Clear Sky Science · en
Ab initio determination of phase stabilities of dynamically disordered solids: rotational C2 disorder in Li2C2
Why this shifting solid matters
Many modern technologies rely on solids that can quietly change their internal structure when heated or squeezed. These changes, called phase transitions, are central to ideas like solid-state cooling and safer batteries. This study looks at a simple compound, lithium carbide (Li2C2), that switches from a neatly ordered form to a more restless, dynamically disordered form as temperature rises. By watching this transformation atom by atom in computer simulations, the authors show how the internal "restlessness" of tiny molecular units can tip the balance between two crystal structures.
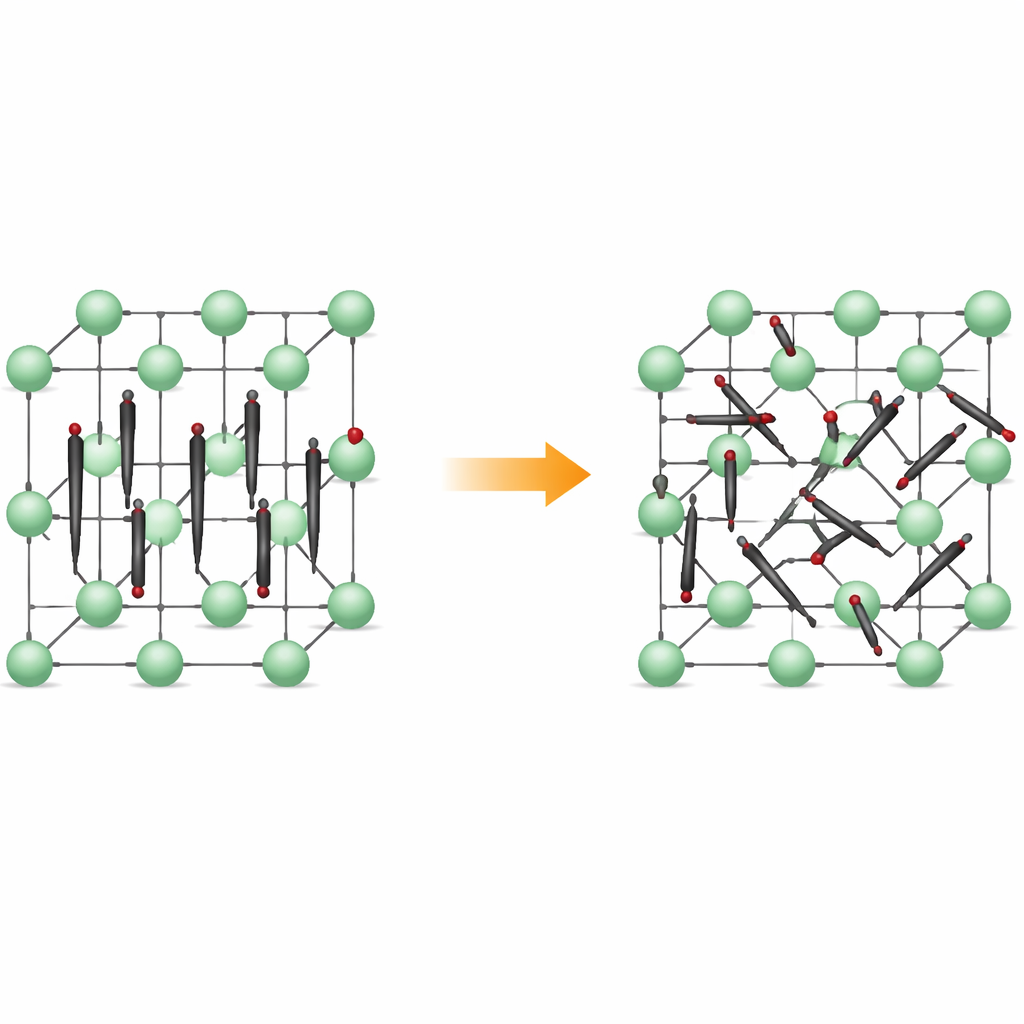
From neat rows to restless motion
At low temperatures, Li2C2 forms an orthorhombic crystal: its carbon atoms pair up into small C2 dimers that all point in nearly the same direction, like aligned matchsticks. Lithium ions sit in between, creating a regular, three-dimensional framework. When the material is heated, it transforms into a cubic form, where the positions of the dimer centers stay ordered on a lattice, but the dimers themselves no longer keep a fixed direction. Instead, they rotate among several preferred orientations, spending time in shallow energy valleys that correspond to specific alignments. The material remains a solid, but its internal structure becomes dynamically disordered.
Following the change along a smooth path
To understand which phase is more stable at a given temperature, one must compare their free energies, which combine energy and entropy (a measure of disorder). Standard methods based on small vibrations around fixed positions struggle when atoms roam or rotate significantly. Here, the authors use a technique called stress–strain thermodynamic integration, built on first-principles molecular dynamics. They construct a smooth deformation path that continuously reshapes the simulation cell from the low-temperature orthorhombic structure to the high-temperature cubic one. Along this path, they run long simulations at fixed temperatures and measure how the internal stress responds to the imposed strain. Integrating this stress response yields the free energy difference between the two phases.
