Clear Sky Science · en
Fish meal replacement with poultry byproduct and black soldier fly larvae proteins: effects on growth, flesh quality, bioactivity, and physiological responses of Nile tilapia
Why feeding farmed fish needs a rethink
Fish like Nile tilapia help feed millions of people, but the feed they eat often relies on fish caught from the ocean and turned into fishmeal. As demand rises and wild fish supplies stagnate, this system strains both wallets and ecosystems. This study explores whether tilapia can grow just as well, and stay just as healthy, when their feed replaces conventional fishmeal with proteins made from poultry by‑products and the larvae of the black soldier fly. The results point to an unexpectedly powerful role for insects in building a more sustainable plate of fish for people.
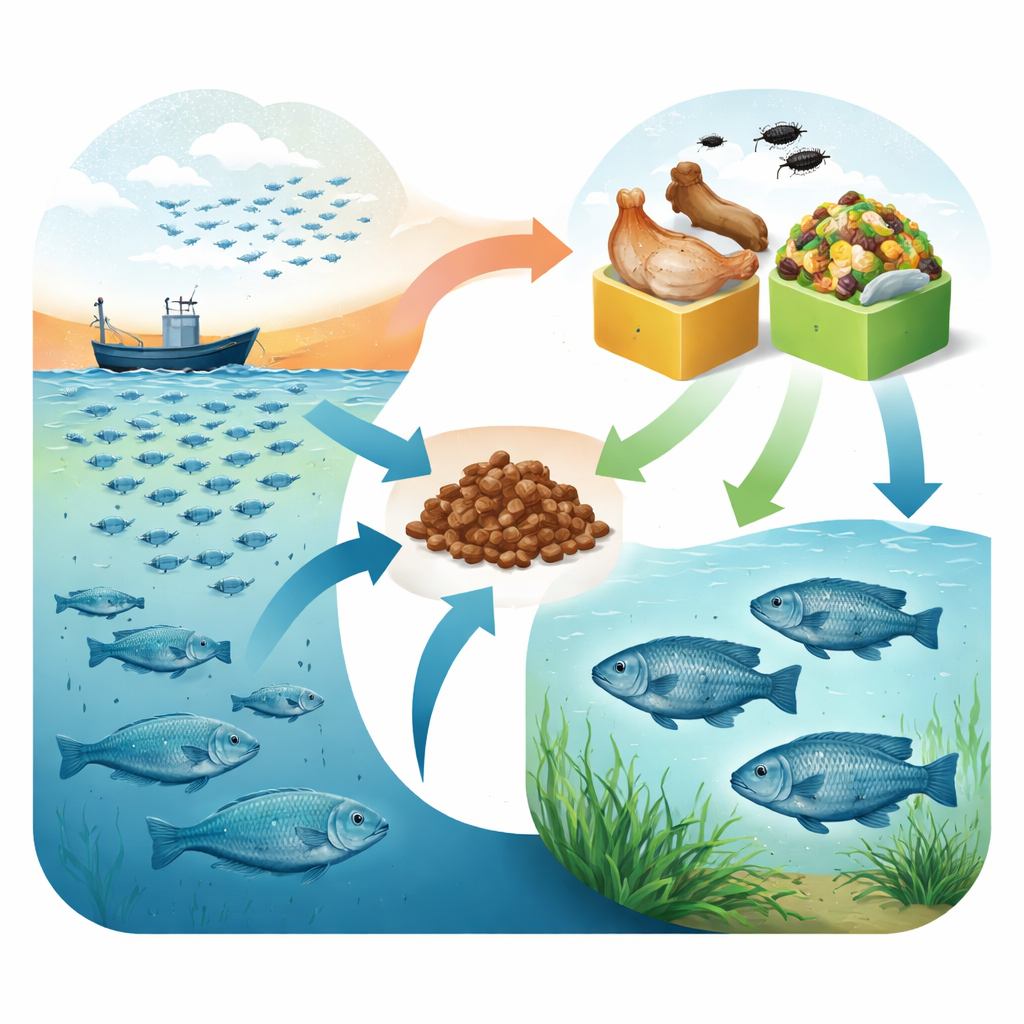
New ways to fuel a common farmed fish
The researchers worked with Nile tilapia fry, a popular farmed species known for its fast growth and flexible diet. They divided 168 young fish into four groups and raised them for ten weeks on different feeds that were otherwise similar in protein and energy. One diet used traditional fishmeal as the main animal protein. A second replaced the fishmeal with poultry processing leftovers, such as skin and organs, rendered into a high‑protein meal. A third swapped fishmeal for powdered black soldier fly larvae, raised on fruit and vegetable scraps. The fourth combined poultry by‑product meal and insect meal in equal parts. This design let the team ask not just whether fish could survive, but how growth, body make‑up, health markers, and flesh quality compared across feeds.
