Clear Sky Science · en
Natural genetic variation impacts complement inhibitory activity of PFam54 orthologs of Asian Borrelia bavariensis
Why this matters for Lyme disease
Lyme disease is already the most common tick-borne illness in the Northern Hemisphere, but not all Lyme-causing bacteria behave the same way in our bodies. This study looks at Borrelia bavariensis, a close relative of the main Lyme agent, and asks a simple question with big implications: do natural genetic differences in Asian and European strains change how well these bacteria dodge one of our body’s first lines of defense, the complement system in the blood? The answer helps explain how these microbes spread, how they cause disease, and where we might target future treatments or vaccines.
A tale of two continents
Borrelia bavariensis circulates between small animals and ticks across Eurasia and can cause Lyme disease in humans. European strains often show a strong preference for the nervous system, turning up in cases of neuroborreliosis, while Asian strains seem less tied to brain and nerve infections. Genetic studies suggest European strains descended from a more diverse Asian ancestor and passed through a bottleneck when they adapted to a different tick species in Europe. That evolutionary detour may have reshaped parts of the bacterial genome, including a cluster of genes called PFam54 on a plasmid named lp54. Many PFam54 genes encode surface proteins that help the bacteria survive in blood by interfering with complement, a protein network that can punch holes in invading cells.
How the bacteria dodge the blood’s attack
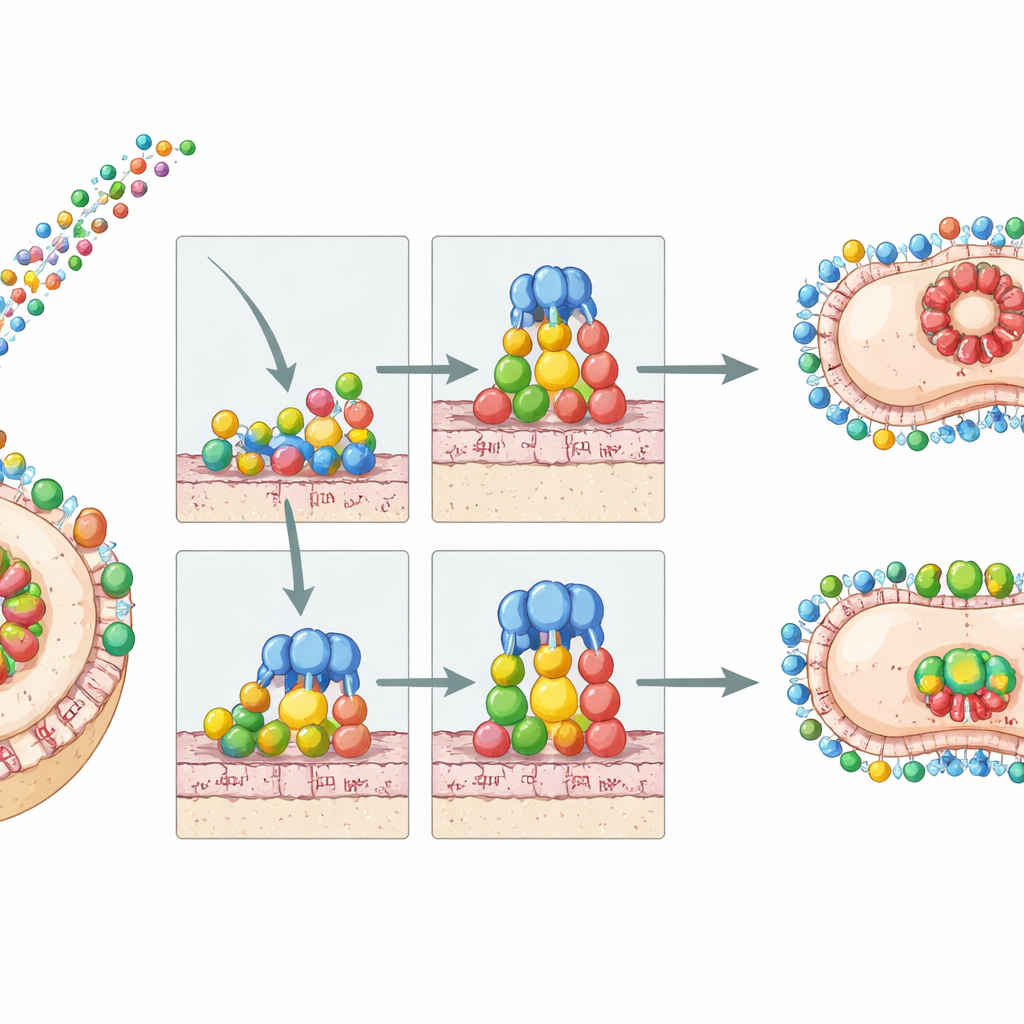
The complement system can be imagined as a molecular alarm that, once triggered, ends in formation of a “membrane attack complex” that drills pores into bacterial membranes. Earlier work in a European reference strain showed that two PFam54 proteins, BGA66 and BGA71, sit on the bacterial surface and latch onto late complement components (C7, C8, C9), blocking assembly of this terminal pore. However, Asian B. bavariensis strains often carry a different assortment of PFam54 genes, and some even lack the exact BGA66 and BGA71 genes found in Europe. The researchers asked whether these Asian variants still work as complement shields, and whether new, Asia‑specific PFam54 family members might provide alternative protection.
Probing diversity with genetics and structure
