Clear Sky Science · en
Multi-omics analysis of NEDD1 in hepatocellular carcinoma: biological function, prognostic value, and clinical significance
Why this matters for patients and families
Most liver cancers are discovered late, when treatment options are limited and survival is poor. Doctors urgently need better warning signs in the blood or tissue to catch tumors earlier and to choose the right therapy for each person. This study focuses on a little-known protein called NEDD1 and asks a simple question with big consequences: can this molecule help explain why some liver cancers grow faster and shrug off modern immune-based treatments, and could it become a new target for diagnosis and therapy?
A new suspect in liver cancer growth
The researchers began by combing through large public cancer databases and samples from their own hospital to see how much NEDD1 is present in different tissues. They found that NEDD1 levels were higher in many cancer types and especially in hepatocellular carcinoma, the most common form of liver cancer, compared with healthy liver. Patients whose tumors had more NEDD1 tended to live for a shorter time and were more likely to see their disease return or worsen. Statistical models that combined NEDD1 with routine clinical information suggested that this protein carries independent prognostic weight, meaning it may help doctors better predict how an individual’s cancer will behave.
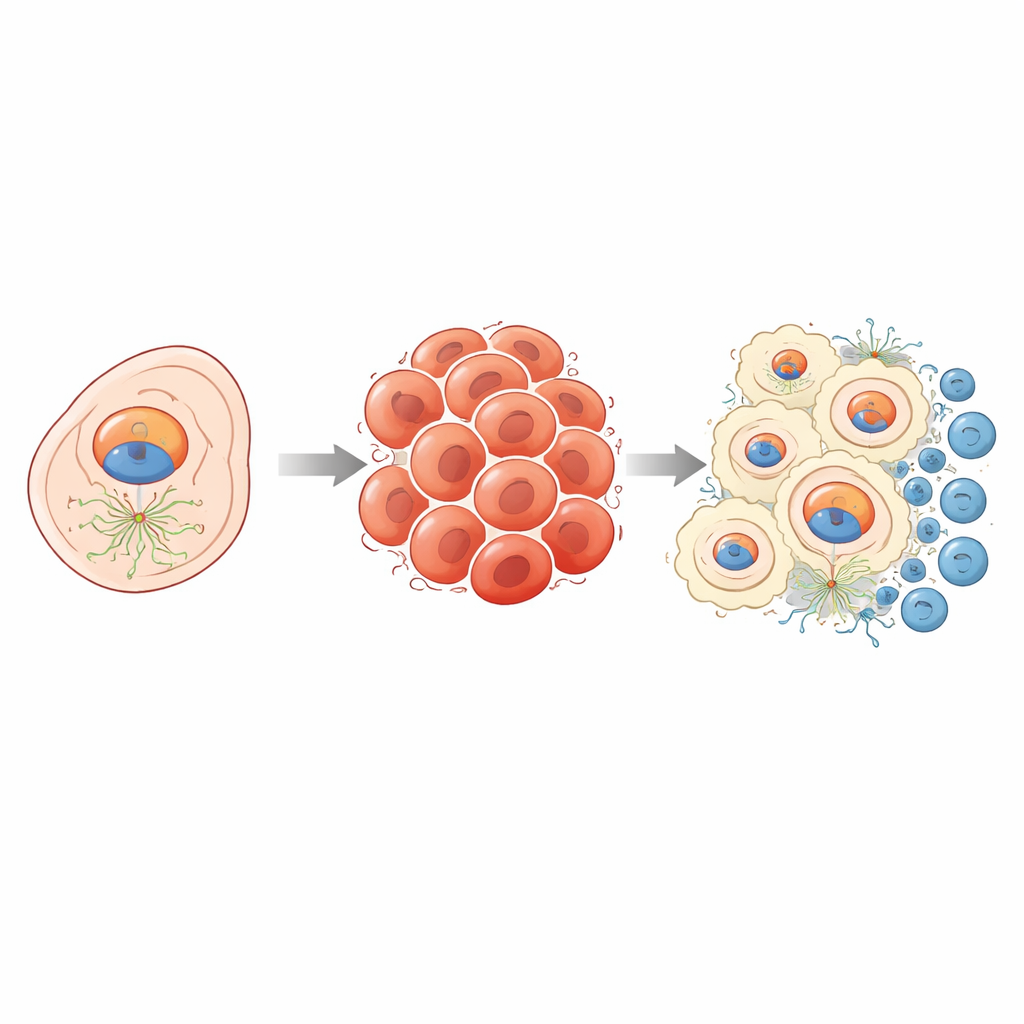
How NEDD1 helps tumors divide and spread
NEDD1 normally helps organize the inner scaffolding that cells use to pull their chromosomes apart during division. When this process is overactive in cancer, cells can multiply unchecked. By comparing tumors with high and low NEDD1 levels, the team found that genes involved in the cell cycle, cell adhesion, and well-known growth pathways were switched on alongside NEDD1. In lab experiments, liver cancer cells made to reduce NEDD1 slowed their growth, formed fewer colonies, and were less able to migrate—behaviors tied to lower aggressiveness. In mice, tumors built from NEDD1-silenced cells grew much more slowly, and tissue staining showed a shift toward a less invasive cell state, with markers of spread and proliferation declining.
Hidden layers of control and treatment clues
The study also explored why NEDD1 becomes overactive. The authors discovered that a regulatory stretch of DNA near the NEDD1 gene was less chemically “methylated” in liver tumors than in normal liver tissue. Such loss of methylation is often linked to genes being switched on too strongly. Patients whose tumors combined low methylation with high NEDD1 had particularly poor outcomes, hinting that this epigenetic change may be one trigger for NEDD1 overdrive. On top of that, specific chemical tags called phosphate groups on the NEDD1 protein were more common in tumors, with one site (s523) standing out as tied to worse prognosis. By screening drug-response databases, the team identified several existing compounds—such as axitinib, topotecan, and pevonidistat—that, at least in computer models, might work better in cancers with high NEDD1 activity.