Growth, nutrition, and fish fillets on the plate
At the end of the trial, tilapia fed the insect‑based diet matched the fishmeal group in key measures such as final body weight, weight gain, and feed conversion—the amount of food needed to add a unit of body mass. Fish fed poultry by‑products alone or the mixed diet grew somewhat more slowly and, in the mixed group, survival was modestly lower, likely linked to handling stress rather than the feed itself. When the whole fish were analyzed, all diets produced acceptable levels of protein and fat, though fish fed poultry by‑products tended to carry more body fat. Fillets from every group were microbiologically safe: total bacteria stayed well below international limits, and no harmful E. coli, yeasts, or molds were detected. In fact, tilapia on the insect diet had the lowest counts of coliform bacteria, a sign of cleaner flesh.

Inside the fish: gut, blood, and immune balance
Looking beyond size and fillet quality, the team examined how the alternative feeds affected internal organs and blood chemistry. Measures such as red and white blood cell counts, liver enzymes, kidney waste products, and blood fats stayed within normal ranges across all groups, indicating that the new ingredients did not harm basic physiology. Microscopic views of the intestine, liver, kidney, and spleen showed normal structure in every treatment. Importantly, fish fed insect meal had intestinal surfaces as large as those in fishmeal‑fed fish, supporting efficient digestion and absorption. In the liver, a signaling protein called NF‑κB, often switched on during inflammation, remained quiet in all groups, suggesting that none of the diets triggered harmful inflammatory stress.
Natural defenses and antioxidant protection
One striking advantage of the insect diet emerged in the fish’s internal chemistry. The black soldier fly feed contained the highest level of natural phenolic compounds—plant‑derived molecules and related substances known for antioxidant activity. This pattern carried through into the fish muscle: tilapia eating insect meal accumulated more phenolics and showed stronger capacity to neutralize damaging free radicals. They also had the lowest levels of malondialdehyde, a by‑product of fat oxidation that signals rancidity and tissue damage. At the same time, their livers showed reduced amounts of the pro‑inflammatory signal TNF‑α and higher levels of IL‑10, which helps dial down inflammation. Together, these changes paint a picture of fish whose immune systems are balanced—ready to defend against disease without being locked in costly, chronic inflammation.
What this means for future fish farming
For consumers, the message is that tilapia can be raised on feeds made from insects without sacrificing growth, health, or fillet safety—and in some respects, such as antioxidant status and immune balance, the fish may even fare better. For farmers and policy makers, black soldier fly larvae meal stands out as a promising, cost‑effective way to reduce reliance on ocean‑caught fishmeal while turning low‑value food waste into high‑value protein. Poultry by‑product meal can also contribute, though it may need more careful balancing. As aquaculture expands to feed a growing population, this work suggests that the future of farmed fish may depend less on what we take from the sea and more on how cleverly we recycle what we already have on land.
Citation: Aref, S.M., Alian, H.A., Khodary, F.M. et al. Fish meal replacement with poultry byproduct and black soldier fly larvae proteins: effects on growth, flesh quality, bioactivity, and physiological responses of Nile tilapia. Sci Rep 16, 9536 (2026). https://doi.org/10.1038/s41598-026-43600-x
Keywords: Nile tilapia, insect protein, black soldier fly larvae, poultry by-product meal, sustainable aquafeed