Seeing entropy through atomic motion
The calculations reveal that at around 600 K the low-temperature orthorhombic phase is still slightly favored, while at 650 K the cubic phase wins by a few thousandths of an electron volt per formula unit. Interpolating between these results gives a transition temperature of about 611 K. This is lower than experimental estimates but still in reasonable agreement, given the small free-energy differences involved. The internal energy of the cubic phase is actually higher; what stabilizes it is a large entropy gain, traced directly to the rotational disorder of the C2 dimers. By analyzing how the orientation of each dimer loses memory of its initial direction over time, the authors show that the dimers reorient on sub-picosecond timescales, blurring the line between the usual categories of "vibrational" and "configurational" entropy.
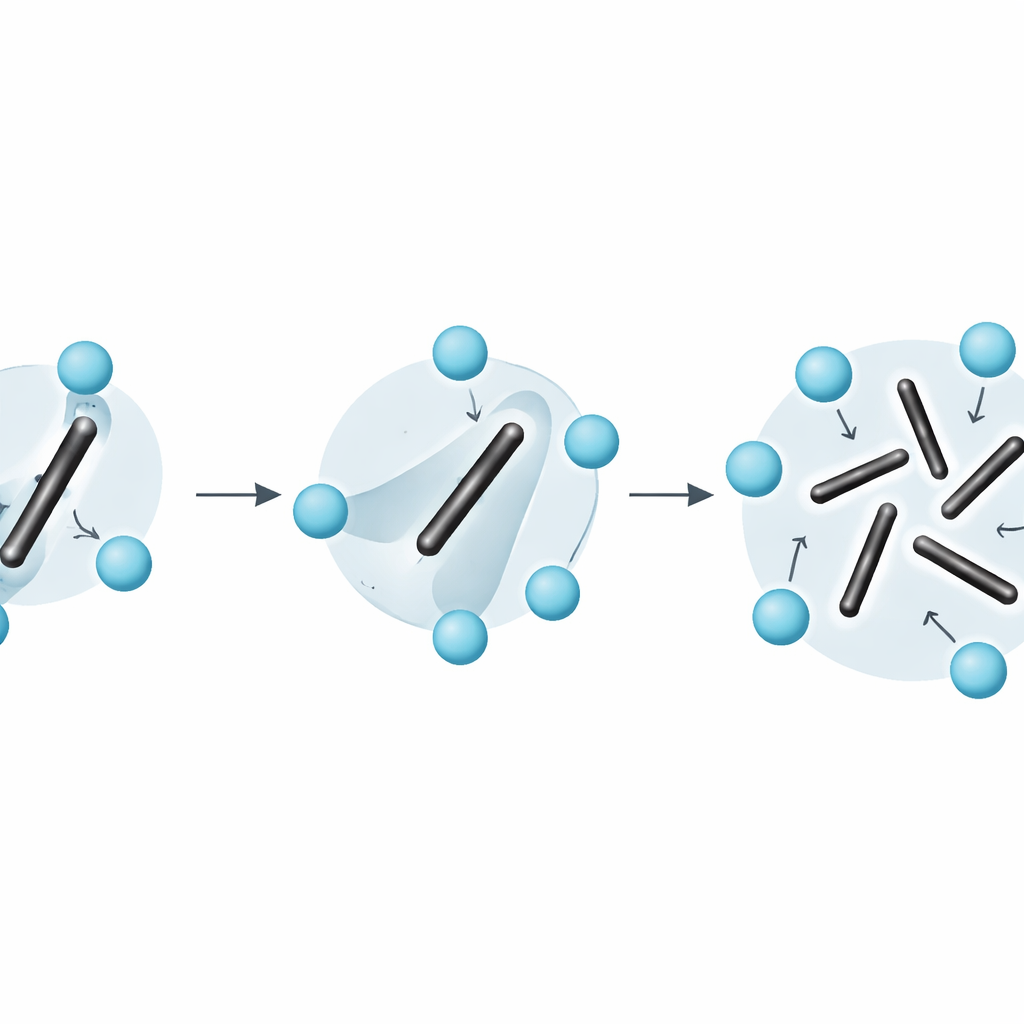
Beyond simple pictures of solid disorder
The work also highlights that common shortcuts—such as treating entropy as a simple sum of vibrations around fixed configurations plus separate counting of static orientations—break down for materials like Li2C2. Because dimer rotations are fast and strongly coupled to ordinary vibrations, the system cannot be cleanly split into separate "vibrating" and "rearranging" parts. The stress–strain integration method sidesteps this difficulty: it extracts the full free energy directly from the microscopic dynamics, without needing to guess how entropy should be partitioned.
What the study teaches us
In everyday terms, the study shows how a solid can stay rigid while its internal building blocks become increasingly free to twist and turn, and how this internal freedom can make a more disordered structure thermodynamically preferred. For Li2C2, the high-temperature cubic phase is stabilized not because it is energetically cheaper, but because it offers many more ways for the C2 dimers to orient and move. By proving that stress–strain thermodynamic integration can capture this subtle balance between order, energy, and entropy, the work opens a path to predicting similar transitions in other dynamically disordered solids that may underpin future cooling devices, batteries, and smart materials.
Citation: Klarbring, J., Filippov, S., Häussermann, U. et al. Ab initio determination of phase stabilities of dynamically disordered solids: rotational C2 disorder in Li2C2. Sci Rep 16, 8965 (2026). https://doi.org/10.1038/s41598-026-43795-z
Keywords: solid-state phase transition, dynamical disorder, molecular dynamics, lithium carbides, thermodynamic integration