Clear Sky Science · en
Unlocking the reactivity of municipal solid waste incineration bottom ash through physicochemical co-activation toward improved cementitious performance
Turning Trash into Stronger Buildings
Modern cities burn huge amounts of household garbage to recover energy, but this process leaves behind a gritty residue called bottom ash. Much of this ash is landfilled, wasting space and materials. The study described here explores how that ash could instead be upgraded into a useful ingredient for concrete, helping to cut both waste and the carbon footprint of construction.

The Hidden Potential in Burned Waste
When household trash is incinerated, the heavier leftovers collect at the base of the furnace as bottom ash. This material is a chaotic mix of grains and fragments that contain many of the same basic elements found in cement, such as calcium, silicon, and aluminum. On paper, that makes it a promising candidate to replace part of the cement in concrete. In practice, however, untreated bottom ash is bulky, chemically unstable, and sprinkled with problematic components like metallic aluminum and salts. These can create gas bubbles, extra pores, and even cracking inside concrete, weakening structures and raising concerns about long-term safety and pollution.
Why Simple Fixes Are Not Enough
Researchers have tried two main types of fixes. One is purely physical: grinding the ash into finer powder so it packs better and exposes more surface for reaction. The other is purely chemical: soaking or treating it with alkaline solutions to wash out harmful substances and tweak its surface chemistry. Each method brings some improvement, but neither alone reliably makes bottom ash a strong, consistent performer when blended into cement. Coarse, glassy grains remain stubbornly unreactive, while leftover metals and salts can still generate gas and leave behind a porous, fragile structure.
A Two-Step Makeover for Ash Particles
The team behind this study proposed a combined route they call physicochemical co-activation. First, the ash is milled in a rotating drum, breaking down clumps, shrinking particles, and creating a network of microcracks. This exposes fresh surfaces rich in reactive components that were previously shielded. Next, the milled ash is soaked for a day in a mildly concentrated solution of calcium hydroxide, a common, inexpensive alkaline chemical. During this bath, some surface layers dissolve away, trapped silicon and aluminum species are released into the liquid, and extra calcium ions attach to the newly opened surfaces. After rinsing and drying, this pretreated ash replaces 30 percent of the cement in standard mortar mixes.
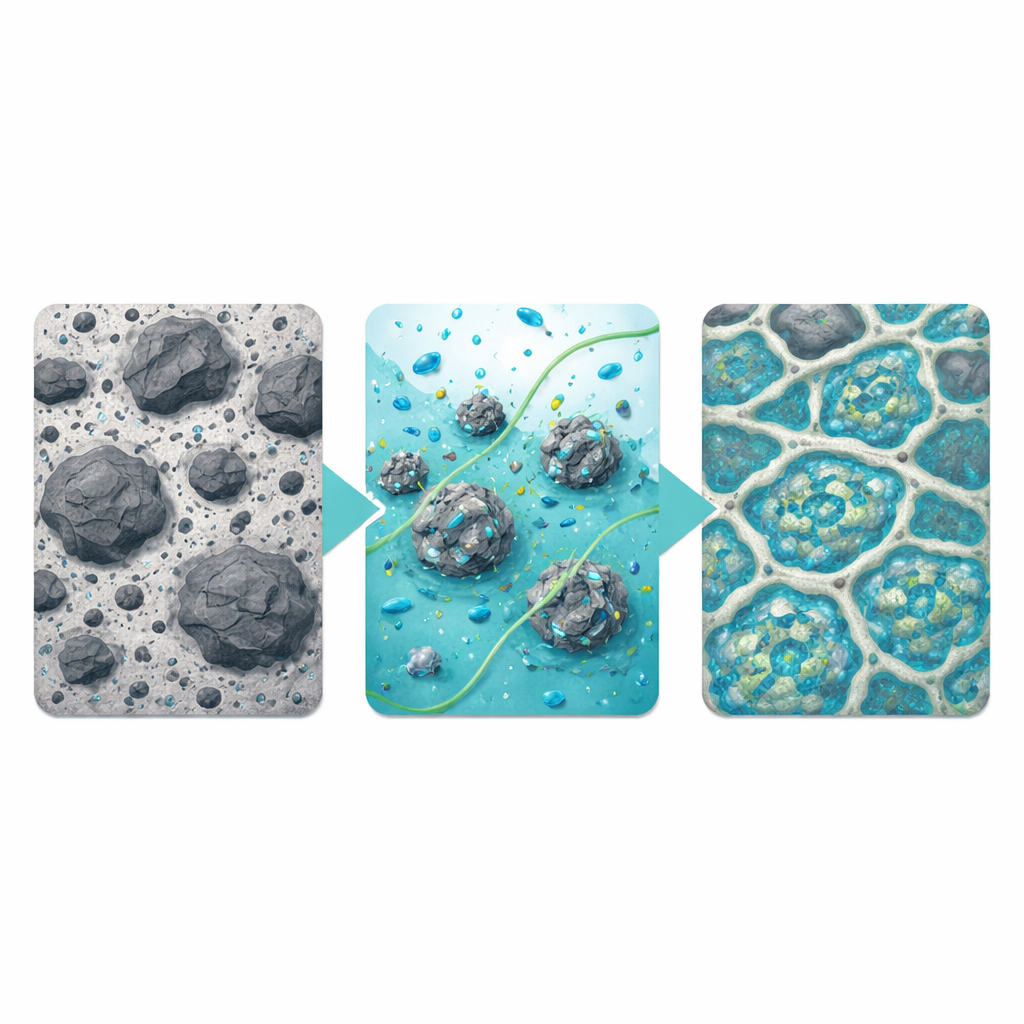
Watching Heat, Strength, and Pores Change
