Clear Sky Science · en
Assessment of Mycobacterium tuberculosis dodecin scaffold as a multimerization platform on the immunogenicity of HPV L2 antigens
Why this research matters for future vaccines
Human papillomaviruses (HPVs) cause nearly all cervical cancers and a growing share of other cancers, yet current vaccines are complex to make and do not fully cover all dangerous virus types. This study explores a new way to build a simpler, more broadly protective HPV vaccine that could be cheaper, more stable, and easier to use worldwide, especially in low- and middle-income countries. By rearranging how key viral protein fragments are displayed to the immune system, the researchers aim to coax the body into making powerful antibodies against many HPV types at once.
A new building block for HPV protection
Today’s HPV shots rely on a viral shell protein called L1, assembled into virus-like particles that strongly stimulate antibodies but mainly against the few HPV types included in each vaccine. The team focused instead on a second viral protein, L2, which contains short regions that are similar across many HPV types and can therefore trigger broader protection. They used a previously designed antigen called Trx-8mer, where small conserved L2 pieces from eight HPV types are stitched together and displayed on a stable carrier protein. Earlier work had already shown that bundling seven of these Trx-8mer units into a ring-shaped complex (a vaccine called PANHPVAX) produces strong, cross-protective antibody responses and is now in early-stage human testing.
Turning a bacterial protein into a vaccine platform
In this study, the researchers tried to go beyond seven copies and pack even more L2 antigens into each particle, on the theory that highly repetitive patterns are particularly good at activating B cells, the antibody-producing cells of the immune system. They chose a tiny, heat-resistant protein from the tuberculosis bacterium, called dodecin, which naturally assembles into hollow balls made of twelve identical subunits. By genetically fusing Trx-8mer to dodecin, they created several new vaccine candidates designed to self-assemble into multimeric nanoparticles displaying many copies of L2. These particles could be made in bacteria, purified at high temperature thanks to their robustness, and verified by multiple biophysical methods to form the intended nanoscale structures.
When direct fusion is not enough
