Clear Sky Science · en
A micro-CT–integrated 3D simulation framework reveals fluid transport mechanisms and void dynamics in root canal biomaterials
Why tiny gaps in root canals matter
Root canal treatments are meant to save damaged teeth by cleaning out infected tissue and sealing the inner space so germs cannot sneak back in. Yet even with modern materials and meticulous technique, many root canals still fail over time. One major suspect is something almost impossible to see during treatment: a hidden network of microscopic gaps and bubbles inside the filling. This study presents a new way to peer into those hidden spaces and virtually watch how fluids might move through them—offering clues to why some teeth stay healthy for years while others become painful again.
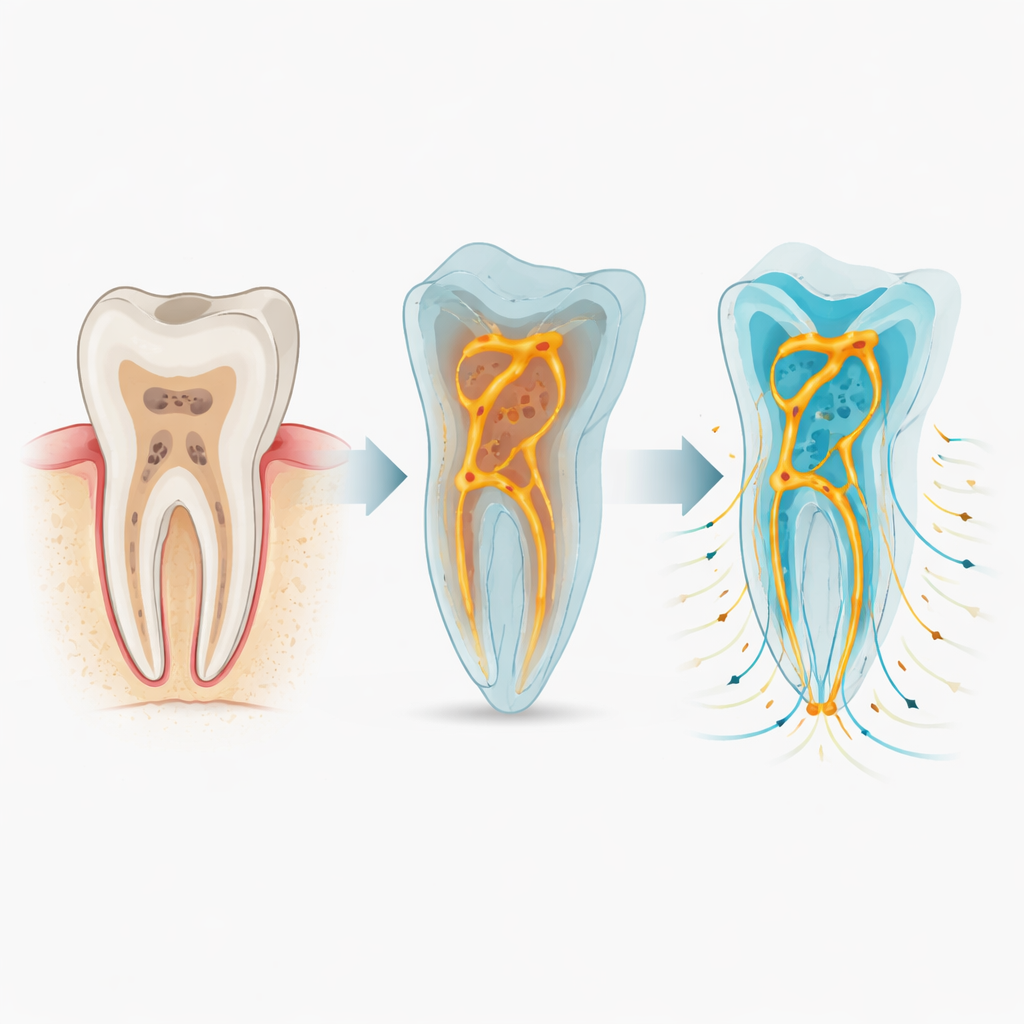
A new way to look inside treated teeth
Traditionally, researchers have tested how well root fillings seal by soaking teeth in dyes, pumping liquids through them, or exposing them to bacteria. These methods can be messy, destructive, and difficult to reproduce. They often give conflicting results, and they reduce a complex three‑dimensional problem to simple end points like how far a dye has travelled. The authors of this paper argue that what really matters is not just how much empty space exists in a filling, but how those spaces are shaped and connected—and how fluids actually move through them over time.
Turning X‑ray scans into virtual flow experiments
To tackle this, the team developed a workflow they call 3D‑SALAM. First, they took extracted human teeth that had been cleaned and filled in a standard way and scanned them with high‑resolution micro‑CT, a kind of 3D X‑ray that can reveal features only a few micrometres across. These scans show the solid tooth and filling material, but they also pick up the tiny voids and channels trapped inside. The researchers then used specialised software to isolate just the empty spaces and convert them into detailed digital models. Finally, they ran computer simulations that mimic how liquids and air would behave inside these miniature mazes under different conditions.
What happens when water and air compete
The virtual experiments showed that gaps inside filled canals are not simple straight leaks; they form tangled, uneven networks, especially near the crown of the tooth. When the voids were assumed to be already filled with water, gentle conditions led to slow but fairly even spreading of a dye, driven mainly by random molecular motion. When pressure was added—similar to what might happen during certain lab tests or under chewing forces—water rushed through the largest channels first, leaving some side pockets to fill much later. In other simulations, the voids started full of air, more like what happens right after placing a filling. Here, how easily the walls “liked” water made a big difference: more water‑friendly surfaces allowed liquid to creep into fine crevices, reaching over 90 percent of the empty volume, while water‑repelling surfaces left stubborn bubbles behind.
