Clear Sky Science · en
Cardiac phenotype in hereditary transthyretin amyloidosis: correlations between fibril types and 99mTc-DPD uptake
Why this heart study matters
Some inherited conditions quietly damage the heart over many years before symptoms appear. One such disease, called hereditary transthyretin amyloidosis, can cause the heart to stiffen and eventually fail. Doctors can now spot this damage using a special bone scan, but the test does not work equally well for every patient. This study explores why, by looking closely at the tiny protein fibers that build up in tissues and how they relate to what doctors see on heart scans and in blood tests.
Two ways a protein can misbehave
In hereditary transthyretin amyloidosis, a blood protein that normally carries hormones and vitamin A can misfold and clump together, forming long, threadlike deposits called amyloid. In Sweden, most affected patients share the same gene change, known as Val30Met, yet they often develop two different patterns of illness. Some people fall ill relatively young, mostly with nerve problems such as numbness and weakness. Others get sick later in life and more often develop heart disease. Earlier work showed that their amyloid fibers differ: one type (called type A) is a mix of whole and cut-up protein pieces, while the other (type B) is made of intact protein only. These structural differences were suspected to influence how strongly the heart lights up on a bone scan that uses a tracer called 99mTc-DPD.
Looking in fat tissue to understand the heart
The researchers studied 152 patients with hereditary transthyretin amyloidosis who had both a 99mTc-DPD heart scan and a small needle biopsy of belly fat. The fat samples, rather than heart tissue, were used to determine amyloid fiber type and to estimate how much amyloid was present. At the same time, the team collected data on heart structure and function, including ultrasound measurements of heart wall thickness and blood tests for markers of heart damage and stress. They then asked how well fiber type and scan results agreed with one another and with the patients’ actual heart health.
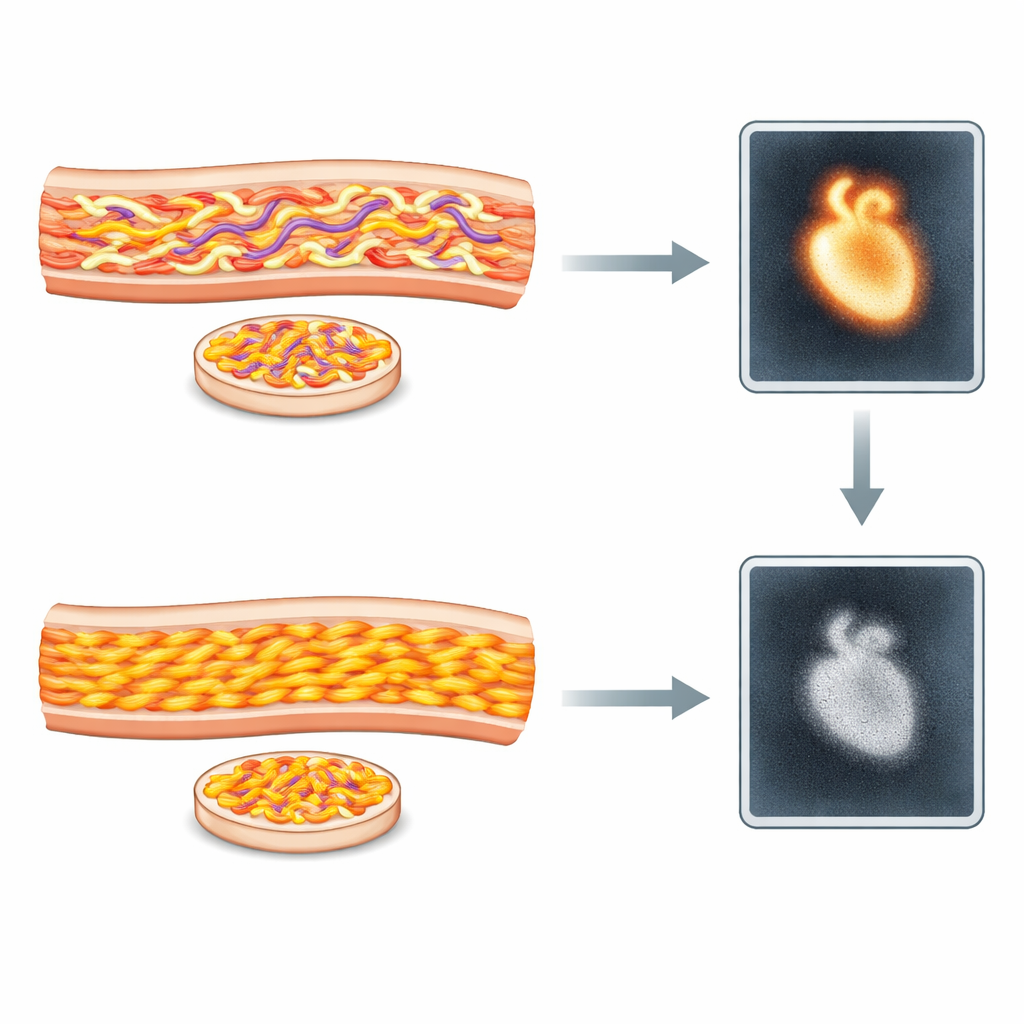
What the scans revealed about heart strain
Patients whose fat tissue contained type A fibers were generally older and had thicker heart walls, higher levels of heart injury markers, and more signs of heart failure than those with type B fibers. Every patient with type A fibers had a clearly abnormal 99mTc-DPD scan, showing strong tracer uptake in the heart. By contrast, most patients with type B fibers (about 85 percent) had normal scans, matching their milder heart involvement. However, a notable minority of type B patients—about 15 percent—did show clear tracer uptake. These individuals looked very similar to the type A group in terms of age and degree of heart damage, suggesting that the scan may reflect how sick the heart is rather than just which kind of fiber is present.
Hidden complexity behind a simple test
The amount of amyloid seen in belly fat tended to be higher in patients with type B fibers, even though their hearts were usually less affected, underlining that fat samples do not always mirror what is happening in the heart. Among patients with positive scans, higher amyloid load in fat was modestly linked to stronger tracer uptake and to one blood marker of heart strain. Still, there were important unknowns. Because it is technically difficult to type fibers directly in heart tissue, the team could not confirm whether the heart always holds the same fiber type as the fat, or whether a mixture of different deposits might be present. The scan itself detects small calcium deposits rather than the protein fibers directly, adding another layer of complexity.
What this means for patients and doctors
This work shows that the microscopic structure of amyloid deposits closely matches the overall disease pattern in hereditary transthyretin amyloidosis. Patients with mixed, fragmented fibers are more likely to have serious heart problems and strong tracer uptake on scans, while those with only intact fibers usually have milder heart involvement and normal scans. At the same time, the study highlights that the 99mTc-DPD scan is often a better guide to actual heart disease than fiber typing alone. Importantly, a normal scan does not fully rule out this inherited condition, especially in patients with the Val30Met variant, meaning doctors must still rely on a combination of genetic testing, tissue sampling, imaging, and blood markers to reach a confident diagnosis.
Citation: Löfbacka, V., Wixner, J., Westermark, P. et al. Cardiac phenotype in hereditary transthyretin amyloidosis: correlations between fibril types and 99mTc-DPD uptake. Sci Rep 16, 9196 (2026). https://doi.org/10.1038/s41598-026-43816-x
Keywords: transthyretin amyloidosis, cardiac amyloidosis, bone scintigraphy, protein fibrils, hereditary heart disease