Clear Sky Science · en
Exploring the dual role of extracellular vesicles in coagulation and immune modulation in glioblastoma
Why blood clots matter in brain cancer
People diagnosed with glioblastoma, the most aggressive form of brain cancer, often face an unexpected danger: dangerous blood clots in the legs or brain. These clots can shorten survival and complicate already demanding treatments. This study asks a simple but crucial question: what is it about glioblastoma cells that makes the blood around them so prone to clotting, and how do tiny particles they shed into the blood also reshape the body’s immune defenses?
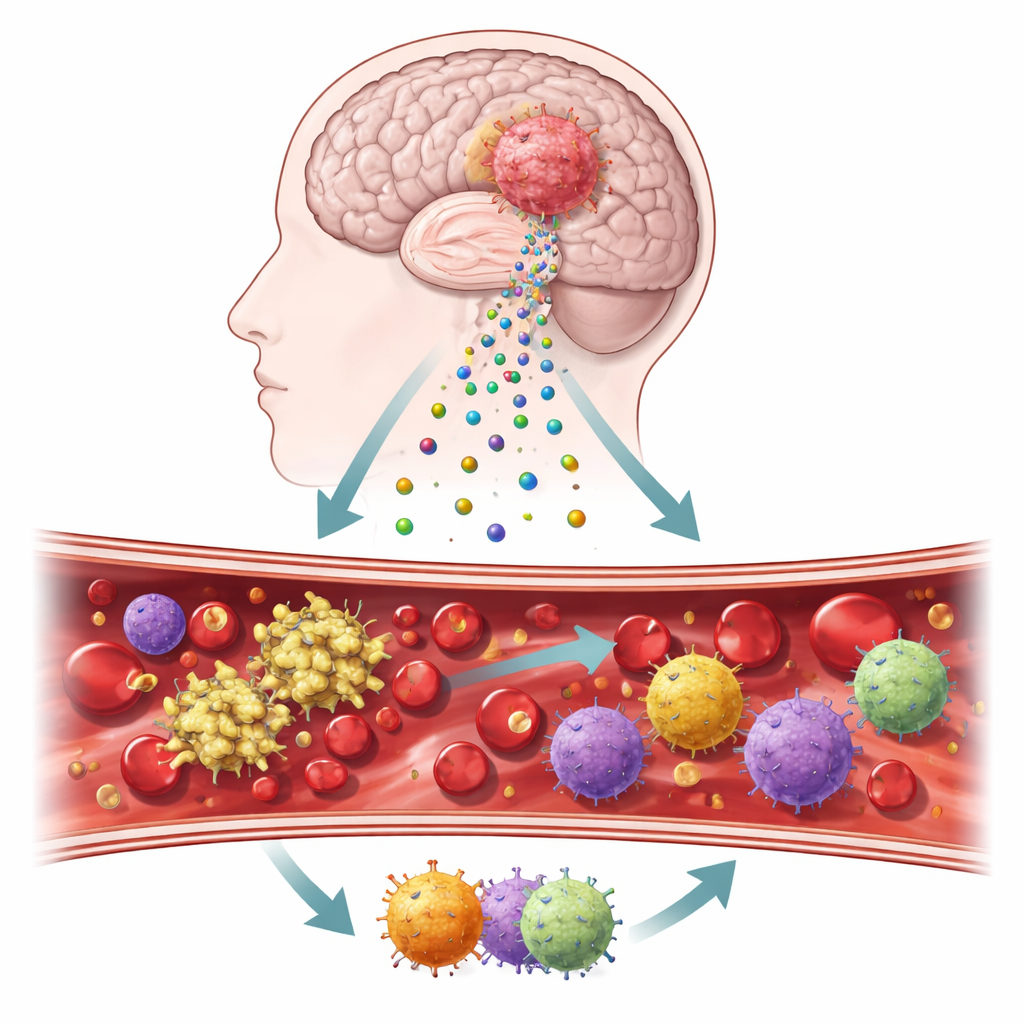
Tiny messengers leaving a brain tumor
The researchers focused on microscopic bubbles called extracellular vesicles, which bud off from tumor cells and float through blood and brain tissue. These vesicles carry on their surface two key proteins, tissue factor and podoplanin, that are known to spark clot formation and influence inflammation. Using tumor samples, blood collected during surgery, and cell cultures grown from patients’ tumors, the team built a stepwise workflow to track how these molecules behave in real patients and in controlled laboratory tests. They also compared tumor tissue to normal brain to see how strongly these clot‑linked signals were turned on.
A quiet bloodstream but a primed tumor zone
Standard hospital tests suggested that, before surgery, many patients’ blood looked surprisingly normal: common clotting times and clot firmness values were largely within reference ranges. Yet when the scientists examined the tumors themselves, a different picture emerged. Glioblastoma tissue showed consistently high levels of tissue factor and podoplanin and very low levels of a natural brake called tissue factor pathway inhibitor. Patient‑derived tumor cell lines largely kept this pattern in the dish, confirming that the “clot‑friendly” signature is an intrinsic feature of the cancer cells. When these cells, or the vesicles they released, were mixed with plasma in the lab, clots formed faster, especially through the so‑called extrinsic pathway that relies on tissue factor.
