Clear Sky Science · en
Elucidating the relationship between affinity and potency in the performance of therapeutic IgE
Turning Allergy Antibodies Against Cancer
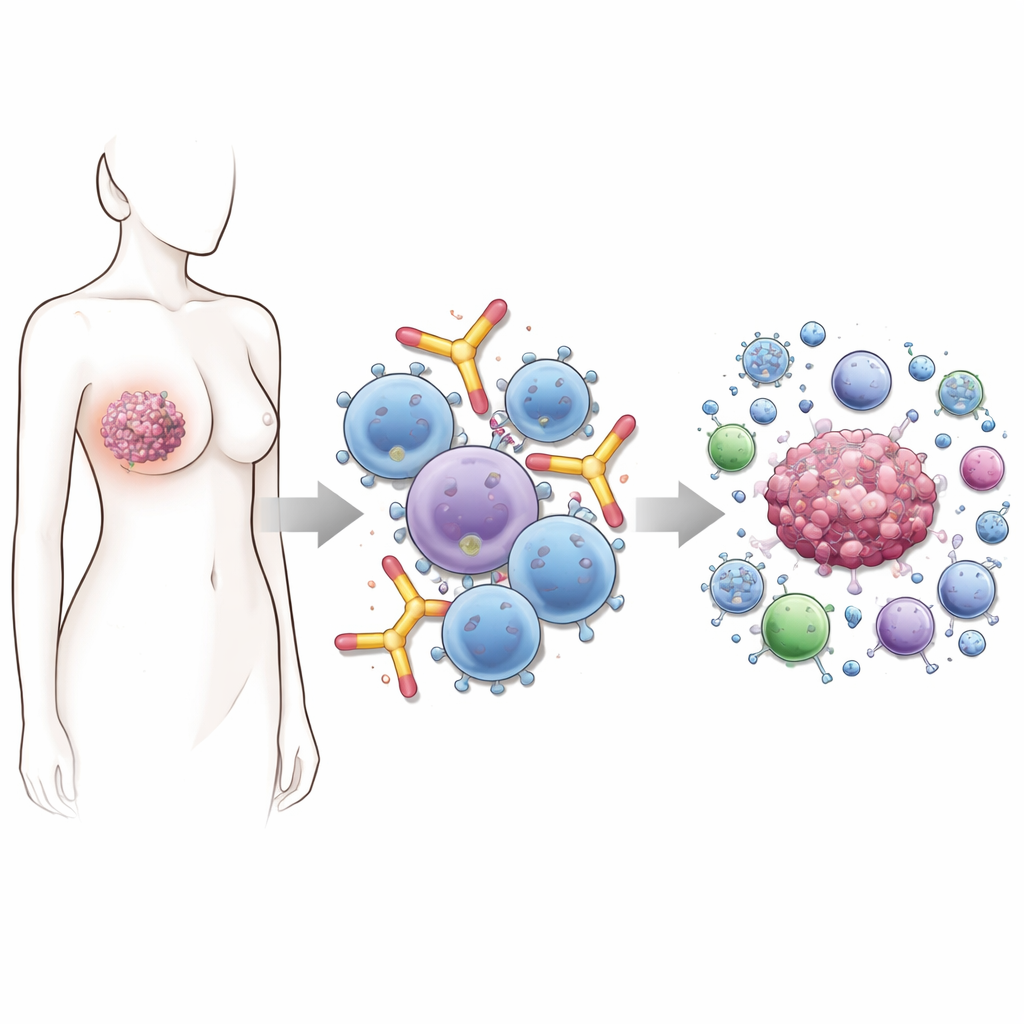
Allergy sufferers know the power of IgE antibodies—the molecules that help trigger itchy eyes and sudden sneezes. This study asks a surprising question: can that same powerful biology be redirected to fight cancer, and if so, how precisely should these antibodies be tuned to work best? The researchers homed in on breast cancers that make a protein called HER2, including cases where HER2 is present at very low levels and current drugs often fail.
Why This Antibody Class Is So Special
Most antibody drugs on the market today belong to the IgG class. They are workhorses of modern cancer therapy, but they have limits—especially when tumors dodge attack by lowering the amount of target protein on their surface. IgE antibodies are different. They naturally latch very tightly onto immune cells such as mast cells, basophils, monocytes, and macrophages, priming them for rapid responses. In allergy, that means explosive reactions to pollen or food. In cancer, the same wiring could mean rapid, focused attacks on tumor cells as soon as IgE encounters its target on the tumor surface.
Designing a Sharper Cancer-Seeking IgE
The team had previously created an IgE antibody, called EPS 226, that recognizes HER2 on breast cancer cells and can slow tumor growth in experimental models. In this new work, they set out to answer a subtle but crucial design question: does making the antibody grip HER2 more tightly actually improve its ability to rally the immune system, or does the natural “many-hands” binding of IgE (called avidity) already do most of the job? To explore this, they used a targeted mutagenesis and selection approach in bacteria to generate millions of small variations of the HER2-binding region and then fished out 12 new IgE variants that bound HER2 more strongly than the original antibody.
