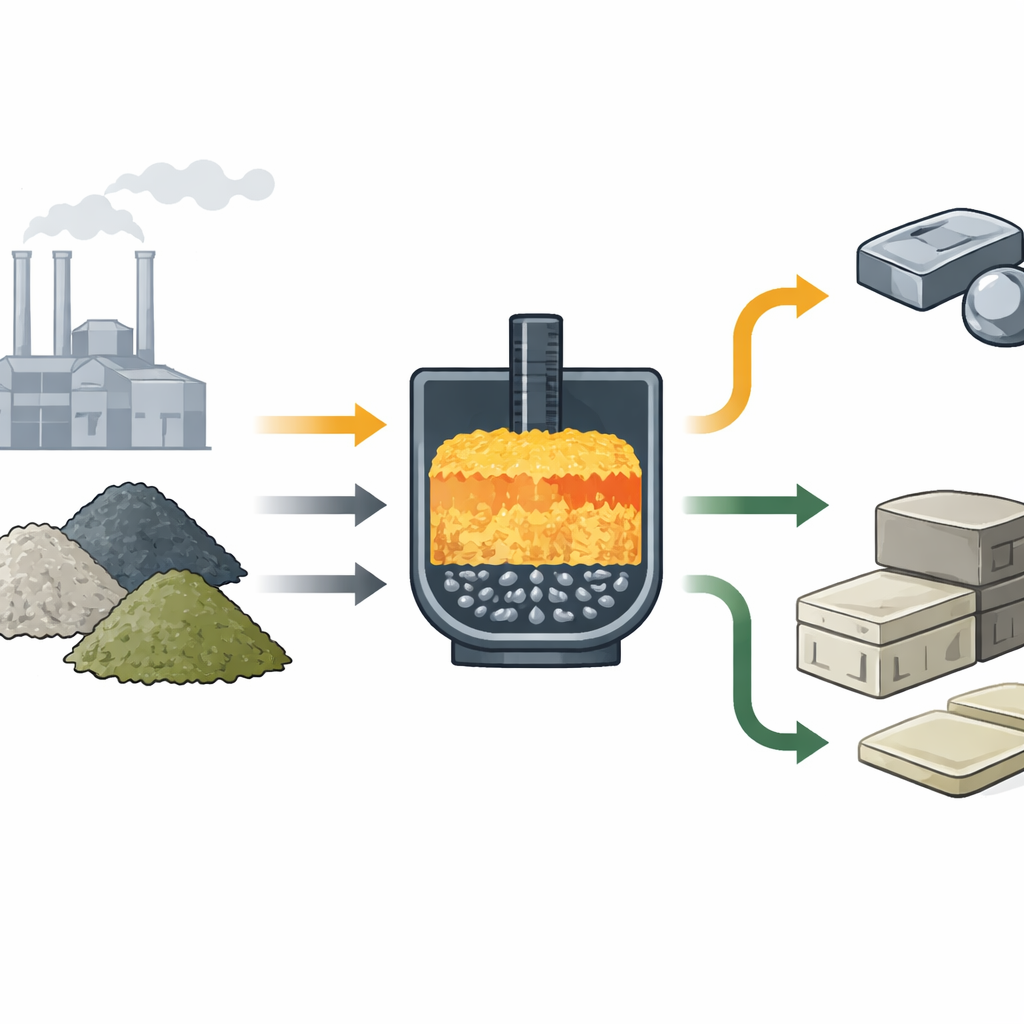
Designing the Right Slag Cocktail
The core idea is to blend the three slags so that, when heated, they act as their own “flux”—the material that controls how easily the melt flows and how well molten metal separates from molten slag. The team used open‑source software (R) and industrial thermodynamic tools to choose mixing ratios that give a simple target: a balance between lime‑rich and silica‑rich components that makes the melt fluid enough at 1450 °C without adding new minerals from mines. They mapped all workable combinations in triangular diagrams that show where the mixture is fully molten and how sticky, or viscous, it will be. These diagrams act like a recipe map for plant operators, showing which blends fall into a “sweet spot” for smooth smelting.
