Clear Sky Science · en
Spectroscopic fingerprinting of extracellular vesicles from diverse cellular origins by ATR-FTIR for vibrational biomarkers of vector–host interactions
Tiny Messengers With Big Stories
All around us, every cell in our bodies—and even in mosquitoes—is constantly releasing nanosized bubbles called extracellular vesicles. These particles shuttle molecular messages between cells and can reveal what is happening inside tissues long before we feel sick. This study shows how a quick, label‑free kind of infrared “listening” can read the chemical fingerprints of these vesicles, even telling apart human and mosquito particles that may be involved in spreading viral diseases like dengue or Zika.
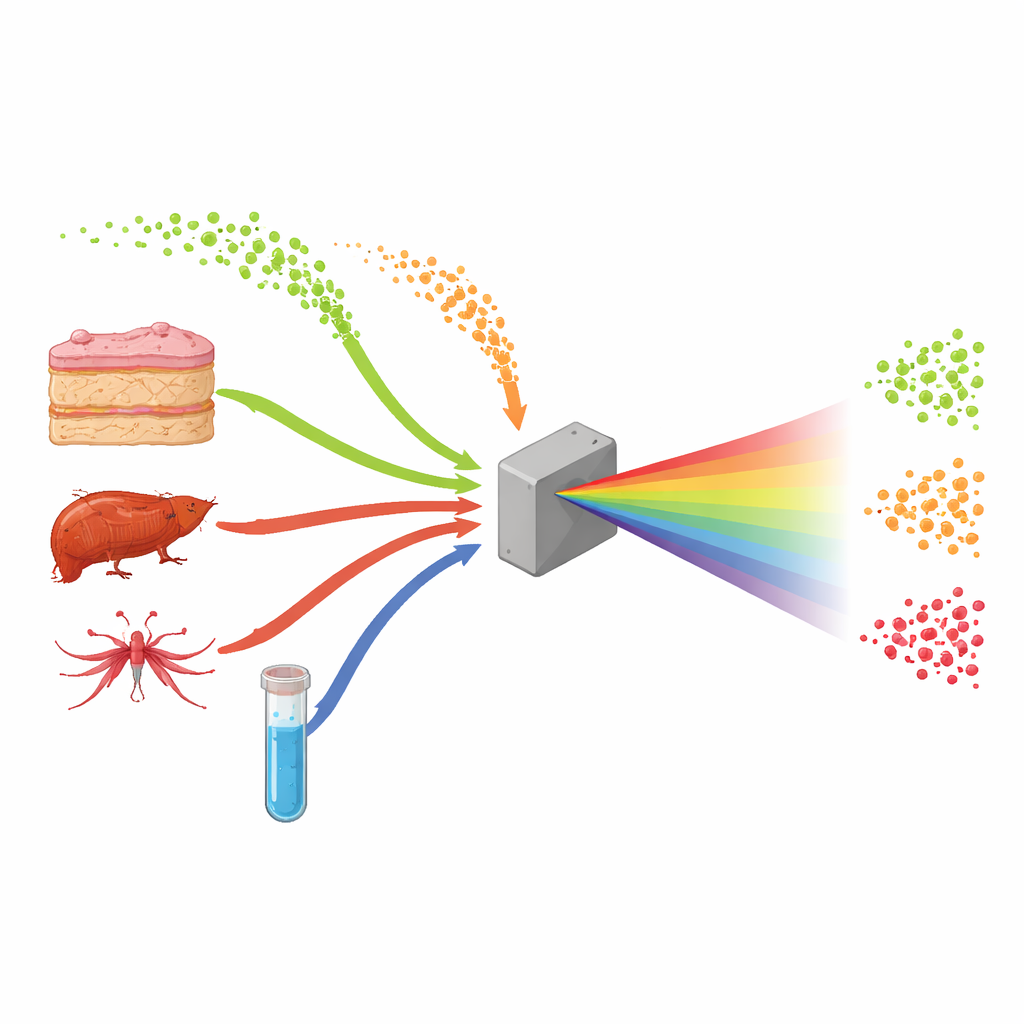
Nanobubbles Bridging Humans and Mosquitoes
Extracellular vesicles are tiny, membrane‑wrapped spheres that carry proteins, fats, and genetic material from one cell to another. They help coordinate normal body functions, but viruses can also hijack them to move silently between cells and even between species. While vesicles from human tissues have been widely explored as disease markers, those made by mosquitoes and other biting insects remain poorly understood, despite their importance for transmitting viruses from vector to host. The authors set out to compare vesicles from both sides of this relationship—human cells and mosquito cells—as well as artificial vesicles built in the lab, to see whether their molecular makeup could be distinguished using infrared light alone.
Reading Vesicle Chemistry With Light
Instead of using time‑consuming and expensive methods that require many labels and reagents, the team turned to a technique called ATR‑FTIR spectroscopy. In simple terms, a tiny droplet containing billions of vesicles is placed on a special crystal and illuminated with infrared light. Different chemical bonds within the vesicles—those in fats, proteins, and genetic material—vibrate and absorb light at specific wavelengths, creating a pattern much like a bar code. The researchers first carefully grew three kinds of cells: human skin fibroblasts, human liver‑like hepatocytes, and mosquito cells from the species Aedes albopictus. They purified the vesicles they released, checked their size and shape with nanoparticle tracking and electron microscopy, and created a fourth group of synthetic vesicles made from defined lipids and simple cargo to act as a clean reference.