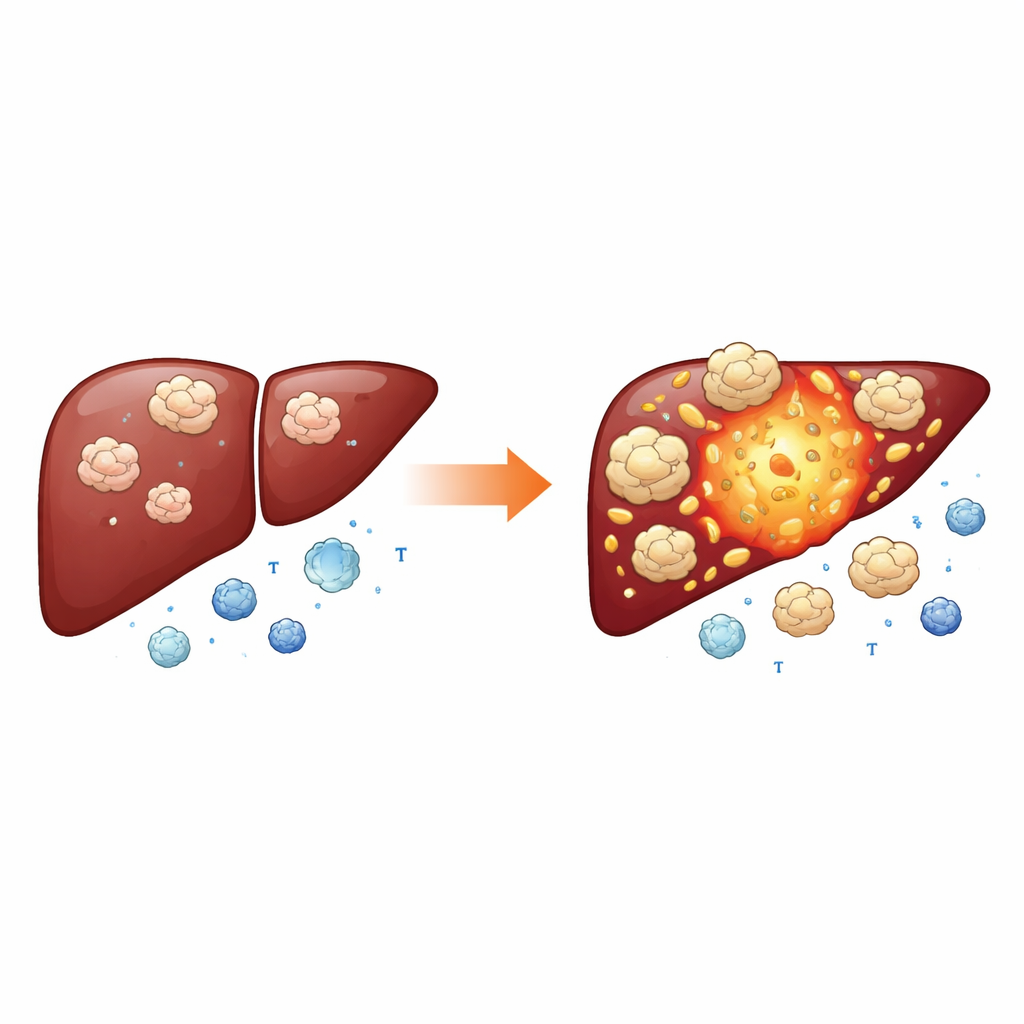
Shaping an immune “safe zone” for the tumor
Modern immunotherapies try to unleash the body’s own T cells against cancer by blocking brakes like PD-1 and CTLA-4. Intriguingly, tumors with more NEDD1 also had higher levels of these immune checkpoint molecules, and patients with low NEDD1 appeared more likely to benefit from checkpoint-blocking drugs. To understand why, the researchers turned to single-cell and spatial gene-mapping data, which reveal which cells in a tumor carry which genes and where they sit. They found that NEDD1 often acts together with another protein, MZT2B, forming a functional pair that appears not only in tumor cells but also in a special group of macrophages enriched for the molecule APOE. These macrophages have been linked in other cancers to spread and resistance to immunotherapy. The NEDD1–MZT2B pair was sometimes concentrated in macrophage-rich niches, sometimes in tumor-cell clusters, suggesting a flexible module that different cell types can use to support tumor growth and dampen immune attack.
What this could mean for future care
Put simply, this work paints NEDD1 as both a growth engine inside liver cancer cells and a possible architect of an immune shelter around the tumor. Its overactivity, likely driven by changes in DNA marking and protein tagging, is tied to faster disease progression, weaker responses to immunotherapy, and a distinct pattern of cooperation with certain immune cells. While much remains to be tested in larger patient groups and more refined models, NEDD1 now emerges as a promising biomarker to help stratify risk and guide treatment—and as a potential target for new drugs aimed at slowing liver cancer and making immune therapies work better.
Citation: Chen, Y., Wan, Z., Xie, H. et al. Multi-omics analysis of NEDD1 in hepatocellular carcinoma: biological function, prognostic value, and clinical significance. Sci Rep 16, 11383 (2026). https://doi.org/10.1038/s41598-026-42505-z
Keywords: hepatocellular carcinoma, NEDD1, tumor microenvironment, cancer immunotherapy, biomarkers