Clear Sky Science · en
Mapping nocturnal arousal across sleep and pain disorders
Why restless nights matter
Most of us think of sleep as a smooth, quiet state, but under the surface the brain is constantly tuning how deeply we rest. Tiny bursts of activity—brief "stirrings" that don’t fully wake us—help keep sleep stable. This study asks how those hidden stirrings unfold across the night in several conditions linked to disturbed sleep: a dream‑enacting movement disorder, narcolepsy, night terrors and sleepwalking, and the chronic pain condition fibromyalgia. By following these moment‑to‑moment changes instead of just counting hours in each sleep stage, the authors hope to reveal patterns that could one day guide diagnosis and treatment.
The hidden rhythm inside deep sleep
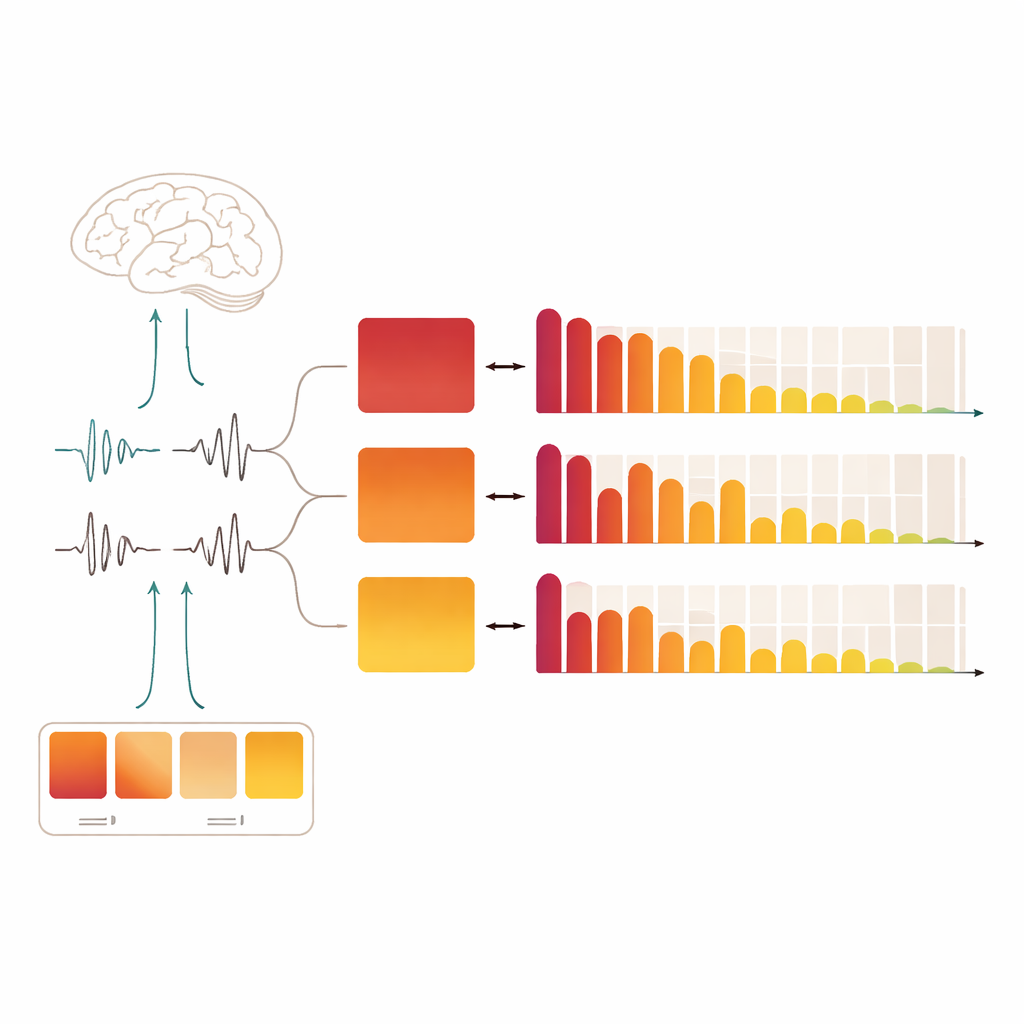
During non‑rapid eye movement (non‑REM) sleep, the brain alternates between quieter periods and short flares of activity. Sleep researchers call this the cyclic alternating pattern: brief "A‑phases" of activation followed by calmer "B‑phases." Within the A‑phases, three flavors appear. The first subtype, A1, is strongly tied to slow, high‑amplitude brain waves and is thought to help keep deep sleep stable. The second and third, A2 and A3, reflect progressively stronger activation and move the brain closer to waking. Rather than treating these events as random noise, recent work views them as a key part of how the brain balances staying asleep with staying responsive to the world.