Different tumor cells, different clotting strategies
Not all glioblastomas behaved the same way. Some patient‑derived cell lines had very high tissue factor, others had more podoplanin, and some had little of either. Cells rich in tissue factor were the most potent at shortening clotting time, while podoplanin‑rich cells contributed modestly through effects on platelets. When one such cell line was sorted into subgroups expressing only tissue factor, only podoplanin, both, or neither, the cells did not stay that way for long. Over three weeks, they drifted back toward mixed profiles, revealing a high degree of plasticity. This dynamic reshaping was accompanied by shifts in oxidative stress within the cells, hinting that chemical stress in the tumor may push cells toward more pro‑clotting states.
Vesicles as shapers of brain and blood immunity
The team then zoomed in on how tumor‑derived vesicles influence the brain’s own immune cells, called microglia, and circulating white blood cells and neutrophils. Vesicles from highly pro‑clotting tumor lines were readily taken up by microglia and slowed their migration, while nudging them toward a more inflammatory, “M1‑like” state and signs of cellular aging. In the bloodstream, vesicles rich in tissue factor activated leukocytes, boosted production of reactive oxygen species, and promoted the release of sticky web‑like structures from neutrophils known as extracellular traps. Vesicles carrying podoplanin were less powerful at driving these explosive immune reactions but strongly encouraged a senescence‑like program, in which immune cells stop dividing and change their behavior. Even vesicles lacking both key proteins could still provoke some senescence, implying that other cargo also matters.
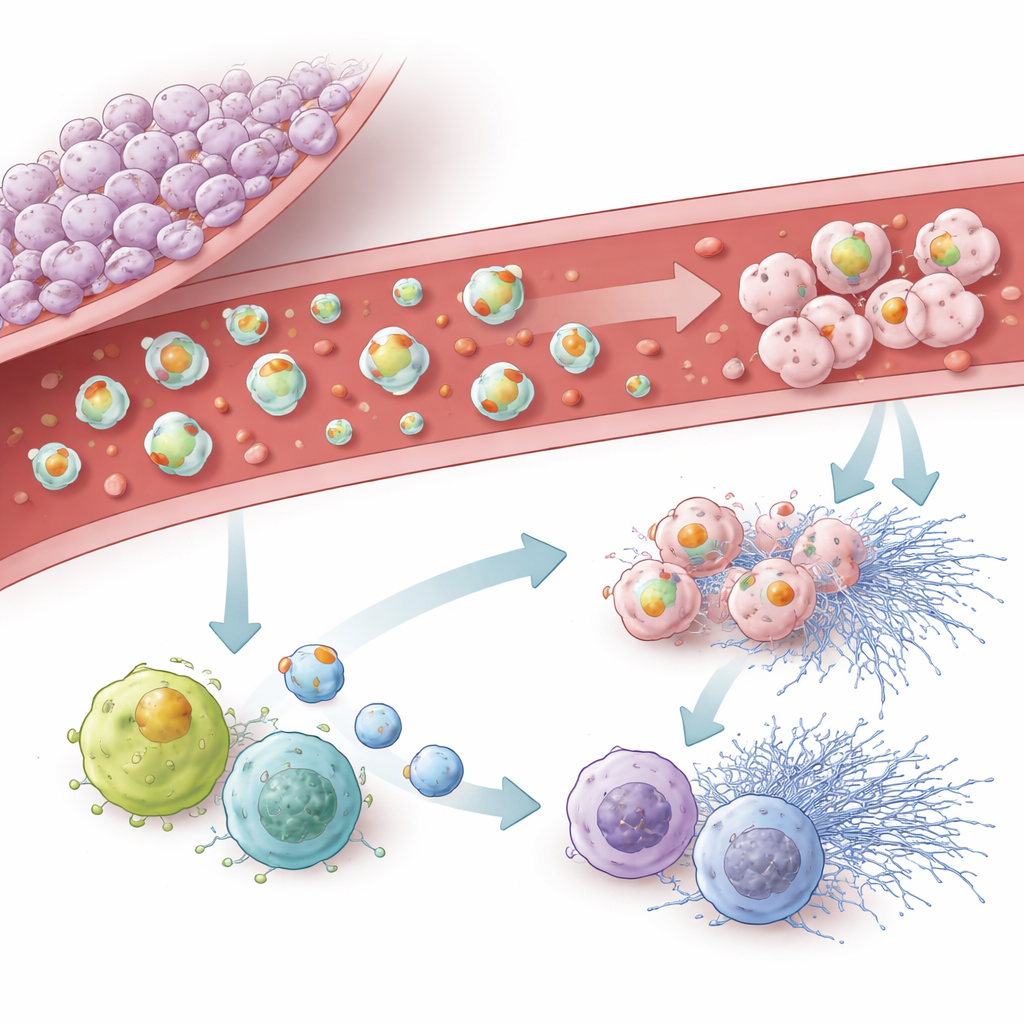
What this means for patients and future care
Put together, the findings show that glioblastoma does not simply thicken the blood everywhere. Instead, the tumor and the vesicles it releases create a localized, highly complex zone where clotting and inflammation feed each other. Tissue factor emerges as the main engine of clot formation and inflammatory activation, while podoplanin adds its own influence on platelets, immune aging, and microglial behavior. Because these vesicles circulate and mirror the state of the tumor, they could serve as future blood‑based markers to identify patients at especially high risk for clots. They may also point toward more tailored prevention strategies that target the tumor’s pro‑clotting machinery without broadly thinning the blood.
Citation: Wolff, A., Waitz, G., Kaps, P. et al. Exploring the dual role of extracellular vesicles in coagulation and immune modulation in glioblastoma. Sci Rep 16, 9534 (2026). https://doi.org/10.1038/s41598-026-42867-4
Keywords: glioblastoma, blood clots, extracellular vesicles, tumor microenvironment, immune response