The team first scanned PFam54 genes across dozens of Asian and European B. bavariensis genomes. As expected, Asian strains showed more sequence diversity in this family, but the overall patterns suggested the proteins remain under pressure to keep their basic function. Using AlphaFold to predict 3D structures, they found that the Asian versions of BGA66 and BGA71, as well as several related proteins, all folded into very similar all-helical shapes despite many amino acid changes. These mutations tended to cluster on surface-exposed regions—exactly where interactions with complement would occur—hinting that fine-tuning of binding, rather than wholesale loss of function, might have evolved.
Putting the proteins to the test
To see what these differences meant in practice, the researchers expressed Asian PFam54 proteins in bacteria and tested them in human serum. Two Japanese B. bavariensis isolates, NT24 and JHM1114, proved highly resistant to killing by active human complement, much like the European type strain. Purified proteins from these strains were then mixed with human complement components in controlled assays. Asian BGA66 could still block formation of the membrane attack complex, though less efficiently than its European counterpart. An Asian BGA71 variant partially impaired a late step in the pathway but needed higher doses to show an effect. Strikingly, a novel Asian-only protein, BGA67b, strongly inhibited the terminal pathway without directly blocking C9 polymerization, indicating a different way of stopping pore assembly. When these PFam54 proteins were produced on the surface of an otherwise complement-sensitive surrogate strain, they made that strain serum resistant, confirming their protective role.
What this means for patients and future research
In everyday terms, this work shows that both Asian and European B. bavariensis strains come equipped with effective molecular “shields” that protect them from being blown apart by the blood’s complement system. The specific shield proteins and their efficiencies differ, but the overall strategy—interfering late in the attack sequence to prevent pore formation—is preserved across continents. That means complement evasion by PFam54 proteins alone does not explain why European strains are more often linked to nervous system disease. Other factors, such as additional surface proteins, tissue targeting mechanisms, or differences in host or tick ecology, must be involved. At the same time, the study highlights a small set of conserved protein shapes and interaction surfaces that are crucial for bacterial survival in blood, marking them as promising targets for drugs or vaccines designed to unmask the bacteria to our innate immune defenses.
Citation: Langhoff, L., Kapfer, P., Röttgerding, F. et al. Natural genetic variation impacts complement inhibitory activity of PFam54 orthologs of Asian Borrelia bavariensis. Sci Rep 16, 9080 (2026). https://doi.org/10.1038/s41598-026-43598-2
Keywords: Lyme disease, Borrelia bavariensis, complement system, immune evasion, PFam54 proteins