Clear Sky Science · en
Investigating Escherichia coli Colicin E9 immunity protein interactions with DNA gyrase of Pseudomonas aeruginosa: advanced computational approach for developing novel antimicrobial strategies
Turning Bacterial Weapons into New Medicines
As antibiotic resistance spreads, doctors are running out of ways to stop dangerous infections. Some of the most stubborn offenders, like Pseudomonas aeruginosa, can shrug off many drugs. This study looks to bacteria themselves for fresh ideas, exploring how one microbe’s built-in protective protein might be repurposed to disable a vital enzyme in another microbe. By using advanced computer simulations, the researchers show how a small "immunity" protein can cling tightly to a key bacterial enzyme, hinting at a new route for future antimicrobials.
A Tiny Shield Against a Deadly Toxin
Certain strains of Escherichia coli produce powerful protein toxins called colicins that can kill neighboring bacteria. To avoid poisoning themselves, these bacteria also make matching immunity proteins. One such protector, known as the Colicin E9 immunity protein (often called Im9), locks onto the toxin’s cutting domain and prevents it from damaging the host’s DNA. Because this partnership is so specific and strong, scientists have long suspected that understanding it in detail could reveal new ways to control harmful bacteria. In this work, the authors ask whether Im9 might also latch onto DNA gyrase, an essential enzyme in Pseudomonas aeruginosa that manages how its DNA is coiled and copied.
Targeting a Vulnerable Enzyme in a Hard-to-Treat Germ
Pseudomonas aeruginosa is a major hospital pathogen, able to survive in harsh environments and resistant to many drugs. DNA gyrase is one of its most important enzymes, responsible for keeping the bacterium’s long DNA strands properly twisted so they can be replicated. Because blocking this enzyme can halt bacterial growth, it is already a proven target for some antibiotics. The authors used a deep-learning tool to scan the three-dimensional structure of Pseudomonas DNA gyrase and flag likely "hotspots"—clusters of amino acids on its surface that are most important for binding partners. These regions form the enzyme’s active pocket, where normal DNA processing occurs and where a potential inhibitor would ideally attach.
Simulating a Molecular Handshake
