Clear Sky Science · en
Prescription‑dose stratification improves deep learning‑based VMAT dose prediction in locally advanced NSCLC
Why Smarter Radiation Plans Matter
When doctors treat advanced lung cancer with radiation, they walk a tightrope: deliver enough energy to control the tumor while keeping delicate nearby organs safe. Designing these plans is slow, complex, and depends heavily on the planner’s skill. This study explores how artificial intelligence can predict the pattern of radiation in the chest and, crucially, shows that teaching the AI with carefully grouped dose levels makes its predictions more accurate and clinically useful.
How Lung Cancer Is Treated with Focused Beams
Most people with locally advanced non-small cell lung cancer cannot have their tumors removed by surgery. Instead, they often receive chemotherapy together with highly shaped radiation beams that sweep around the chest in arcs, a technique known as VMAT. The goal is to bathe the tumor in a high, uniform dose while limiting exposure to the lungs, heart, esophagus, and spinal cord. Achieving this balance usually requires multiple rounds of trial-and-error adjustments, which consume time and can delay treatment decisions. If doctors could quickly estimate what dose pattern is realistically achievable for a given patient, they could choose the best treatment strategy earlier.
Teaching an Algorithm to See Dose Patterns
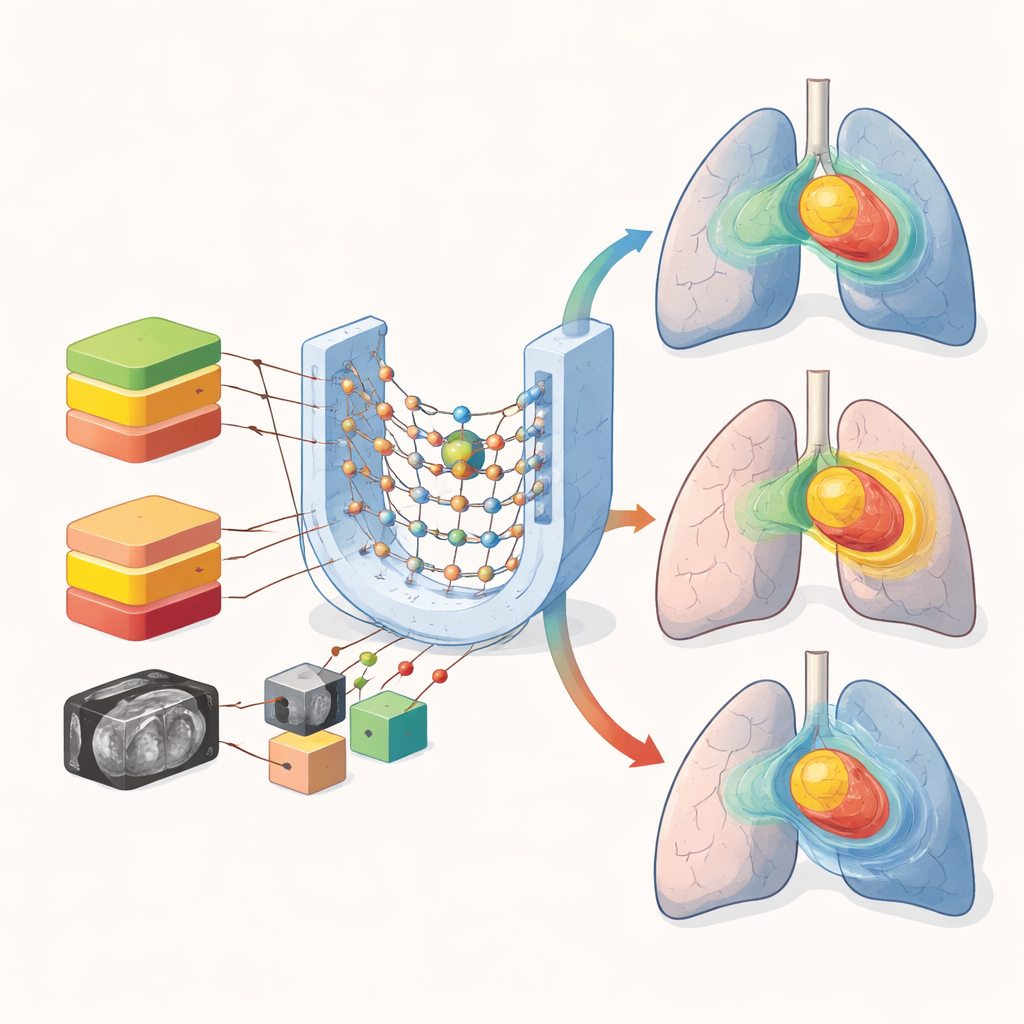
The researchers turned to deep learning, a form of AI that learns patterns directly from images. They collected CT scans and finished treatment plans from 72 lung cancer patients treated with VMAT. For each person, they recalculated how the same beam setup would deliver three different total doses: 50, 54, and 60 gray (Gy), the standard units for radiation. Each case was converted into a three-dimensional grid that marked the tumor, key organs, and the underlying CT image. These nine channels of information were fed into a 3D U-Net, a common neural network design that can learn how dose tends to flow through the body based on anatomy.
Why Grouping by Prescription Dose Matters
A central question was how to handle the fact that patients are prescribed different total doses. The team built four otherwise identical AI models. Three were “single-prescription” models, each trained only on plans with one dose level (50, 54, or 60 Gy). The fourth model mixed 50 and 60 Gy cases together without telling the network which prescription it was seeing. When tested on new patients, the prescription-specific models did a better job matching the true dose in the tumor, keeping both coverage and hot spots close to the clinical plans, typically within a few Gy. In contrast, the mixed model made noticeably larger mistakes in the tumor’s highest-dose regions and around the spinal cord, where the dose must stay below strict safety limits.
Protecting Vital Organs While Speeding Planning
Interestingly, for many organs such as the lungs, heart, and esophagus, the mixed and stratified models performed similarly. Average dose errors to these larger structures remained modest across all approaches. The main weakness of mixing prescriptions emerged in small, sensitive structures like the spinal cord, where steep dose falloff is essential. Visual checks of the predicted dose maps showed that all models captured the overall shape of the high-dose region in the tumor, but mixing prescriptions led to more blurring in sharp gradients and low-dose areas of the lungs. These differences, though subtle numerically, can matter when doctors are deciding whether a higher prescription is safe.
What This Means for Patients and Clinics
For patients, this work does not replace the careful dose calculations already done by clinical systems, but it could help ensure that time and effort are focused on plans that are truly feasible and safe. By showing that grouping training data by prescription dose significantly sharpens AI predictions—especially for tumor coverage and spinal cord protection—the study highlights a practical design choice that can make computer assistance more trustworthy. With further testing in larger and more diverse patient groups, such models could become everyday tools that give doctors a fast, anatomy-based preview of likely radiation trade-offs, supporting better, more consistent treatment planning.
Citation: Chaipanya, T., Nimjaroen, K., Chamchod, S. et al. Prescription‑dose stratification improves deep learning‑based VMAT dose prediction in locally advanced NSCLC. Sci Rep 16, 8707 (2026). https://doi.org/10.1038/s41598-026-43192-6
Keywords: lung cancer radiotherapy, deep learning dose prediction, VMAT planning, treatment plan optimization, organs at risk