Clear Sky Science · en
Dietary patterns influence the in silico GABA production capacity of Bifidobacterium adolescentis HD17T2H and other human gut bacteria
Why Your Dinner Might Matter to Your Mood
Many people have heard that the gut is like a “second brain,” but it is still unclear how what we eat shapes that hidden world of microbes and, in turn, our mental and digestive health. This study explores one key messenger, gamma-aminobutyric acid (GABA), a chemical that calms nerve activity and is made not only in the brain but also by gut bacteria. The researchers asked a deceptively simple question: if you change your diet, how does that alter the ability of gut microbes to make GABA?
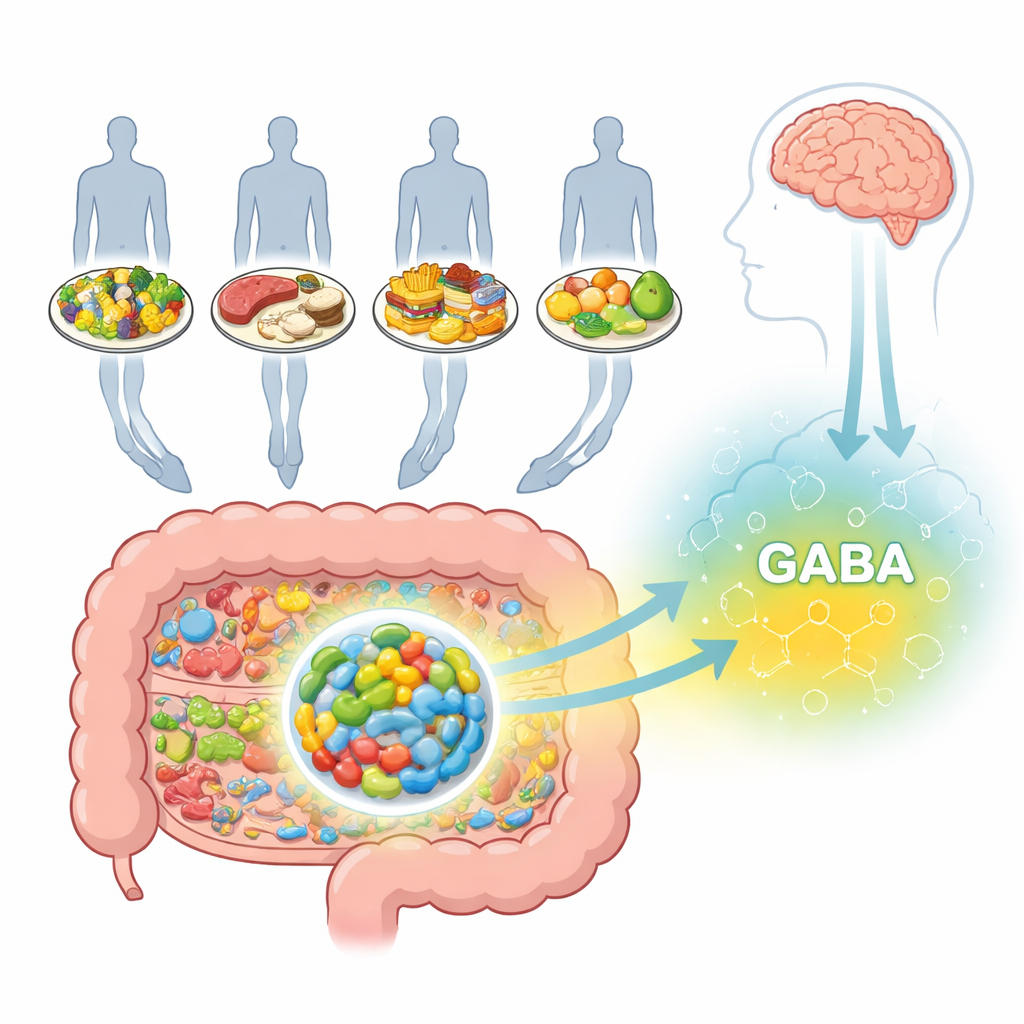
A Calming Chemical Made in the Gut
GABA is best known as a soothing signal in the brain, where it helps keep nerve cells from becoming overactive and has been linked to anxiety and depression. But GABA is also active in the gut itself, where it influences movement of the intestine, fluid secretion, and the release of hormones. Some gut bacteria can turn common nutrients into GABA using well-characterized biochemical routes. One such microbe, Bifidobacterium adolescentis HD17T2H, is a standout GABA producer and belongs to a group of bacteria often found in probiotic products. Yet until now, scientists did not have a systematic picture of how different eating patterns might change this bacterium’s capacity to make GABA.
Using Computer Models as a Virtual Gut
Instead of running dozens of laborious lab experiments, the team built a detailed computer model of B. adolescentis based on its genome. They then “fed” this virtual microbe eleven realistic whole-day diets drawn from a nutrition database, including vegetarian, vegan, high-protein, average European, Mediterranean, low-carbohydrate, and ketogenic patterns. With mathematical tools that estimate how nutrients flow through metabolism, the researchers calculated how much GABA the bacterium could in principle secrete under each diet, while still growing. They also ran thousands of virtual “supplementation” tests, adding extra amounts of single nutrients in silico to see which ones most strongly boosted GABA output.
How Different Diets Shape GABA Potential
The simulations revealed striking differences between eating patterns. Under baseline conditions, a vegetarian diet gave the highest GABA production potential for this bacterium, closely followed by a high-protein diet and a diet designed for type 2 diabetes. At the other end of the spectrum, a ketogenic, very low-carbohydrate plan yielded the lowest baseline GABA output, with a vegan diet also on the lower side. When the team artificially increased the supply of individual compounds, two broad nutrient classes consistently pushed GABA production upward: carbohydrates and nitrogen-rich molecules, especially amino acids. In low-carbohydrate diets, adding sugar-like compounds had the biggest effect, suggesting that carbon energy was the limiting factor. In contrast, in carbohydrate-rich diets such as vegetarian and vegan patterns, extra amino acids and related nitrogen sources had the strongest impact, indicating that nitrogen then became the bottleneck.
