Clear Sky Science · en
PGE2 regulates ferroptosis and osteogenesis of MC3T3-E1 cells via NOS2
Why Tooth Straightening Affects Your Bones
Braces and clear aligners do more than move teeth—they ask the bone around each tooth to remodel itself. That slow biological construction work is one reason orthodontic treatment can take years. This study looks inside bone-forming cells to understand how a common inflammatory signal, prostaglandin E2, might both speed movement and quietly weaken new bone, pointing toward future ways to make orthodontic care faster and more stable.
Signals Released When Teeth Are Pushed
When an orthodontist applies force to a tooth, the ligament and bone around the root experience a controlled injury. Cells in this area release chemical messengers, including prostaglandin E2, which help coordinate how bone is broken down on one side of the tooth and rebuilt on the other. The authors focused on bone-forming cells known as osteoblasts, using a standard mouse cell line as a model. They wanted to know how prostaglandin E2 shapes two key outcomes inside these cells: their ability to build mineralized bone and their tendency to undergo a recently discovered, iron-dependent form of cell death called ferroptosis.
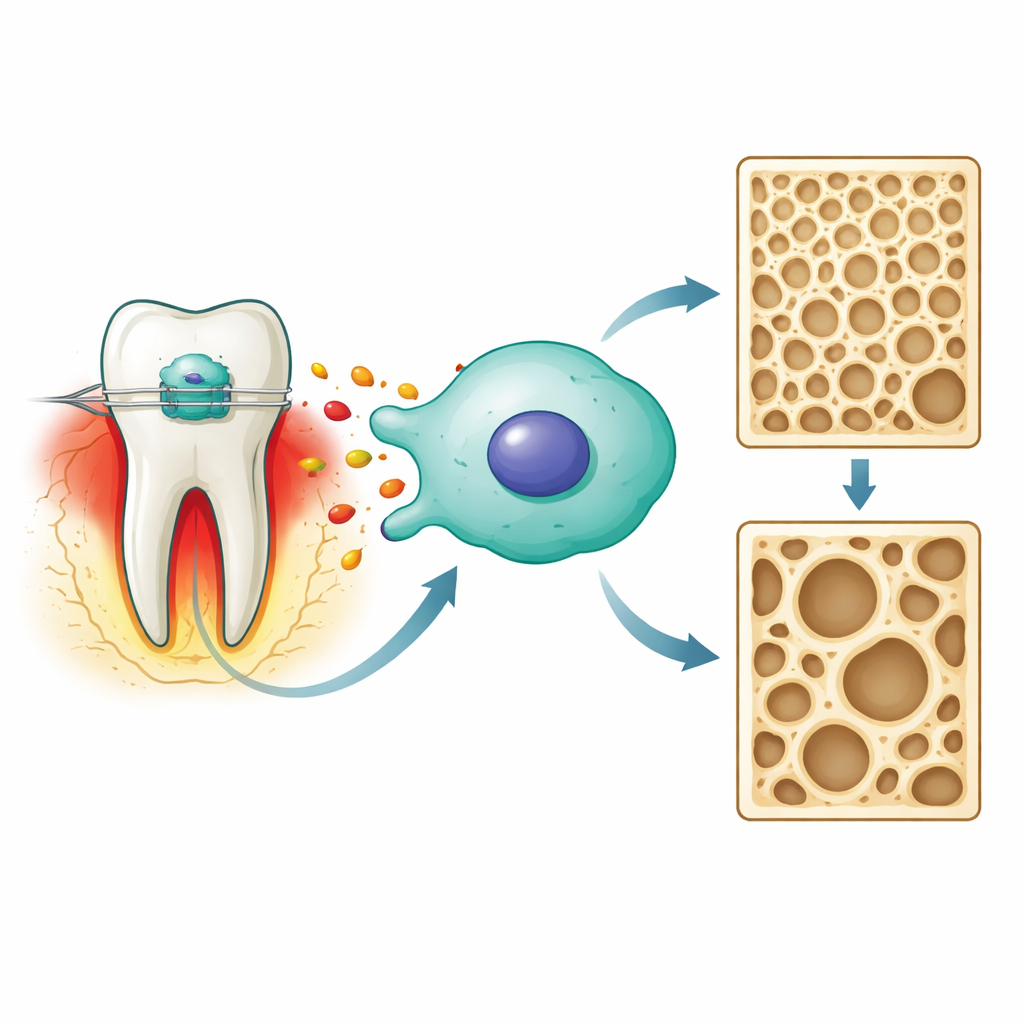
A Stress Pathway Centered on a Single Enzyme
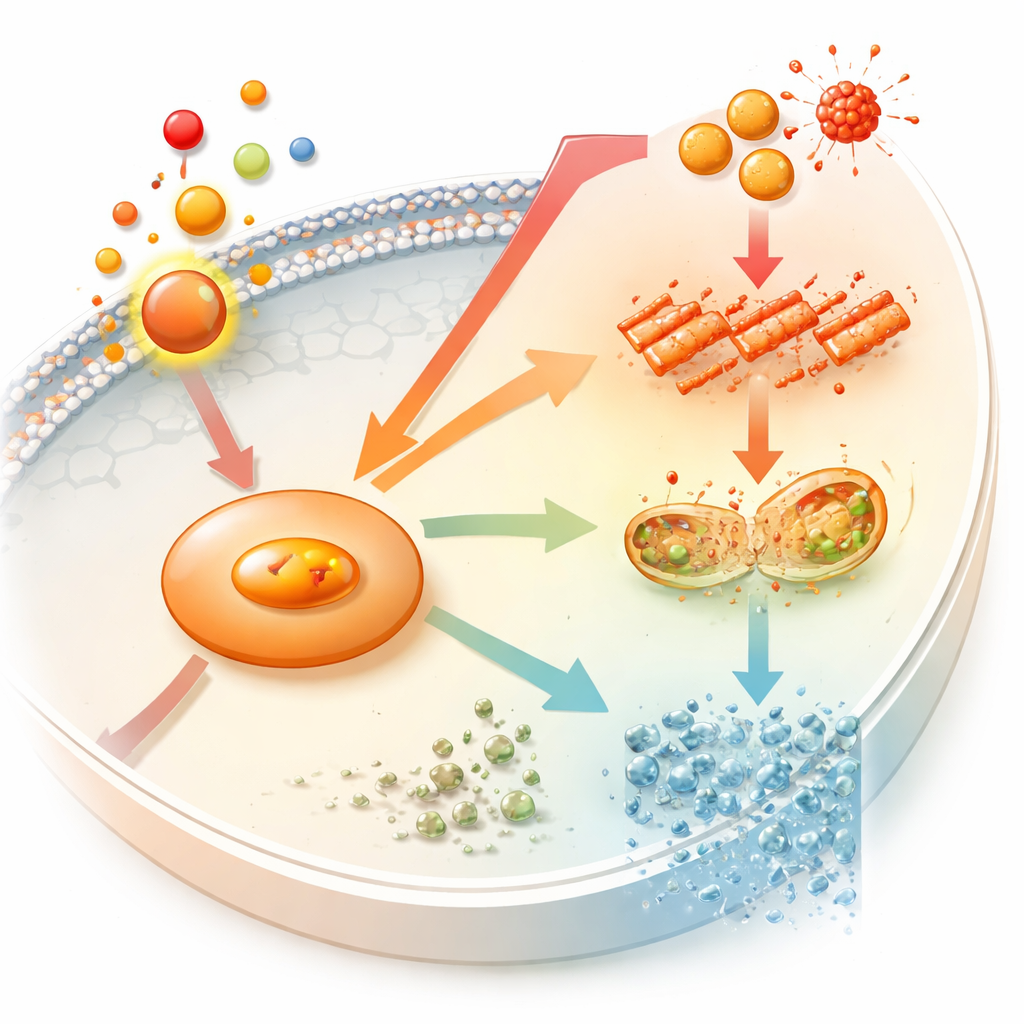
Using gene data from rats undergoing tooth movement, the team first looked for molecules linked both to orthodontic force and to ferroptosis. One candidate, an enzyme called nitric oxide synthase 2 (NOS2), stood out as a central hub. In the dish, exposing osteoblast-like cells to a clinically relevant dose of prostaglandin E2 created a state of “sterile” inflammation—no germs involved, only mechanical stress. Under this signal, NOS2 levels rose, as did other stress markers and damaging reactive oxygen molecules, while protective antioxidant and iron-handling proteins fell. Together, these shifts are hallmarks of ferroptosis beginning inside the cell.
When Bone Builders Switch Off