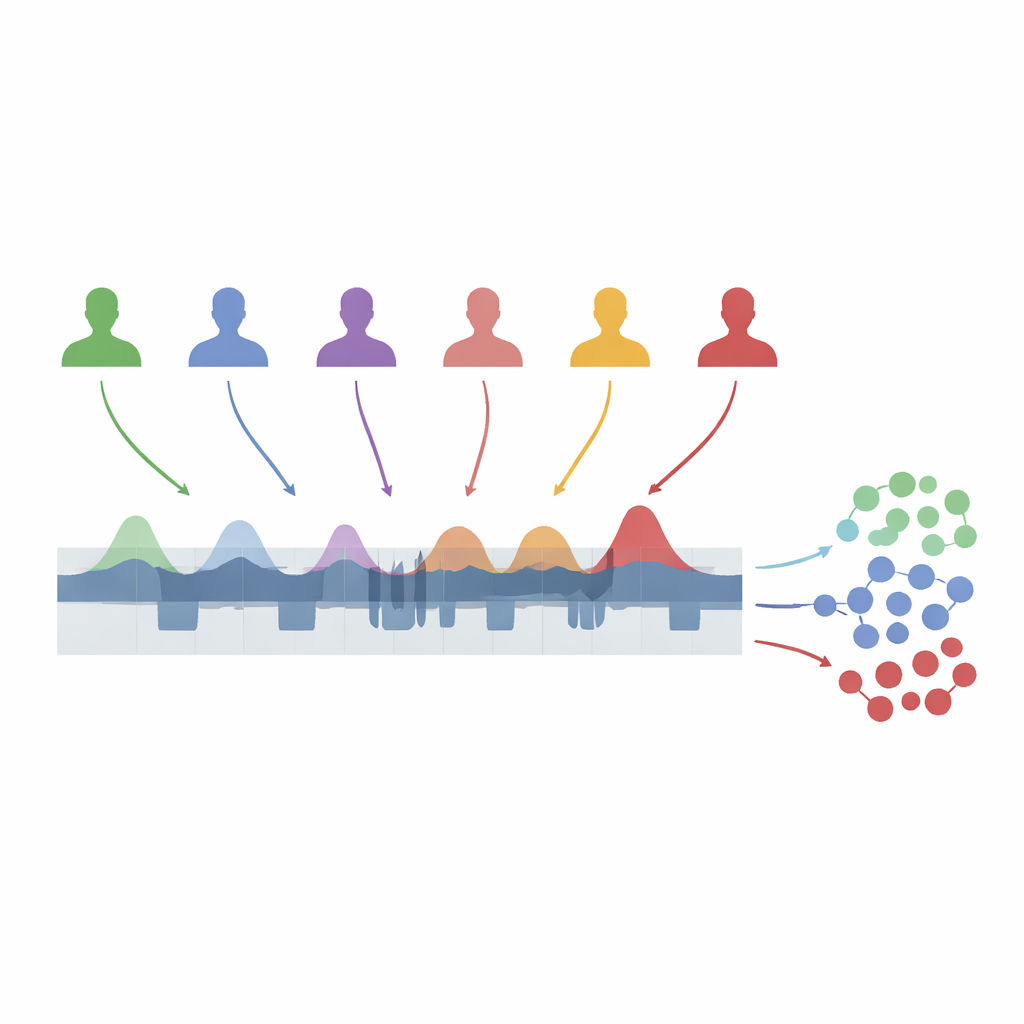
A new way to track night‑long arousal
The team analyzed overnight brain‑wave recordings from 109 adults: healthy volunteers and patients with idiopathic REM sleep behavior disorder, narcolepsy type 1, non‑REM parasomnias, and fibromyalgia. Using an automated algorithm trained on expert‑labeled data, they detected each moment when the brain entered A1, A2, or A3 during non‑REM sleep. They then divided each person’s night into one‑minute chunks and calculated, for each chunk, what fraction of time was spent in each subtype. This "A‑phase index" gives a running measure of how much the brain is being briefly activated, without trying to reconstruct every full cycle of activation and quiet. To compare people with very different bedtimes and sleep lengths, the authors stretched each night onto a standard scale from lights‑out to lights‑on.