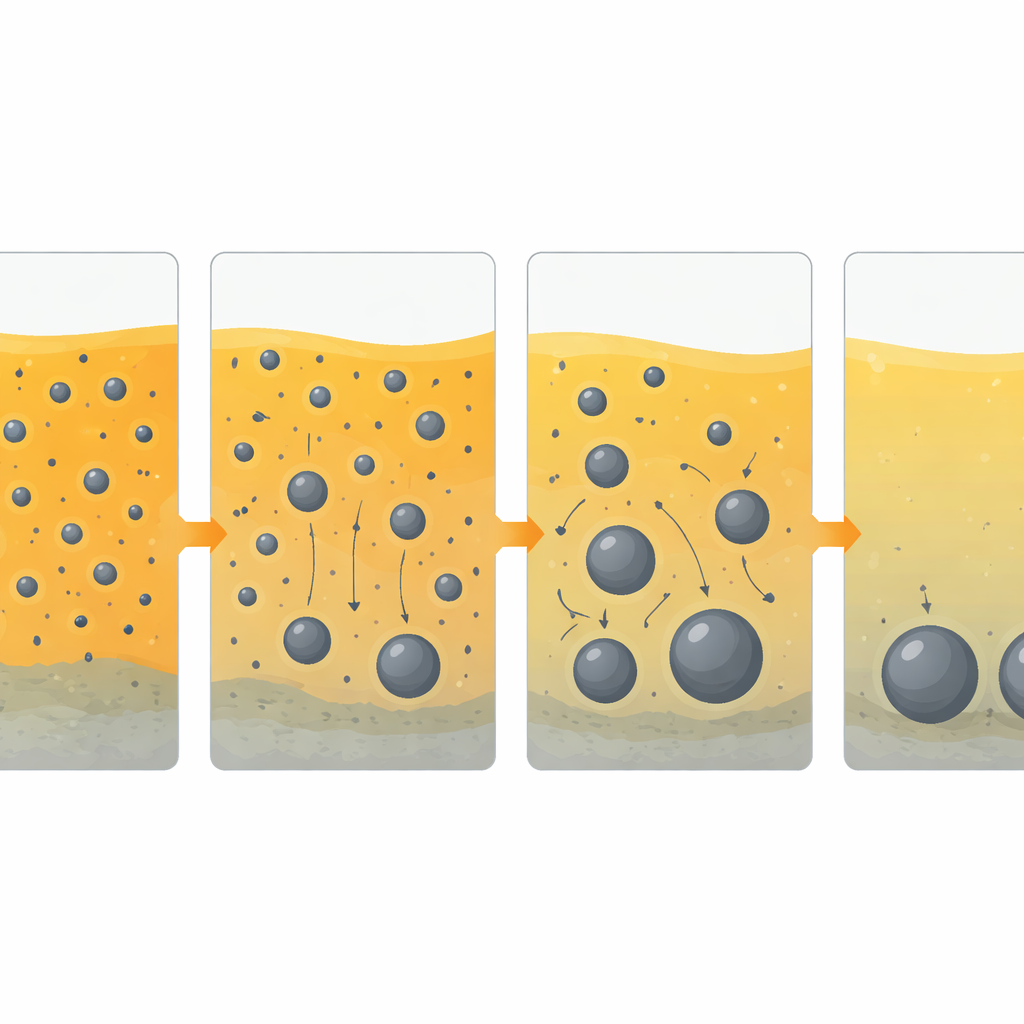
Watching Metal Droplets Grow Inside the Melt
To test their designs, the researchers first ran small smelting trials in tube furnaces, then scaled the best mixture up to a two‑kilogram test in an induction furnace—similar in principle to industrial electric steelmaking. They used X‑ray computed tomography, a three‑dimensional imaging method akin to a medical CT scan, to look inside the solidified products without cutting them apart. In these images, dense metal appears bright and the lighter slag appears dark, revealing how metal droplets form, move, collide and merge into larger nuggets. By comparing ten different mixes, they found that the size and position of metal nuggets depend on a delicate balance between how thick the slag is and how strongly metal and slag repel each other at their boundary.
Finding the Best Mix for Metal Recovery
Chemical modeling and experiments showed that iron is almost completely reduced from all mixtures, but in some cases it remains trapped as many tiny droplets scattered through a viscous slag. Blends with just the right amount of magnesium‑bearing components and moderate viscosity allowed droplets to coalesce into big, dense nuggets that sink to the bottom and are easy to recover. One mixture—about 41% LD slag, 10% Waelz slag and 48% fayalite slag—stood out. In the scaled‑up induction furnace test, this recipe produced a roughly 700‑gram metal nugget containing mostly iron, with notable amounts of manganese, copper and vanadium. Zinc, meanwhile, left with the gas phase as a fume that could be collected separately.
From Black Glass to Bricks and Porcelain
The slag left after smelting is not just cleaner; it is also useful. The team tested how easily metals leach from both the raw and a gently roasted version of the slag using standard environmental procedures from the United States and Europe. The unroasted slag met the strict criteria to be considered non‑hazardous and suitable for use as a granular construction material under US, UK and German rules. When the slag was reheated in air at 900 °C, its internal minerals changed into phases commonly found in ceramic bodies and porcelain, including albite and anorthite. This points to further uses in tiles, glazes, or other engineered ceramics, although these applications still need practical testing.
Closing the Loop for Heavy Industry
In practical terms, the study shows that mixtures of three major industrial slags can be melted in an electrified furnace to recover a vanadium‑bearing pig iron—potentially a feedstock for steelmaking—while generating a secondary slag that is safe for construction and promising for ceramics. By carefully tuning simple composition ratios instead of adding fresh fluxes, the method cuts both costs and environmental impacts. The work offers a blueprint for how metallurgical plants might turn long‑lived waste piles into streams of new products, helping to close material loops in one of the world’s most resource‑intensive sectors.
Citation: Romero, J.L., Recksiek, V., Debastiani, R. et al. Pyrometallurgical valorization of waelz, fayalite, and linz-donawitz slag mixtures. Sci Rep 16, 9539 (2026). https://doi.org/10.1038/s41598-026-44763-3
Keywords: slag recycling, circular economy, pyrometallurgy, metal recovery, industrial waste valorization