Speed, surface, and shape all change the picture
The researchers also explored how fast fluid is pushed into the canal. At very low speeds, capillary action—similar to water climbing up a paper towel—dominated, favouring small passages but sometimes bypassing larger ones. At very high speeds, thicker, viscous flow took over, making the overall filling more uniform and shrinking the amount of trapped air, but also creating steeper local differences in how quickly different regions filled. Between these extremes, there was a sweet spot where water‑friendly surfaces achieved the best balance: most voids filled, and only a small amount of air remained. The size of the gaps themselves mattered too. Teeth with many small voids could be filled effectively at lower speeds, whereas those with larger cavities needed stronger flow to achieve similar coverage.
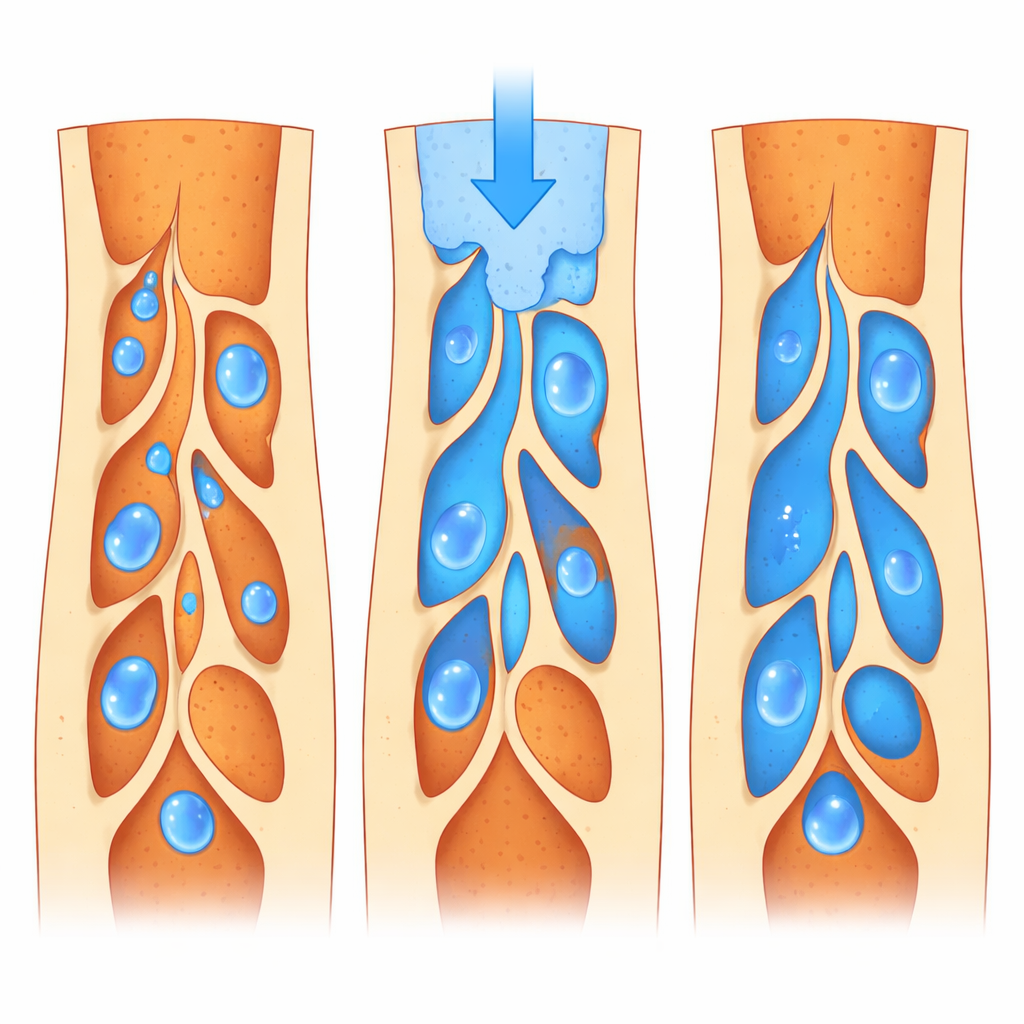
From static pictures to predictive tools
For everyday dentistry, this work does not yet dictate which specific material or technique is best. Instead, it introduces a powerful research tool that turns still images of a filled root canal into a living model of fluid movement. By combining 3D imaging and physics‑based simulation, 3D‑SALAM can show how tiny structural details—void shapes, surface behaviour, and flow conditions—might influence the long‑term seal of a treatment. The same approach could be adapted to other medical materials where fluid and micro‑gaps interact, such as bone scaffolds or dental implants. In essence, the study points toward a future where we can test and refine new biomaterials in a virtual laboratory before they ever reach a patient’s mouth.
Citation: Raoof, A., Raoof, M., Fathi, H. et al. A micro-CT–integrated 3D simulation framework reveals fluid transport mechanisms and void dynamics in root canal biomaterials. Sci Rep 16, 8695 (2026). https://doi.org/10.1038/s41598-026-43796-y
Keywords: root canal sealing, micro-CT imaging, fluid transport, biomaterial porosity, computational simulation