How different disorders reshape a night’s journey
In healthy sleepers, A1 events were most common early in the night, when sleep pressure is high, and then steadily faded toward morning. This fits with the idea that strong, slow waves help stabilize the first cycles of deep sleep and then are needed less as the brain recovers. Across all participant groups, A2 and A3 were less prominent and changed more modestly over time. In contrast, all four clinical conditions showed a blunted version of the healthy A1 pattern. People with REM sleep behavior disorder and those with narcolepsy had noticeably fewer A1 and A2 events in the mid‑depth stage of non‑REM sleep, and those with REM sleep behavior disorder also lacked A1 in the deepest stage. Fibromyalgia showed a broad drop in both A1 and A2, especially in this mid‑depth stage. Non‑REM parasomnias stood out: they showed an early‑night spike in A1 and A2 during light sleep, but later a reduction of A1 in deep sleep. Together, these profiles suggest that each disorder has its own "fingerprint" of how the brain’s brief arousal bursts are woven through the night.
What the patterns might mean for the brain
These disorder‑specific fingerprints point toward different kinds of imbalance between systems that promote deep, restorative slow waves and systems that push the brain toward arousal. Reduced A1 and A2 in REM sleep behavior disorder and narcolepsy may reflect weaker recruitment of stabilizing slow‑wave responses, leaving sleep more fragile even when total deep sleep time looks normal. In parasomnias, the mix of extra activation in light sleep and weaker buffering in deep sleep fits with the idea of unstable boundaries between sleeping and waking states, which can give rise to dramatic behaviors such as sleepwalking. In fibromyalgia, widespread A1 and A2 loss may help explain why sleep can feel unrefreshing despite apparently normal amounts of deep sleep: the fine‑grained, protective microstructure is altered, potentially heightening sensitivity to pain and other sensations.
From descriptive maps to future sleep tools
The authors stress that their work is exploratory: group sizes were modest, healthy volunteers were not perfectly matched to patients, and they focused only on the active A‑phases, not the full cycles that include the quieter B‑phases. Still, the results show that following the rise and fall of brief arousal events across the night can reveal distinctive signatures of different sleep and pain disorders. If confirmed in larger, prospective studies, this kind of night‑long mapping could contribute to new biomarkers—objective measures that help classify patients, track disease progression, and guide treatments that tune the balance between stability and arousal in sleep rather than simply knocking people out.
Citation: Biabani, N., Mendonça, F., Mutti, C. et al. Mapping nocturnal arousal across sleep and pain disorders. Sci Rep 16, 8668 (2026). https://doi.org/10.1038/s41598-026-42639-0
Keywords: sleep microstructure, nocturnal arousal, cyclic alternating pattern, sleep disorders, fibromyalgia and sleep