Surprisingly, simply attaching the L2 cargo directly to the dodecin scaffold did not outperform the existing heptameric reference vaccine PANHPVAX in mice. Although the multimeric particles formed as planned and induced neutralizing antibodies against both vaccine and non-vaccine HPV types, the titers were generally lower than those seen with PANHPVAX. Adding a built-in helper T-cell epitope, which sometimes boosts antibody responses, also failed to improve performance. Structural modeling and functional data suggested that bulky, directly fused Trx-8mer units might crowd each other and hide critical L2 regions from B-cell receptors, blunting the immune response even though more antigen copies were present in theory.
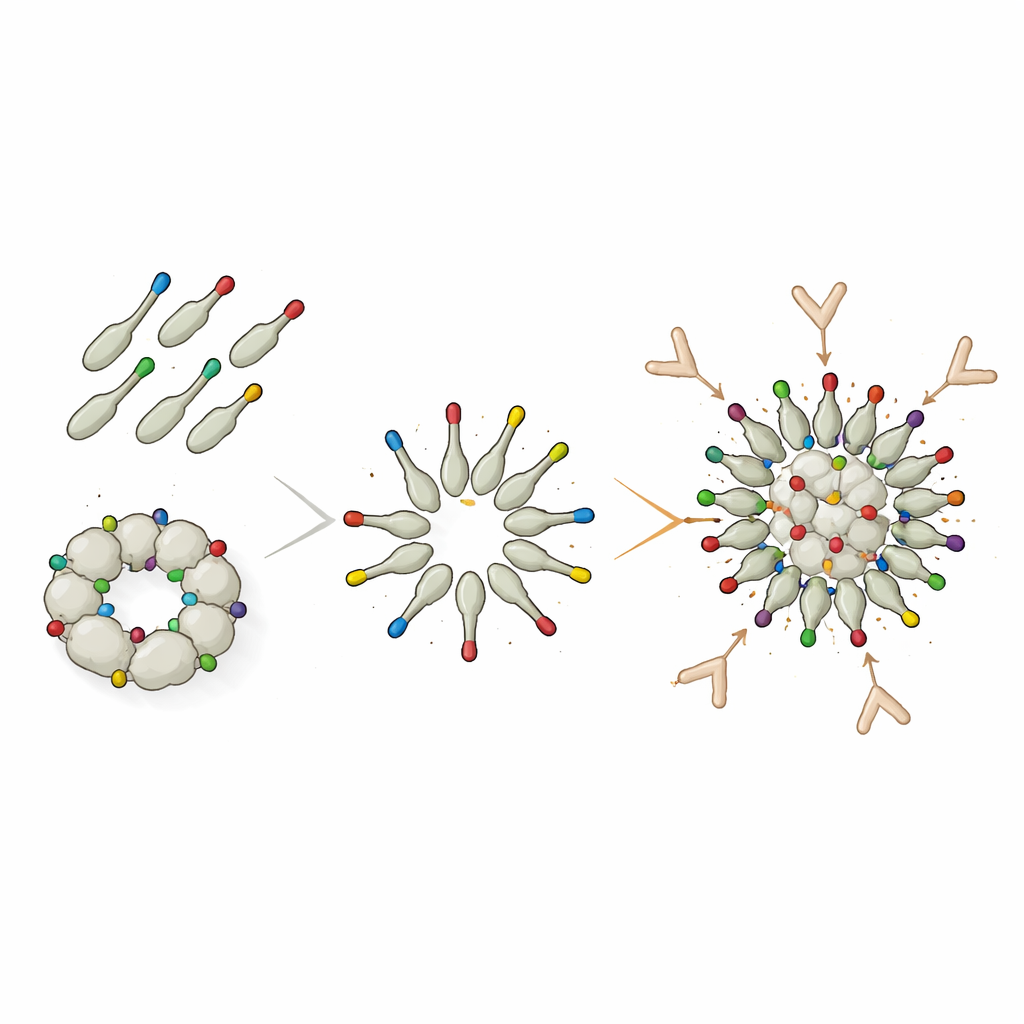
Click-on decoration dramatically boosts responses
To overcome these structural constraints, the team switched to a modular “protein glue” system called DogTag/DogCatcher. In this design, dodecin was produced first with only a tiny DogTag peptide on its surface, allowing it to assemble freely into clean nanoparticles. Separately, the Trx-8mer antigen was fused to the matching DogCatcher partner. When mixed, Tag and Catcher lock together via a spontaneous covalent bond, snapping the L2-bearing Trx-8mer units onto the pre-formed dodecin particles. Even though only about half of the Trx-8mer ended up attached to nanoparticles and the rest remained free in solution, mice immunized with this decorated construct produced much higher neutralizing antibody titers against several high-risk HPV types than with the direct-fusion particles. For some types, such as HPV16, the decorated particles even outperformed PANHPVAX.
Implications for affordable, broad HPV vaccines
Overall, the study shows that how antigens are arranged in space can matter more than how many copies are present on paper. Pre-assembling a sturdy nanoparticle scaffold and then snapping antigens onto it gave the L2 fragments more favorable spacing and orientation, leading to stronger and broader antibody responses in mice. Because the dodecin-based particles are small, highly thermostable, and produced in bacteria, they are promising candidates for next-generation HPV vaccines that could be cheaper to manufacture and less dependent on cold storage. With further refinement and testing, this modular platform could support vaccines that protect against many HPV types in a single, accessible shot, and might even be adapted to combine preventive and therapeutic components in the same nanoparticle.
Citation: Kaplan, E., Mariz, F.C., Zhao, X. et al. Assessment of Mycobacterium tuberculosis dodecin scaffold as a multimerization platform on the immunogenicity of HPV L2 antigens. Sci Rep 16, 9086 (2026). https://doi.org/10.1038/s41598-026-42678-7
Keywords: HPV vaccine, nanoparticle immunogen, L2 antigen, dodecin scaffold, protein conjugation