Clear Sky Science · en
Analysis of aortic valve prostheses using advanced cardiovascular imaging—a patient-specific reversed translational approach
Why heart valve choices matter
As more people live long enough to develop worn-out heart valves, doctors are replacing these tiny gateways in the heart with artificial ones. Today, many patients receive valves threaded in through a blood vessel, avoiding open-heart surgery, while others get newer “rapid-deployment” surgical valves designed to speed operations. Yet these devices are not all created equal. This study looks under the hood of four widely used aortic valve prostheses to see how they really shape blood flow, using a life-sized 3D-printed model of a patient’s aorta and cutting-edge imaging tools.
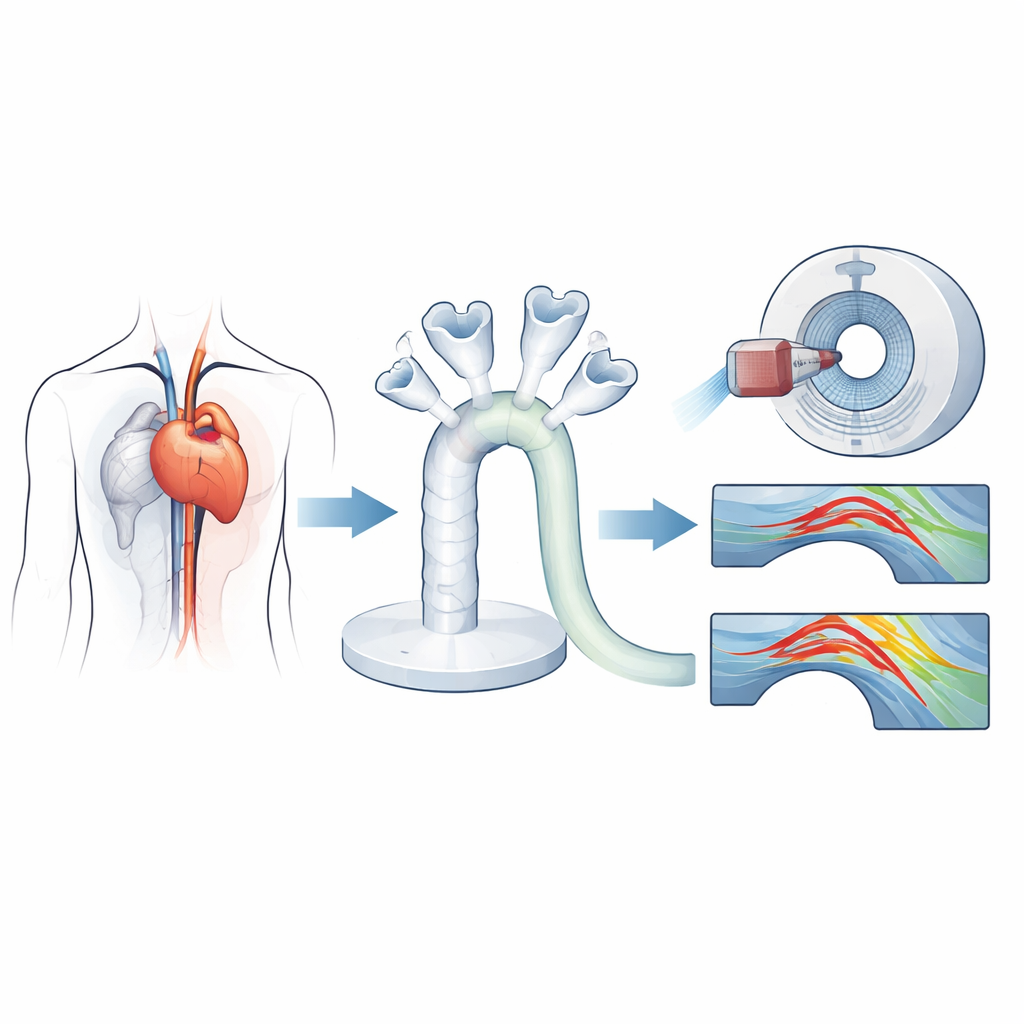
Building a realistic test bed
The researchers began with a high-resolution CT scan of a woman whose natural aortic valve needed replacement and whose valve opening was on the small side—a common and clinically challenging situation. From this scan they digitally reconstructed her aortic root and arch, then 3D-printed a flexible, transparent model of her aorta. Into identical copies of this phantom they implanted four modern biological valves: two rapid-deployment surgical valves and two catheter-based valves delivered as in transcatheter aortic valve replacement. A computer-controlled pump pushed a blood-like fluid through this system, mimicking a normal heartbeat, pressure, and flow rate so that each prosthesis could be tested under the same lifelike conditions.
Watching blood move in four dimensions
To see how each valve reshaped blood flow, the team combined two advanced imaging methods. Vector-flow ultrasound provided real-time maps of how fast and in what direction fluid moved at key cross-sections of the ascending aorta. Four-dimensional flow MRI then captured the full three-dimensional flow patterns over time, allowing the researchers to calculate detailed quantities such as wall shear stress (the friction of blood sliding along the vessel wall), pressure drops along the aorta, kinetic energy loss, and the effective opening area through which blood actually passed. Together, these measurements revealed not just whether blood got through the valve, but how smoothly or turbulently it traveled downstream.
