Clear Sky Science · en
Modeling Clostridioides difficile toxin pathogenesis and antiserum protection in an immunocompetent intestine-on-chip platform
Why this gut infection model matters
Antibiotics save lives, but they can also leave the gut vulnerable to dangerous infections. One of the worst offenders is Clostridioides difficile, a bacterium that can cause severe diarrhea and life‑threatening inflammation of the colon, especially in hospitalized patients. This study introduces a miniature “intestine on a chip” that recreates key features of the human gut, including blood vessels and immune cells, to watch in real time how C. difficile toxins damage the intestine and how antibody treatments might stop that injury.
From stubborn hospital bug to gut damage
C. difficile thrives in people whose normal gut bacteria have been disrupted, often after a course of broad‑spectrum antibiotics. The microbe survives harsh conditions as hardy spores and, once established in the colon, releases two major toxins known as TcdA and TcdB. These toxins attack intestinal cells, loosen the seals between them, and trigger waves of inflammation. The result can range from nagging diarrhea to pseudomembranous colitis, a condition marked by sheets of dead tissue and immune cells coating the bowel wall. Current treatments rely mainly on more antibiotics and sometimes stool transplants, yet many patients suffer repeat infections, underscoring the need for better ways to study the disease and test toxin‑blocking therapies.
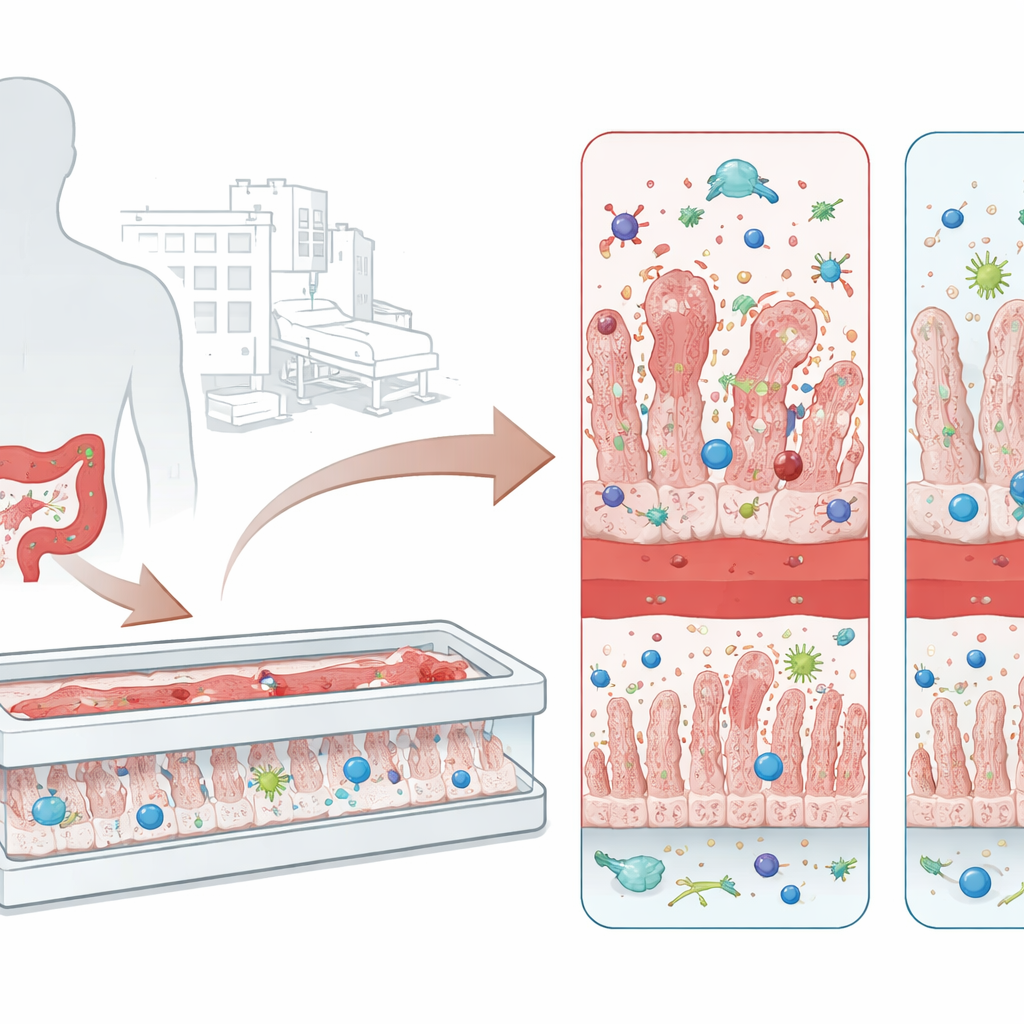
Building a tiny living gut on a chip
Traditional lab models grow a flat sheet of intestinal cells in a dish. While useful, these two‑dimensional cultures lack blood vessels, immune cells, and the gentle fluid flow that shape a real intestine. The researchers instead used a microengineered “intestine‑on‑chip” device with two parallel channels separated by a porous membrane. One side is lined with intestinal cells that grow into finger‑like ridges resembling villi and form a tight barrier. The opposite side carries human blood vessel cells. Into this miniature tissue, the team added monocyte‑derived macrophages that settle into the gut wall and circulating neutrophils that can travel through the vessel channel and infiltrate the tissue, creating an immunocompetent intestine‑on‑chip (i‑IoC) that more closely mimics the living gut.