Testing Power on Cells and in Animals
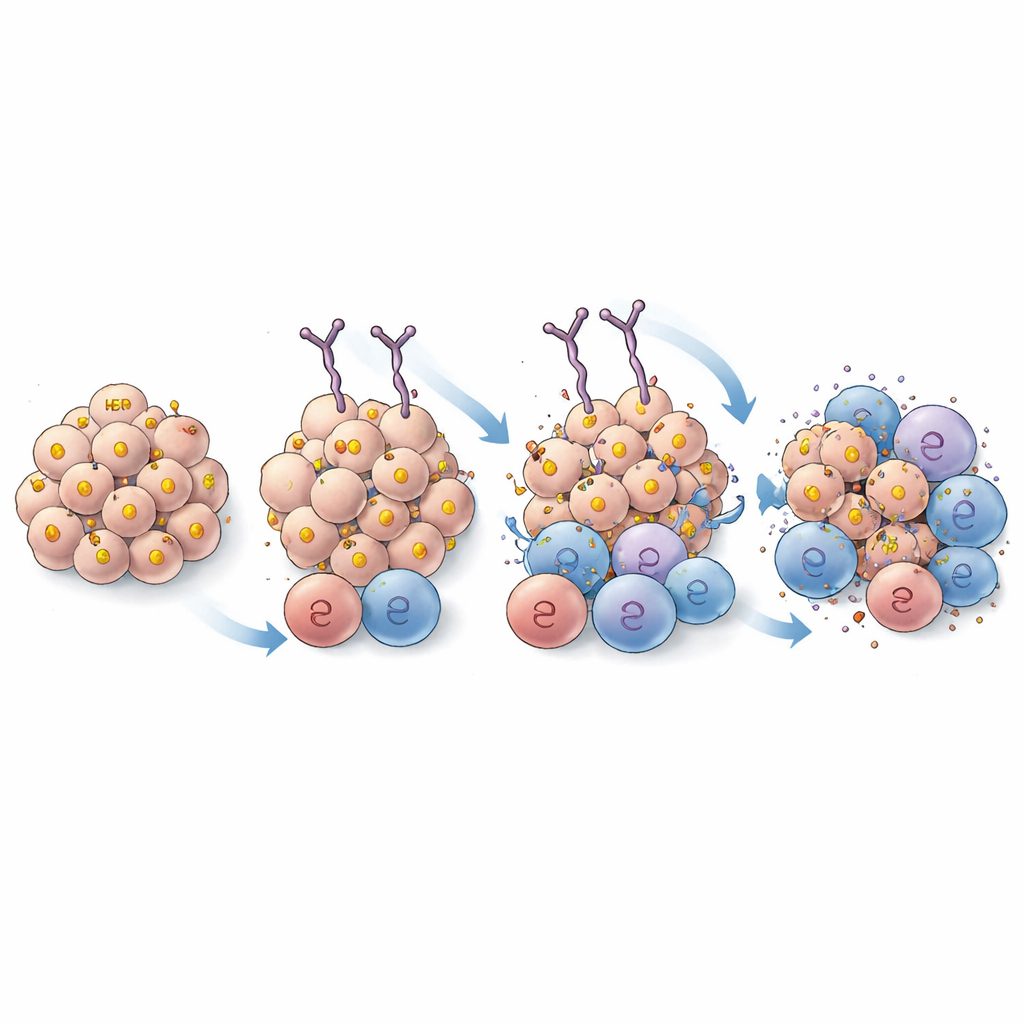
These improved antibodies were then stress-tested in a series of cell-based experiments. When loaded onto mast cells and exposed to HER2-positive cancer cells, most of the tighter-binding IgEs triggered stronger degranulation—an activation response that releases potent immune chemicals. One variant, renamed EPS 232, stood out for its ability to activate cells while still binding effectively to HER2 on human and rat tumor cells. Further tests showed that EPS 232 better armed immune cells to kill cancer: it enhanced cell-killing by basophils and promoted more efficient engulfing of HER2-positive tumor cells by engineered immune cells and by primary human macrophages. Interestingly, increasing binding strength helped only up to a point; variants with extremely high affinity did not outperform EPS 232, suggesting there is a “sweet spot” where grip is strong enough to be effective but not so strong that it hampers how many antibodies and immune cells can engage each tumor cell.
Zooming In on How the Antibody Binds
To ensure that the gains came from stronger grip rather than from recognizing a new patch of HER2, the scientists used high-resolution cryo-electron microscopy to visualize how EPS 232 and the original EPS 226 sit on the HER2 protein. The structures showed that both antibodies dock onto the same region of HER2 and use very similar contact points, with only subtle differences that were not expected to alter the overall binding site. This supports the idea that the boosted performance of EPS 232 arises from improved affinity rather than a change in target.
From Bench to Tumor Control
The real test came in animal models. In mice carrying human breast tumors and supplied with human immune cells, EPS 232 curtailed tumor growth more effectively and at lower doses than EPS 226. In rats with an aggressive breast cancer model that makes ultra-low levels of HER2—similar to some triple-negative breast cancers—an adapted rat version of EPS 232 not only slowed tumor growth more than its predecessor, but also extended survival. Tumors from treated animals showed increased infiltration of T cells and macrophages, indicating that the improved antibody was better at reshaping the tumor environment into one more favorable to immune attack.
What This Means for Future Cancer Therapies
For a general reader, the main message is that fine-tuning how tightly an IgE antibody grabs its cancer target can make a real difference to how well it works, even though IgE already forms many simultaneous contacts with cells. The study identifies EPS 232 as a leading candidate IgE drug against HER2-positive and HER2-low breast cancers, including forms that currently lack good treatment options. More broadly, it shows that “allergy-style” antibodies can be engineered with the right balance of binding strength to turn a hair-trigger system that usually causes sneezing into one that helps the immune system hunt down and eliminate tumors.
Citation: Marano, F., McKenzie, C., Birtley, J.R. et al. Elucidating the relationship between affinity and potency in the performance of therapeutic IgE. Sci Rep 16, 10555 (2026). https://doi.org/10.1038/s41598-026-43772-6
Keywords: therapeutic IgE, HER2 breast cancer, antibody affinity, cancer immunotherapy, triple-negative breast cancer