Different valves, different flow stories
The study found that the two catheter-based valves and the two rapid-deployment surgical valves produced noticeably different flow behaviors, even though all were formally sized to fit the same patient’s anatomy. In general, rapid-deployment valves generated higher average flow speeds than the catheter-based valves, while the catheter-based valves tended to produce a more triangular-shaped opening jet. One rapid-deployment model showed particularly high kinetic energy loss and pressure gradients along the aorta, meaning more energy was wasted pushing blood through and beyond the valve. By contrast, another rapid-deployment valve with a slightly larger effective opening area allowed blood to pass with less resistance and lower energy loss, despite being labeled for the same nominal annulus size.
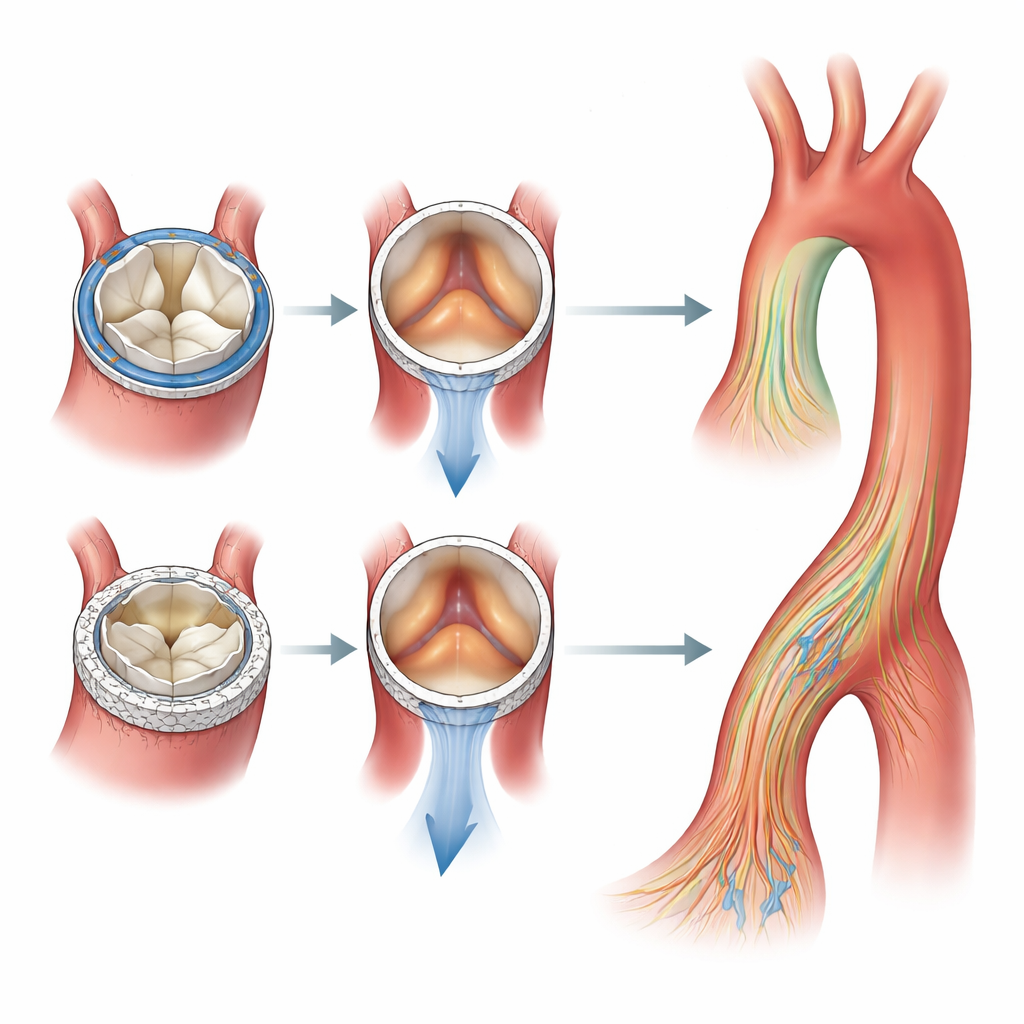
Subtle flow patterns and vessel stress
When the team examined the forces acting on the vessel wall, they observed pockets of elevated shear stress at predictable sites: near the aortic root, along the outer curve of the ascending aorta, around the arch, and in parts of the descending aorta. These hot spots appeared for all four valves, and there were no dramatic differences in where they occurred between surgical and catheter-based devices. Nonetheless, the distribution and magnitude of flow jets and swirling patterns differed in ways that could matter over many years, potentially influencing how the vessel wall remodels or how well a given patient tolerates a particular prosthesis.
Toward more personalized valve selection
For non-specialists, the key message is that choosing a heart valve is not as simple as matching a label size to a measured opening. In this carefully controlled, patient-specific model, valves meant for the same anatomy behaved quite differently, with one rapid-deployment valve emerging as the most energy-efficient and gentle on the vessel overall. The work shows that 3D-printed aortas combined with advanced imaging can act as a kind of test track for new and existing valves, helping doctors anticipate how a device will perform in a particular body. Over time, approaches like this could lead to clearer standards for valve sizing and selection, reduce mismatches between valves and patients, and make it easier to tailor life-saving heart valve therapy to each individual.
Citation: Grefen, L., Herz, C., Flexeder, J. et al. Analysis of aortic valve prostheses using advanced cardiovascular imaging—a patient-specific reversed translational approach. Sci Rep 16, 9334 (2026). https://doi.org/10.1038/s41598-026-44295-w
Keywords: aortic valve replacement, heart valve prosthesis, 3D printed aorta, 4D flow MRI, transcatheter valve