The same prostaglandin E2 treatment also made the cells worse at acting like bone builders. Classic markers of bone formation dropped, and tests that visualize early bone-like mineral and a key enzyme called alkaline phosphatase showed weaker staining and less mineral deposition. When the researchers added ferrostatin-1, a drug known to block ferroptosis, much of this damage was reversed: oxidative stress eased, ferroptosis signals quieted, and osteogenic (bone-forming) activity rebounded. This suggested that, at least in this model, the inflammatory signal hampers bone formation in part by driving cells toward ferroptosis.
Turning NOS2 Up and Down Like a Switch
To see whether NOS2 is merely a bystander or an active driver, the team genetically dialed its levels up and down. Reducing NOS2 in prostaglandin-treated cells dampened ferroptosis markers, lowered oxidative damage, and restored bone-forming behavior to near-normal levels. Overexpressing NOS2 had the opposite effect: it intensified stress and further suppressed bone-building traits. Strikingly, boosting NOS2 alone—without adding prostaglandin E2—was enough to trigger ferroptosis-like changes and blunt osteogenesis. This shows that NOS2 is not just responding to inflammation; it can itself push osteoblasts away from forming sturdy mineralized bone.
What This Means for Future Braces
Together, these experiments outline a new axis in bone biology during tooth movement: prostaglandin E2 raises NOS2, NOS2 fuels oxidative damage and ferroptosis, and bone-forming capacity declines. For patients, this does not mean braces are unsafe, but it does suggest why treatment can be a delicate balance between moving teeth quickly and preserving strong bone around them. In the long run, drugs or materials that fine-tune NOS2 activity or protect osteoblasts from ferroptosis could help orthodontists shorten treatment times while maintaining, or even improving, the quality and stability of the supporting bone.
Citation: Sun, M., Yang, Y., Pang, L. et al. PGE2 regulates ferroptosis and osteogenesis of MC3T3-E1 cells via NOS2. Sci Rep 16, 8893 (2026). https://doi.org/10.1038/s41598-026-43001-0
Keywords: orthodontic tooth movement, bone remodeling, ferroptosis, nitric oxide synthase, osteoblast function