To see whether Im9 could grip these hotspots, the researchers turned to molecular docking programs that virtually "fit" proteins together like 3D puzzles. They first cleaned and completed the available structures of both Im9 and DNA gyrase, repairing a missing loop in the enzyme and running short simulations to relax any strained regions. They then used two complementary docking tools, ClusPro and LightDock, to generate many candidate complexes. From these, they selected the most promising arrangements and subjected them to long molecular dynamics simulations spanning hundreds of nanoseconds. These time-resolved movies allowed them to watch how the two proteins adjusted, flexed, and settled into more stable shapes when bound together.
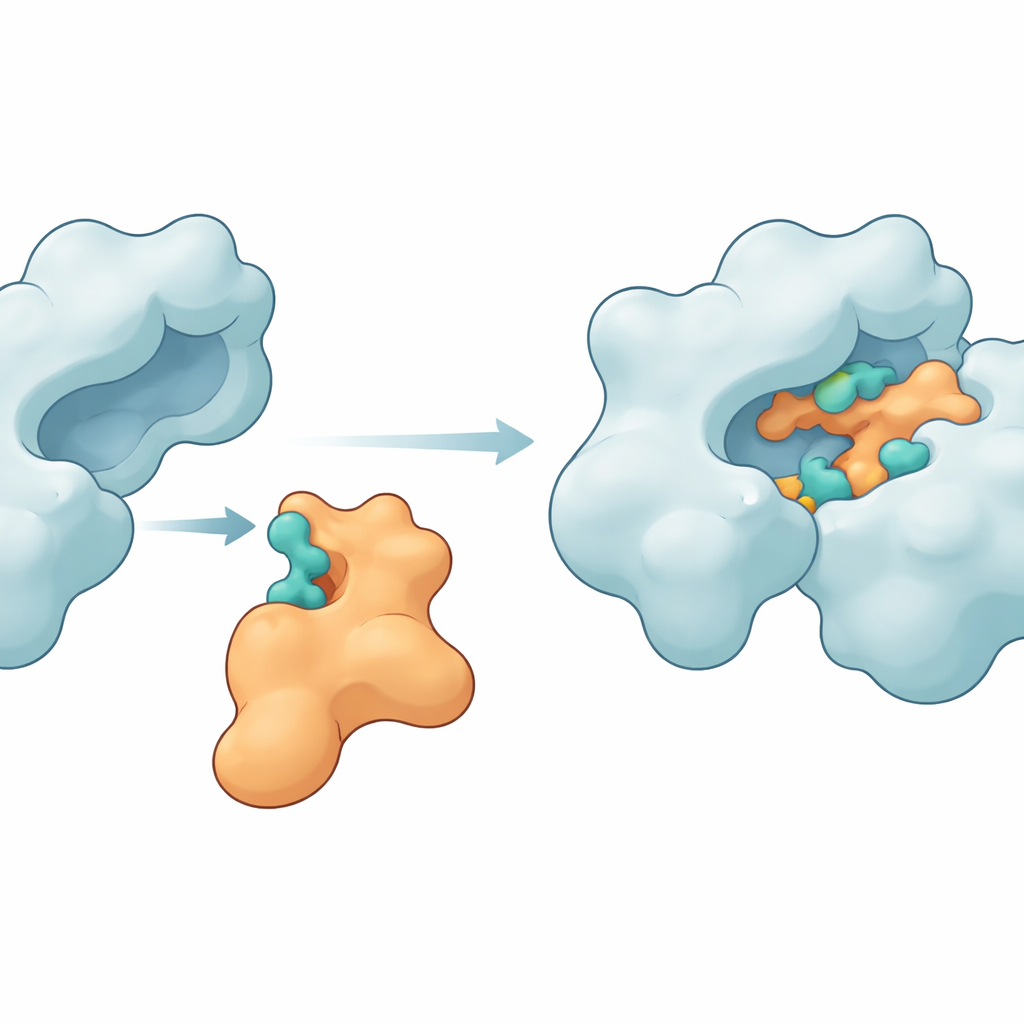
Key Contact Points that Hold the Proteins Together
The simulations revealed that Im9 can indeed form a snug and persistent complex with DNA gyrase. Several amino acids on the enzyme—such as MET27, ASP47, LYS105, LEU198, ASN199, ARG191, and GLU194—repeatedly formed hydrogen bonds and other attractive forces with matching sites on Im9. In one leading model, the two partners maintained between six and ten hydrogen bonds over most of the simulation, a sign of a strong and well-organized interface. Other structural measures, including how compact the proteins remained and how much their shapes fluctuated, showed that the enzyme stayed intact while the immunity protein flexed just enough to mold itself around the gyrase surface. Energy calculations using the MM-GBSA method further supported the idea that these contacts create a favorable, if modest, binding free energy, dominated by electrostatic and van der Waals contributions.
From Computer Models to Future Antimicrobials
Taken together, the results suggest that the Colicin E9 immunity protein can bind stably to the active region of Pseudomonas DNA gyrase, forming a long-lived complex that could, in principle, block the enzyme’s normal role in DNA handling. While these findings are based entirely on computer models and still require experimental testing, they provide a detailed blueprint of where and how a protein-based inhibitor might attach. For non-specialists, the key message is that nature’s own bacterial weapons and shields can inspire new strategies against hard-to-treat infections. By understanding this microscopic handshake in atomic detail, scientists move a step closer to designing novel antimicrobials that shut down crucial bacterial enzymes without harming human cells.
Citation: Alfaraj, R., Alkathiri, F. & Chikhale, R. Investigating Escherichia coli Colicin E9 immunity protein interactions with DNA gyrase of Pseudomonas aeruginosa: advanced computational approach for developing novel antimicrobial strategies. Sci Rep 16, 10786 (2026). https://doi.org/10.1038/s41598-026-44427-2
Keywords: antibiotic resistance, DNA gyrase, protein–protein interactions, computational drug design, Pseudomonas aeruginosa