Clear Sky Science · en
Absence of the luciferase gene in the genome of the kleptoprotein bioluminescent fish Parapriacanthus ransonneti
Fish That Steal Their Glow
When we think about glowing animals, we usually imagine creatures that make their own light using special genes. This study turns that idea on its head. The small reef fish Parapriacanthus ransonneti shines in the dark, but instead of carrying the genetic recipe for its light-making tool, it appears to borrow the finished protein from the tiny crustaceans it eats. By reading this work, a non-specialist can glimpse how flexible life can be—and how evolution sometimes chooses “use it, don’t make it” as a winning strategy.
Borrowed Light from Tiny Prey
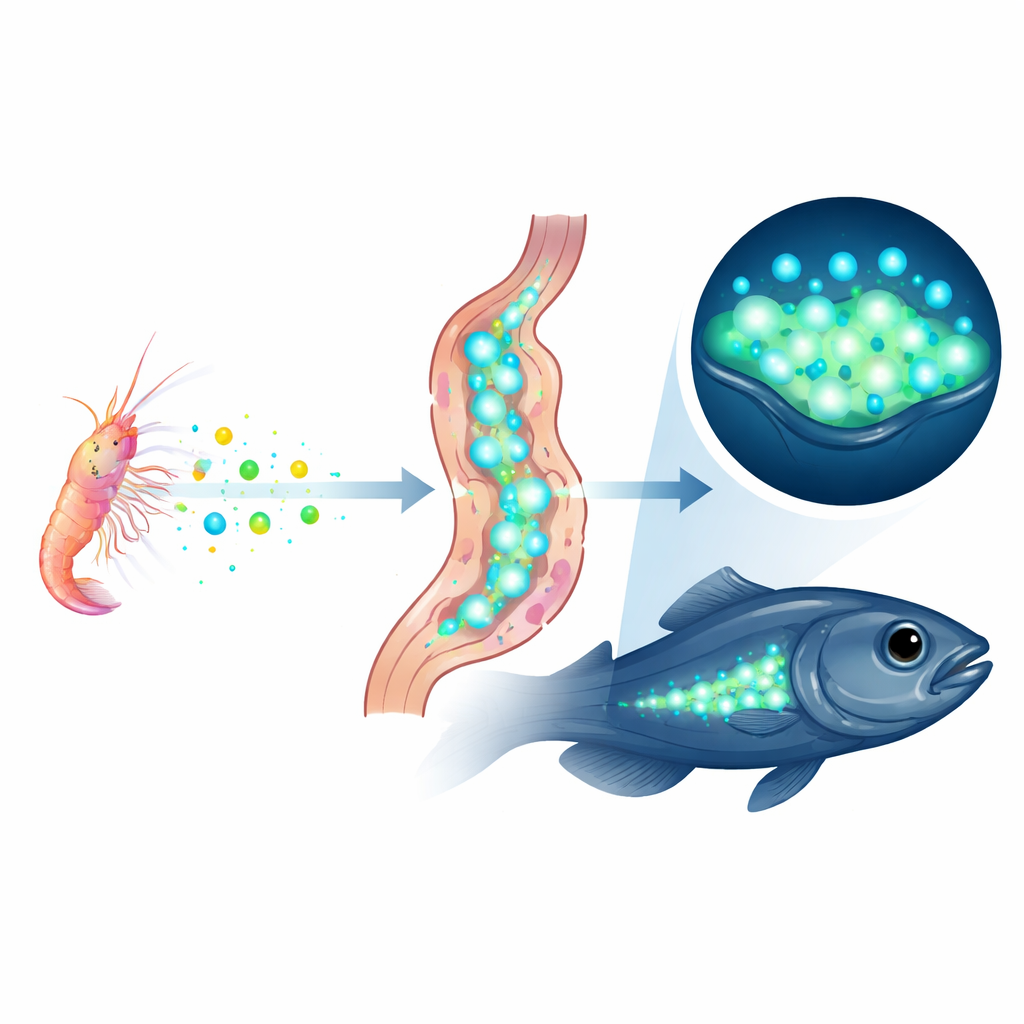
Scientists already knew that this fish glows using the same light-producing protein, called luciferase, as a bioluminescent ostracod, a tiny planktonic crustacean. Earlier work showed that the luciferase protein in the fish is an exact match to the ostracod version, and that the fish lose their glow if they are kept for months without luminous prey. Feeding them a different glowing ostracod swaps the protein in the fish’s light organs for the new one. These clues suggested that the fish do not build luciferase themselves but instead stockpile it from their diet—a process the authors call “kleptoproteinism,” literally protein theft. Still, it was possible that hidden genes for luciferase lurked in the fish genome beyond the reach of previous methods.

Reading the Fish’s Genetic Blueprint
To settle the question, the researchers built a high-quality genetic blueprint, or genome, for Parapriacanthus ransonneti. They carefully raised fish on non-glowing food for over a year to avoid contamination from prey DNA, then extracted and sequenced the fish’s own DNA using state-of-the-art long-read technology. The assembled genome was about 625 million “letters” long, matching independent size estimates and showing very few gaps. They then predicted tens of thousands of genes and checked the overall quality using standard benchmarks, confirming that nearly all expected fish genes were present. In other words, this was a solid, nearly complete map on which a luciferase gene, if it existed, should be visible.
Hunting for a Missing Gene
Armed with this genome, the team went on a focused treasure hunt. They compared known luciferase sequences from several luminous ostracods against the fish’s predicted proteins, its assembled chromosomes, and even the raw, unassembled DNA reads. Multiple search tools with different strengths were used to avoid blind spots. Time and again, no true luciferase match appeared. A few fish genes looked distantly similar, but closer inspection revealed that they belonged to a common family of immune-related proteins, not light-making enzymes, and their evolutionary trees matched ordinary fish history rather than any signal of recent borrowing from crustaceans. The absence of luciferase not only from the polished genome but also from the raw sequencing data makes it extremely unlikely that the gene is simply hiding in an unassembled corner.
Checking for Other Genetic Shortcuts
The scientists also asked whether the fish might have quietly imported other helpful genes from ostracods, such as those that manage the light-producing chemical luciferin. Using fast, large-scale comparison tools, they scanned every predicted fish protein against databases of fish and ostracod proteins and flagged cases where a fish gene resembled an ostracod gene more than those of other fishes. About twenty such candidates emerged, but detailed evolutionary trees showed that these genes still grouped firmly with fish, not within ostracod branches. In short, there was no convincing sign that any genes, including those that process luciferin, had jumped sideways from crustaceans into this fish’s DNA.
A New Way to Shine
Put together, the evidence points to a striking conclusion: Parapriacanthus ransonneti glows without owning the genetic instructions for its key light-making enzyme. Instead, it captures ready-made luciferase proteins from the ostracods it eats and stores them in its light organs, a living example of “you are what you eat” taken to the molecular level. This shows that animals can acquire complex abilities not only by evolving or importing new genes, but also by directly reusing the working parts of other species. The newly assembled genome now offers a platform for future work to uncover how the fish safely transports, protects, and controls these stolen proteins—and how often nature may be using similar tricks in other glowing creatures.
Citation: Bessho-Uehara, M., Yamaguchi, K., Koeda, K. et al. Absence of the luciferase gene in the genome of the kleptoprotein bioluminescent fish Parapriacanthus ransonneti. Sci Rep 16, 9211 (2026). https://doi.org/10.1038/s41598-026-43942-6
Keywords: bioluminescent fish, protein sequestration, kleptobiology, luciferase, genome sequencing