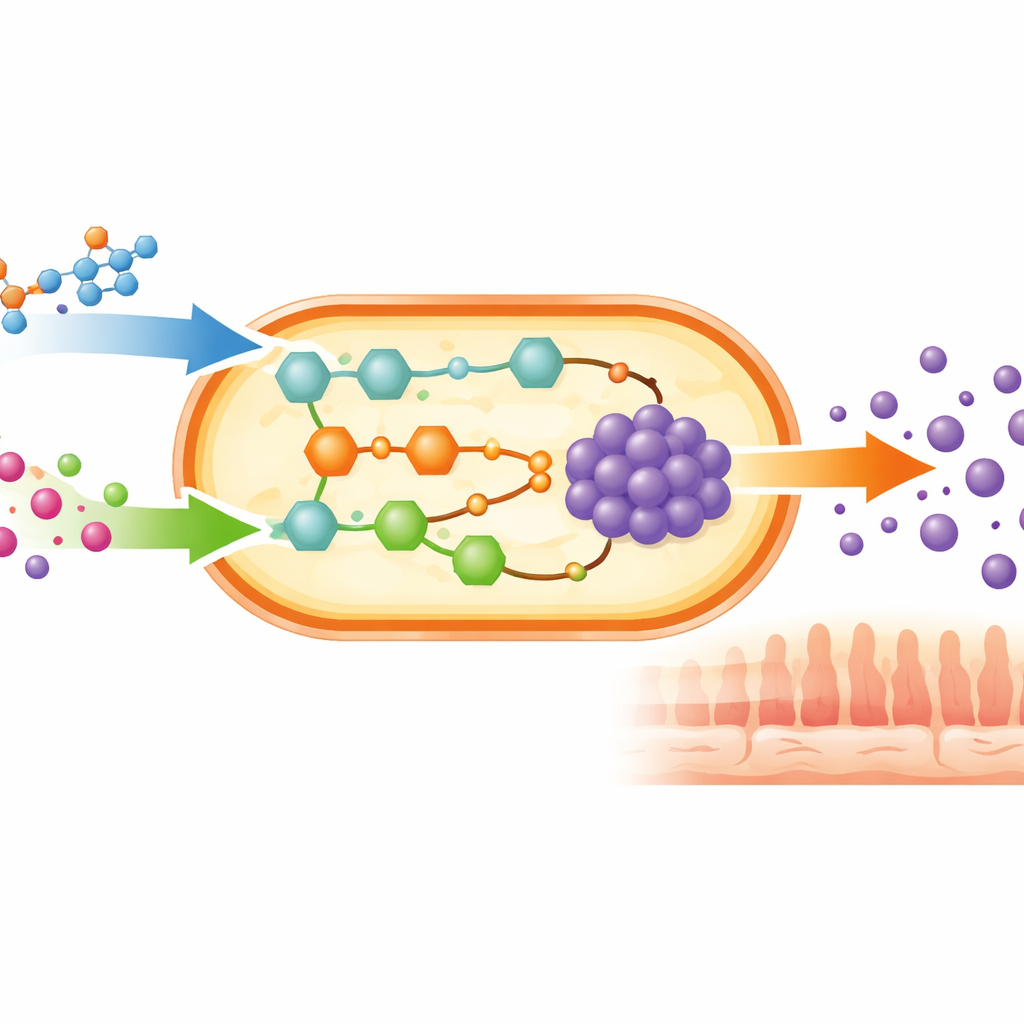
Looking Beyond One Bacterium
To see how these findings might play out in real people, the researchers used detailed dietary records from more than a thousand adults in the Kiel cohort. For each person, they computed how much GABA their personal B. adolescentis population could theoretically produce from their reported nutrient intake. A machine-learning approach confirmed that amino acids were by far the most important dietary drivers, followed by other nitrogen-containing compounds, certain sugars, fatty acids, and B vitamins that help enzymes run efficiently. The team then extended their modelling to whole microbial communities in the same cohort, using a large library of gut microbe models. They found 87 bacterial strains across 47 genera capable of making GABA, including both friendly residents and potential pathogens, underscoring that GABA production is a widespread survival strategy in the gut ecosystem rather than a trait of a few “good” microbes.
What This Means for Health and Future Research
Overall, the study shows that the ability of gut bacteria to make GABA is highly sensitive to diet, and that the key limiting nutrients depend on the overall eating pattern: carbon-rich sugars in low-carb settings and nitrogen-rich building blocks in high-carb ones. This does not mean that people should load up on sugar or protein to chase higher GABA levels—high-sugar diets, for instance, are linked to health risks and may worsen mood disorders despite any microbial effects. Instead, the work provides a mechanistic map of which nutrients matter most for microbial GABA production and highlights that many gut species, not just classic probiotics, may contribute to this calming chemical. Future lab and clinical studies will be needed to test how these in silico insights translate into real-world changes in gut and brain function, and whether carefully designed diets or supplements can safely harness microbial GABA production to support mental and digestive health.
Citation: Homscheid, A., Moors, K.A., Nap, B. et al. Dietary patterns influence the in silico GABA production capacity of Bifidobacterium adolescentis HD17T2H and other human gut bacteria. Sci Rep 16, 8961 (2026). https://doi.org/10.1038/s41598-026-43006-9
Keywords: gut microbiome, GABA, dietary patterns, Bifidobacterium adolescentis, gut brain axis