Patterns in the Vibrations
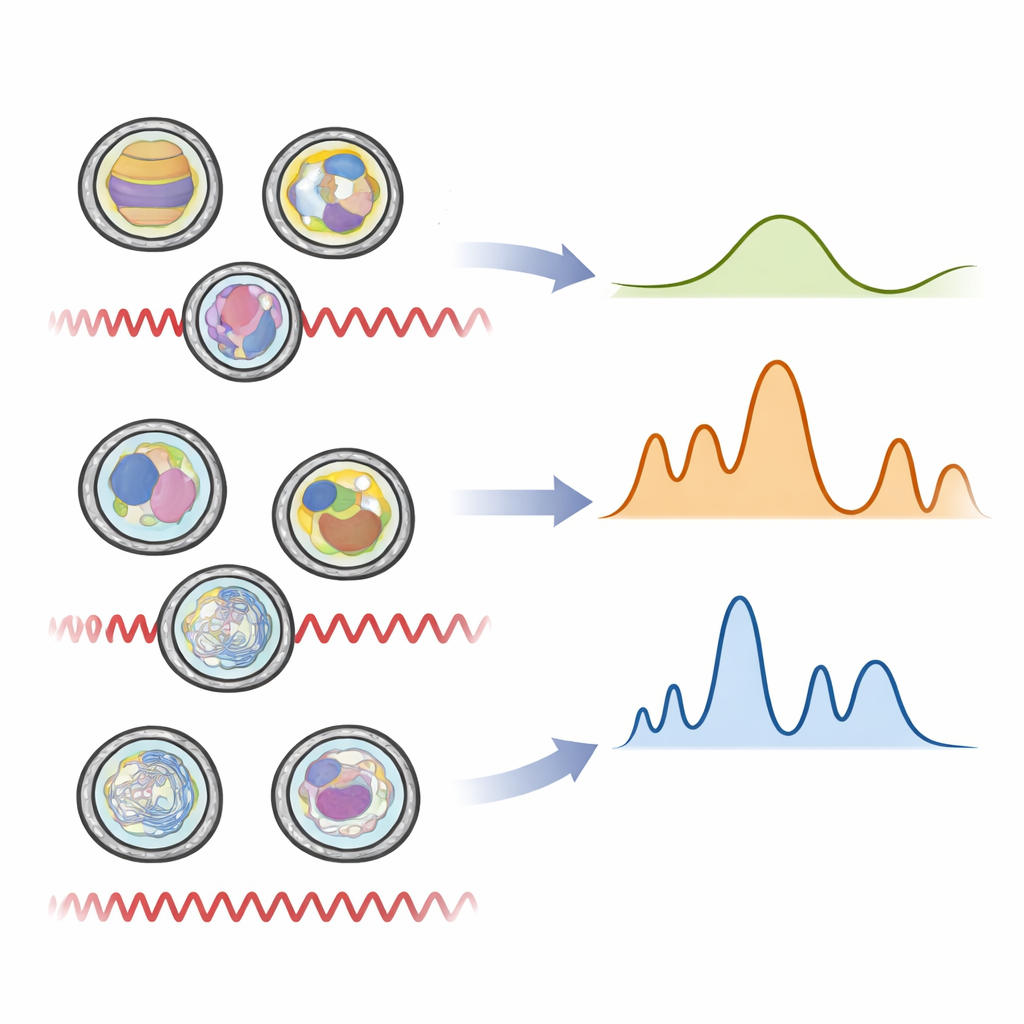
When the infrared spectra from hundreds of vesicle samples were analyzed with advanced statistics, clear groupings emerged. Unsupervised methods, which simply look for natural clusters in the data, already separated mosquito, liver, skin, and synthetic vesicles based on their vibrational patterns. Supervised approaches that explicitly asked the computer to learn the differences went further, pinpointing the exact wavelength regions that mattered most. These key regions corresponded to the chemical signatures of membrane fats, protein “backbones,” sugar decorations on the surface, and nucleic acids inside. Mosquito vesicles showed stronger signals from certain lipids, matching what is known about insect membranes being more flexible and lower in cholesterol. In contrast, vesicles from human liver and skin cells were richer in protein‑related signals, reflecting the more complex signaling and metabolism of mammalian tissues.
From Fingerprints to Fast Identification
By combining these spectral fingerprints with machine‑learning style models, the researchers could correctly identify vesicles from their origin with around 90 percent accuracy or better. The method not only distinguished mosquito from human vesicles, but also separated the two human sources from each other and from the synthetic particles. Importantly, this was achieved without any labels, antibodies, or sequencing—only a tiny amount of sample and a brief infrared measurement. These results suggest that the overall balance of fats, proteins, sugars, and genetic material in vesicles is tightly linked to the cell type that produced them, and that this balance is robust enough to be read out quickly by light.
Why This Matters for Infections and Diagnostics
For a non‑specialist, the key message is that we now have a fast way to “listen in” on the chemical chatter carried by nanosized bubbles from both humans and mosquitoes. The study delivers the first detailed infrared fingerprints of mosquito‑derived vesicles and shows that these patterns can serve as reliable signatures of their origin. In the future, similar measurements could help track which tissues or hosts vesicles and viruses come from in a simple blood or saliva sample, aiding early infection surveillance or monitoring organ damage. As portable infrared instruments and data‑analysis tools improve, this approach could evolve into a practical, label‑free test that screens complex samples for hidden signs of disease and vector–host interactions by reading the vibrational fingerprints of the tiniest messengers.
Citation: Sevinis Ozbulut, E.B., Hoshino, K., Furushima, Y. et al. Spectroscopic fingerprinting of extracellular vesicles from diverse cellular origins by ATR-FTIR for vibrational biomarkers of vector–host interactions. Sci Rep 16, 9195 (2026). https://doi.org/10.1038/s41598-026-44338-2
Keywords: extracellular vesicles, infrared spectroscopy, mosquito vectors, viral transmission, liquid biopsy