Watching toxins injure the barrier
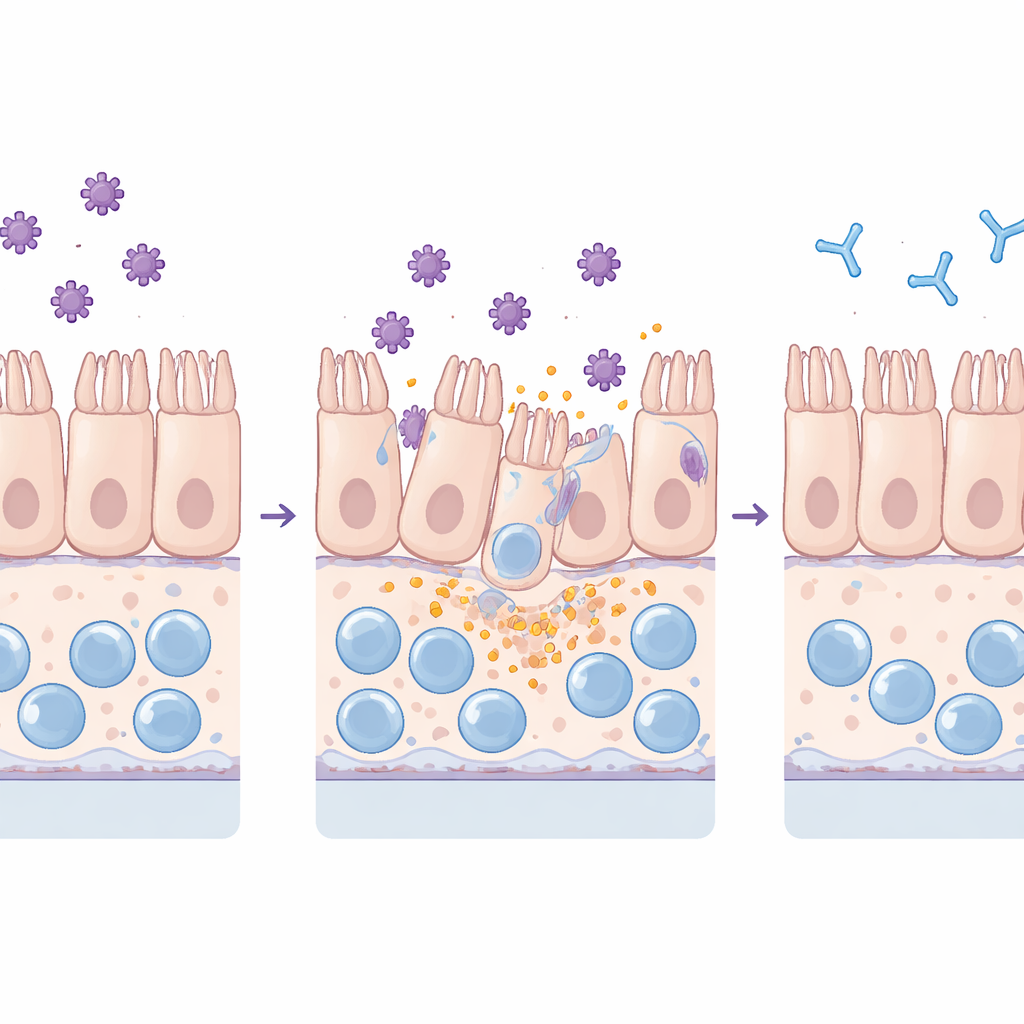
The team exposed both standard flat cultures and the more complex chip to purified TcdA and TcdB. They monitored how well the intestinal barrier kept out fluorescent dye, examined junction proteins that hold neighboring cells together, and measured inflammatory molecules and cell injury markers in the surrounding fluid. Both toxins harmed the tissue, but in distinct ways: TcdA mainly disrupted tight junctions and caused cells to round up, while TcdB led to widespread loss of cells from the layer. Crucially, the chip detected these effects at much lower toxin levels than the flat cultures, reflecting its greater sensitivity and more life‑like behavior under continuous flow.
Immune cells fan the flames—and can be protected
Adding macrophages and neutrophils to the chip amplified the response to toxins. Macrophages boosted the release of key inflammatory messengers such as IL‑6 and IL‑8 but were themselves depleted, consistent with an inflammatory form of cell death. Neutrophils adhered to the vessel wall and migrated into the gut tissue, echoing the immune cell influx seen in patients. Longer toxin exposure damaged not only the intestinal lining but eventually the vessel layer as well. When the researchers pre‑mixed the toxins with toxin‑neutralizing antibody sera, however, structural damage, barrier leakage, cytokine surges, macrophage loss, and neutrophil infiltration were all markedly reduced. In extended experiments, tissues injured by TcdB could partially recover once toxins were removed, whereas TcdA left more persistent damage, hinting at different repair capacities after each toxin.
What this means for patients and therapies
To a lay reader, the key message is that this tiny, living gut model can realistically mimic how C. difficile toxins injure the intestine and stir up the immune system, while also revealing how well antitoxin antibodies can shield the tissue. Compared with simple cell layers, the intestine‑on‑chip is more sensitive, includes blood vessels and immune cells, and can run for days to capture both early damage and attempts at recovery. That makes it a promising test bed for next‑generation treatments and vaccines that neutralize toxins without further disturbing gut microbes, and potentially a tool to tailor therapies using patient‑derived cells in the future.
Citation: Wegner, V.D., Warschinke, M., Brahim, I.B. et al. Modeling Clostridioides difficile toxin pathogenesis and antiserum protection in an immunocompetent intestine-on-chip platform. Sci Rep 16, 9233 (2026). https://doi.org/10.1038/s41598-026-44170-8
Keywords: Clostridioides difficile, intestine-on-chip, gut infection, neutralizing antibodies, organ-on-chip models