To see whether the two-step treatment truly helps, the researchers monitored how much heat the mixtures gave off as they hardened, how strong they became over time, and what their internal structure looked like. Heat measurements showed that grinding alone sped up the early reactions, while chemical soaking alone shifted the timing but did not fully restore activity. When the two steps were combined and the soaking solution was tuned to a moderate strength, the mixture showed a strong, well-timed burst of reaction. After 28 days, the co-activated ash produced mortars whose compressive strength surpassed those made with only ground ash, coming close to the performance of plain cement mortars while using significantly less cement.
Inside the Concrete: From Voids to Dense Skeleton
Microscopes and X-ray scans revealed why the performance improved. In mixes made with only ground or only soaked ash, the hardened material still contained scattered voids, microcracks, and poorly bonded interfaces where ash grains met the cement paste. In contrast, the co-activated ash led to a tightly knit, honeycomb-like framework in which fine reaction products filled gaps and wrapped ash particles in a continuous gel. Measurements of pores across many length scales showed that this treatment reduced overall porosity and shifted the pore system toward much finer, more evenly distributed pores. The researchers also found that if the soaking solution was made too strong, excess crystals formed on particle surfaces, blocking further reaction and leaving behind larger pores that hurt strength.
What This Means for Greener Construction
In plain language, the study shows that giving bottom ash a carefully tuned “double treatment” can turn it from a troublesome waste into a reliable helper in concrete. By combining short mechanical grinding with a mild alkaline soak, and avoiding overly strong solutions, the ash transforms into a fine, reactive powder that helps build a dense, durable cement matrix. This approach uses existing industrial equipment and inexpensive chemicals, suggesting it could be scaled up at waste-to-energy plants and concrete factories. If widely adopted, such treatment could reduce the need for fresh cement, cut greenhouse gas emissions, and divert large volumes of incinerator ash from landfills into long-lasting buildings and infrastructure.
Citation: Zhu, Z., Zhang, Y., Yang, J. et al. Unlocking the reactivity of municipal solid waste incineration bottom ash through physicochemical co-activation toward improved cementitious performance. Sci Rep 16, 9692 (2026). https://doi.org/10.1038/s41598-026-43059-w
Keywords: waste-to-resource, bottom ash concrete, supplementary cementitious materials, low-carbon construction, cement microstructure